Hepatitis B virus surface antigen recombinant chimeric protein, preparation method and application thereof
A technology of hepatitis B virus and chimeric protein, which is applied in the field of biomedicine, can solve the problems of low expression of cell lines, high cost, low yield, etc., to increase reactogenicity and immunogenicity, solve the problem of immune evasion, Strong immunogenic effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] The design of embodiment 1 recombinant chimeric protein HBsAg
[0030] Firstly, according to the sequence and epitope information of HBsAg reported at home and abroad, the coding genes of HBsAg S and PreS1 were selected and synthesized.
[0031] S
[0032]ATGGAGAACATCGCCAGTGGCCTGTTAGGTCCTTTACTGGTGCTGCAGGCC
[0033] GGCTTTTTCCTGCTGACCAAGATCCTGACCATCCCGCAGAGCCTGGACAGC
[0034] TGGTGGACCAGCCTGAACTTCTTAGGCGGCACCCCTGTTTGTCTGGGCCA
[0035] GAACAGCCAGAGCCAGATCAGCAGCCACAGTCCGACCTGTTGCCCTCCGA
[0036] TTTGTCCTGGCTATCGCTGGATGTGCCTGCGCCGCTTCATCATCTTCCTGTG
[0037] CATCCTGCTGCTGTGCCTGATTTTCCTGCTGGTTCTGCTGGACTACCAGGGT
[0038] ATGCTGCCTGTGTGCCCTCTGATCCCGGGCAGTAGCACCACCAGTACCGGT
[0039] CCGTGCAAGACCTGCACCACACCTGCCCAGGGCACCAGCATGTTCCCGAG
[0040] CTGCTGCTGCACCAAGCCGACAGACGGCAACTGCACCTGCATTCCGATCC
[0041] CGAGTAGCTGGGCCTTCGCCAAGTACCTGTGGGAATGGGCCAGCGTGCGC
[0042] TTCAGCTGGCTGAGTCTGCTGGCCCCGTTCGTGCAGTGGTTCGTGGGCTTA
[0043] AGCCCGACCGTGTGGCTGAGCGTGATCTGGATGATGTGGTTCTGGGGCCC...
Embodiment 2
[0071] Preparation of embodiment 2 recombinant chimeric protein
[0072] 1. Preparation of HBsAg
[0073] First, in order to facilitate the prokaryotic expression of the fusion peptide, the above amino acid sequence was optimized according to the amino acid sequence of the above protein, and Suzhou Jinweizhi Biotechnology Co., Ltd. was entrusted to synthesize it.
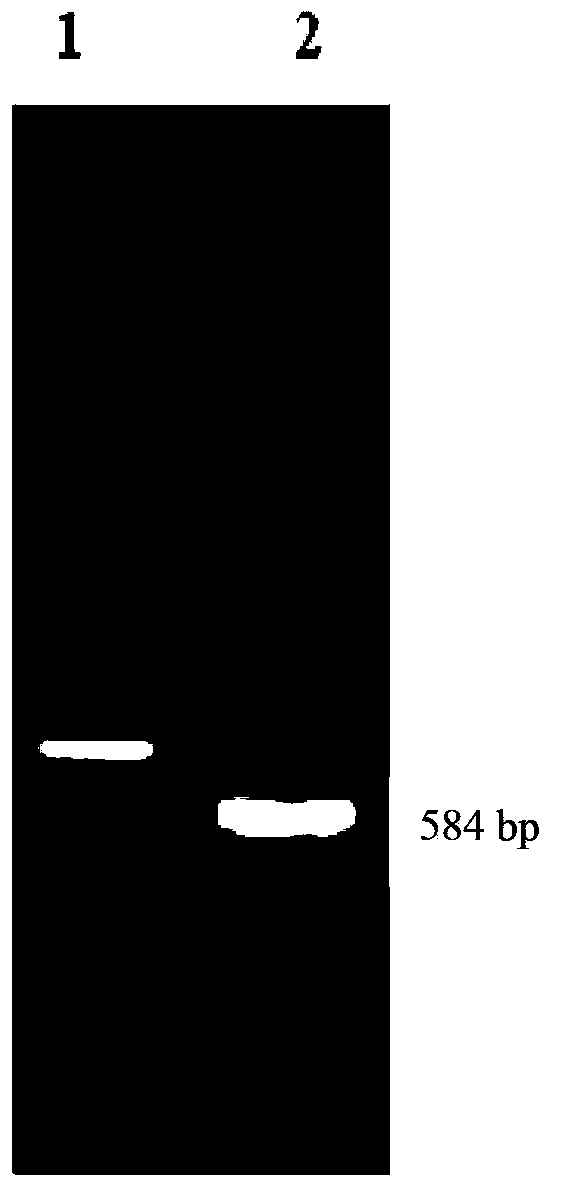
[0074] (1) Using the synthesized S (SEQ ID NO.1) as a template, the upstream primer SP15'-CTAGGATCCATCCTGACCATCCCGCAG-3' (SEQ ID NO.3), the downstream primer SP25'-CCCAAGCTTGAACCACTGCACGAACGG-3' (SEQ ID NO.4) PCR amplification was carried out under the following conditions: pre-denaturation at 94°C for 5 min, denaturation at 94°C for 40 s, annealing at 56°C for 40 s, extension at 72°C for 50 s, 34 cycles, and final extension at 72°C for 10 min. The PCR product (base composition such as SEQ ID NO.2) was detected by agarose gel electrophoresis (see figure 1 ) was double-digested with BamHI and HindIIII, and then com...
Embodiment 4
[0088] The specificity experiment of embodiment 4SS1 colloidal gold immunochromatography test strip
[0089] In order to explore the specificity of the recombinant chimeric protein on the surface of hepatitis B virus developed by the present invention, we will standardize the recombinant chimeric protein solution of the amino acid composition of the present invention such as SEQ ID NO. Test strips (temporarily called "SS1 colloidal gold test strips") were made by conventional methods, and 1,050 clinical samples were tested simultaneously with purchased ELISA kits and hepatitis B colloidal gold test cards. The results showed that SS1 colloidal gold test strips, external Purchased ELISA kits, hepatitis B colloidal gold detection card A, and hepatitis B colloidal gold detection card B detected positive results in 77 cases, 77 cases, 73 cases, and 74 cases, respectively. The re-examination of the test cards by many companies proves that the accuracy rate of the 1050 samples tested...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap