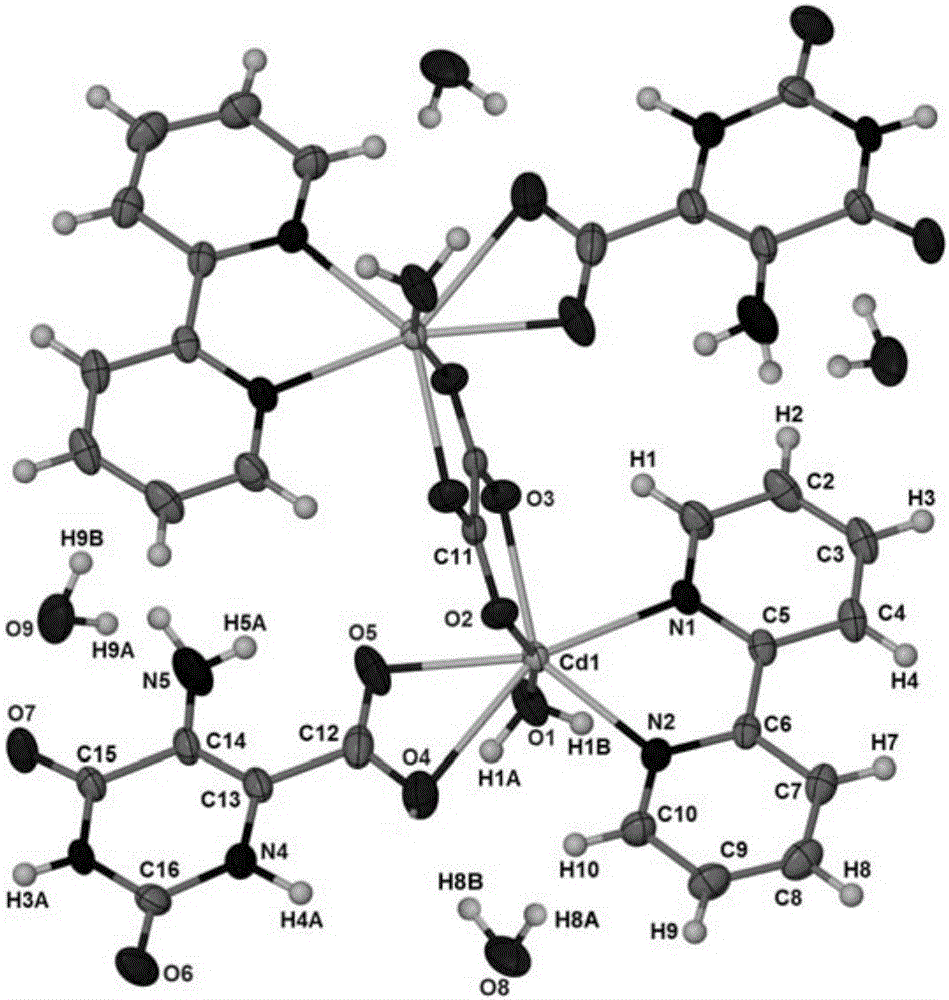
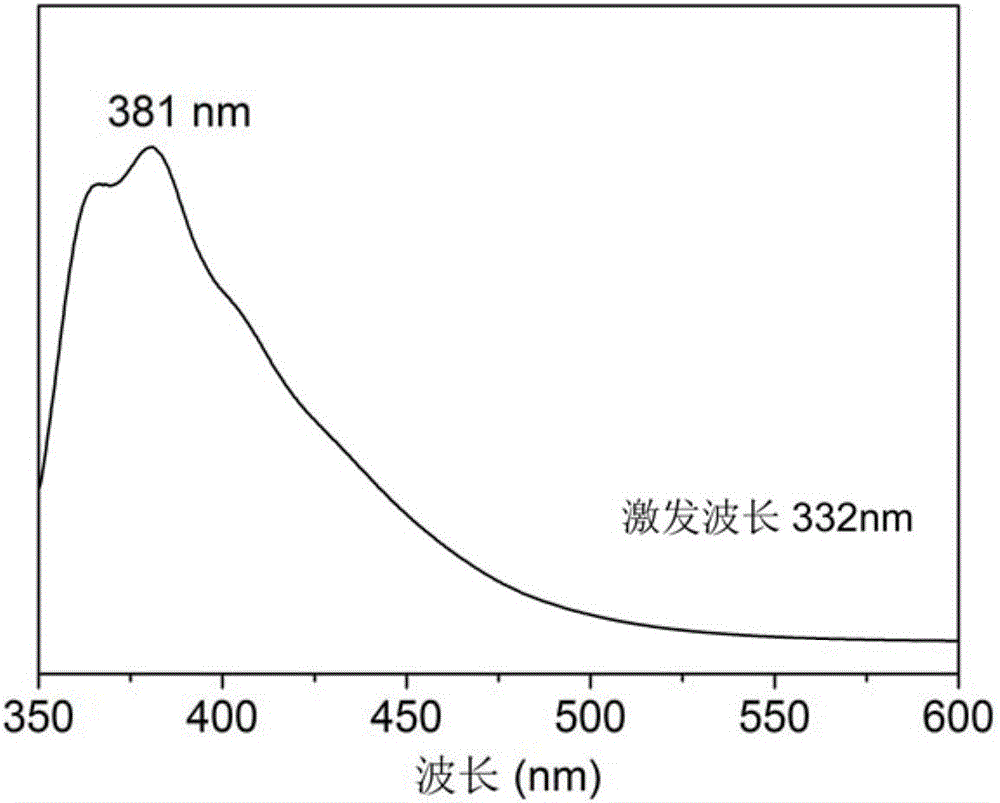
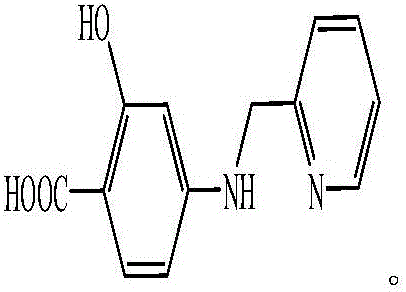
5-aminoorotic acid dinuclear cadmium complex
A technology of aminoorotic acid and cadmium complexes, applied in the field of structural chemistry, to achieve excellent physical and chemical properties and broad application prospects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0019] Weigh CdCl 2 (0.055g, 0.3mmol), 5-aminoorotic acid (0.051g, 0.3mmol), 2,2'-bipyridine (0.047g, 0.3mmol) were added to 15mL of distilled water, stirred at room temperature for 5min, and the reaction mixture Transfer to a high-pressure reaction kettle with a polytetrafluoroethylene liner, seal the reaction kettle, put it in a constant temperature oven at 80 ° C for 5 days, cool it down to room temperature naturally, remove the solution by filtration, and dry it naturally to obtain gray flaky crystals , which is the 5-aminoorotate dinuclear cadmium complex.
Embodiment 2
[0021] Weigh CdSO 4 (0.062g, 0.3mmol), 5-aminoorotic acid (0.051g, 0.3mmol), 2,2'-bipyridine (0.047g, 0.3mmol) were added to 15mL of distilled water, stirred at room temperature for 15min, and the reaction mixture Transfer to a high-pressure reaction kettle lined with polytetrafluoroethylene, seal the reaction kettle, put it in a constant temperature oven at 150 ° C for 2 days, cool it down to room temperature naturally, remove the solution by filtration, and dry it naturally to obtain gray flaky crystals , which is the 5-aminoorotate dinuclear cadmium complex.
Embodiment 3
[0023] Weigh Cd(NO 3 ) 2 (0.024g, 0.1mmol), 5-aminoorotic acid (0.034g, 0.2mmol), 2,2'-bipyridine (0.016g, 0.1mmol) were added to 20mL of distilled water, stirred at room temperature for 10min, and the reaction mixture Transfer to a high-pressure reaction kettle lined with polytetrafluoroethylene, seal the reaction kettle, put it in a constant temperature oven at 120 ° C for 3 days, cool it down to room temperature naturally, remove the solution by filtration, and dry it naturally to obtain gray flaky crystals , which is the 5-aminoorotate dinuclear cadmium complex.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More