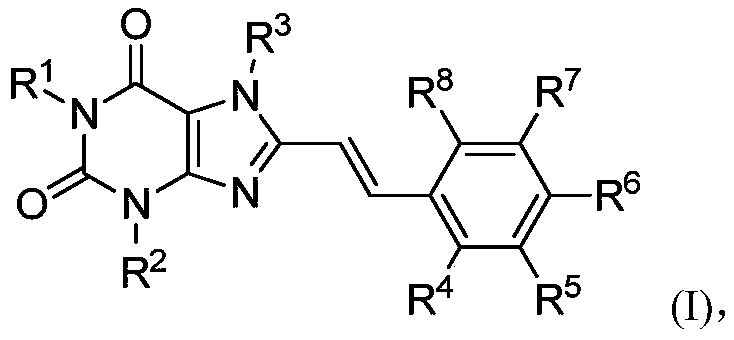
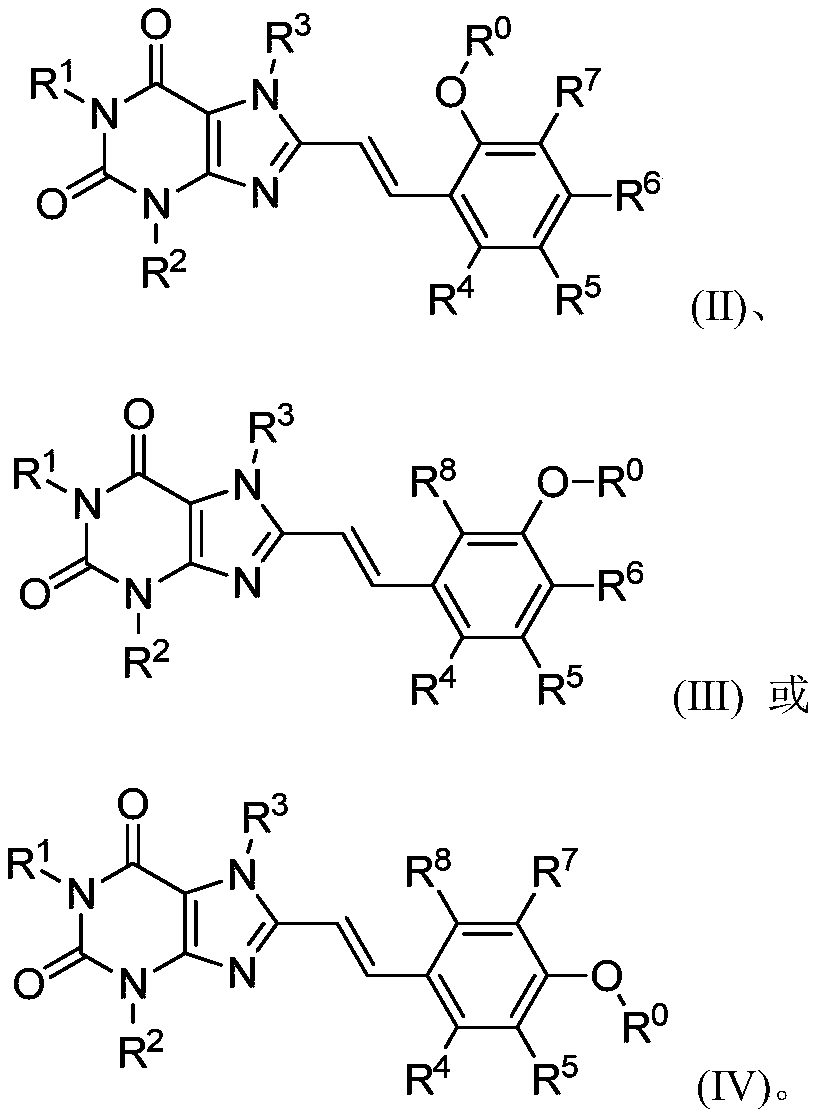
8-substituted styryl xanthine derivative and application thereof
An alkyl and group technology, which is applied in the direction of drug combinations, active ingredients of heterocyclic compounds, cardiovascular system diseases, etc., can solve problems such as distribution restrictions of adenosine A, achieve a good brain/plasma ratio, good clinical application prospects, stable effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment 1
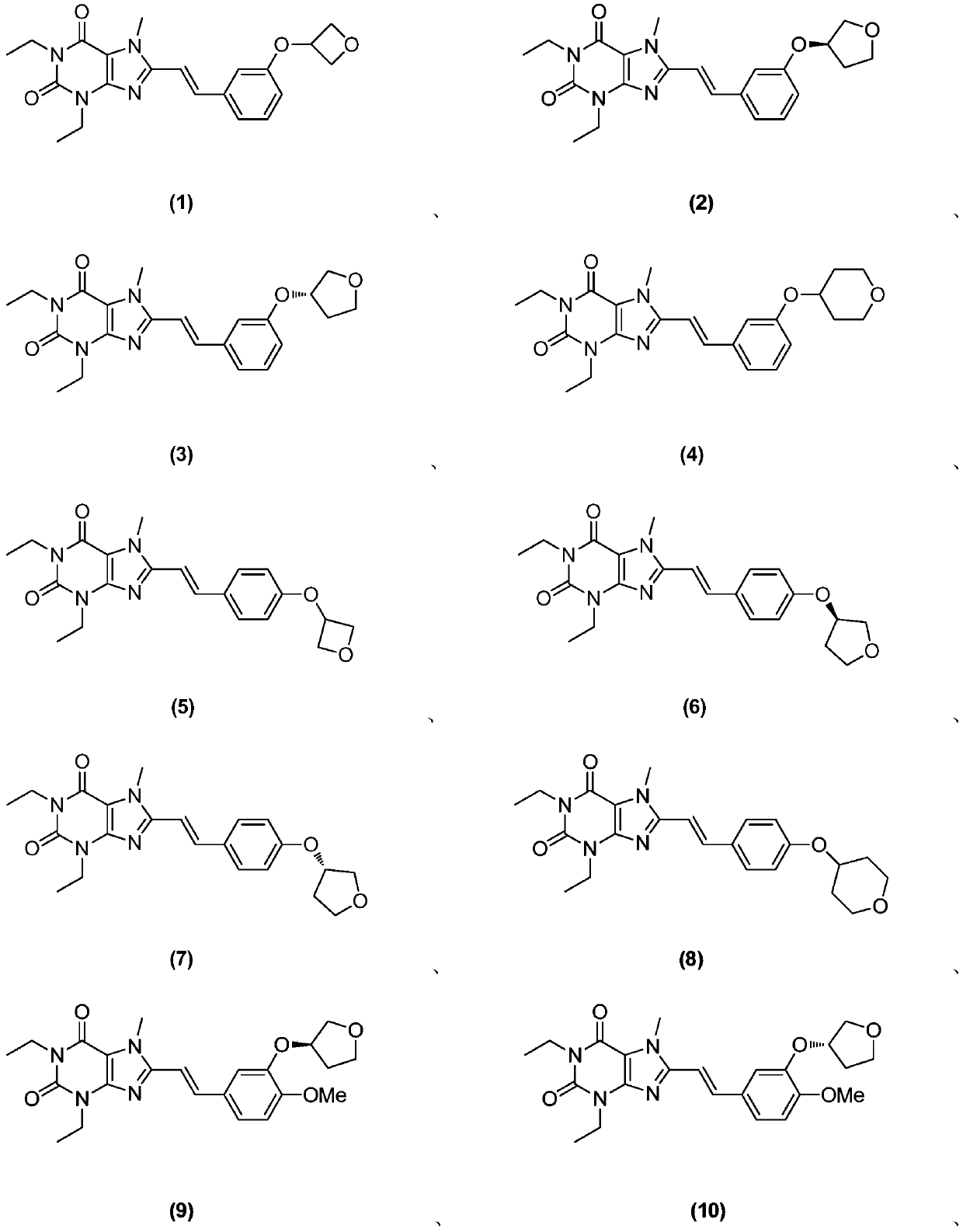
[0269] Example 1 (E)-1,3-diethyl-7-methyl-8-(3-(oxetan-3-yloxy)styryl)-1H-purine-2,6( Synthesis of 3H,7H)-diketones
[0270]
[0271] Step 1) Synthesis of oxetan-3-yl 4-methylbenzenesulfonate
[0272] Add p-toluenesulfonyl chloride (1.0g, 5.2mmol) and triethylamine (2.2mL, 15.7mmol) into a 100mL single-necked flask at 25°C, add dichloromethane (10mL), and then add oxetane in batches Alkan-3-ol (0.5g, 6.7mmol), continue to react for 24 hours; add water (40mL), then add dichloromethane (20mL), separate the liquid, collect the organic phase, spin dry under reduced pressure, and separate by column chromatography Purification (petroleum ether / ethyl acetate (v / v)=10 / 1~5 / 1) gave the title compound as a pale yellow solid (1.02 g, 85.2%).
[0273] MS(ESI,pos.ion)m / z:229.1[M+H] + ;
[0274] 1 H NMR (400MHz, CDCl 3 )δ (ppm) 7.81 (d, J = 8.2Hz, 2H), 7.39 (d, J = 8.1Hz, 2H), 5.38–5.28 (m, 1H), 4.76–4.66 (m, 4H), 2.49 (s ,3H).
[0275] Step 2) Synthesis of 3-(oxetan-3-yloxy)b...
Embodiment 2
[0291] Example 2 (R, E)-1,3-diethyl-7-methyl-8-(3-((tetrahydrofuran-3-yl)oxy)styryl)-1H-purine-2,6 Synthesis of (3H,7H)-diketones
[0292]
[0293] Step 1) Synthesis of (S)-tetrahydrofuran-3-yl 4-methylbenzenesulfonate
[0294] The title compound of this step was prepared by referring to the method described in step 1 of Example 1, that is, p-toluenesulfonyl chloride (3.6g, 18.9mmol), (S)-tetrahydrofuran-3-ol (1.5g, 17.0mmol) and triethylamine (3.5mL, 24.9mmol) was prepared by reaction in dichloromethane (20mL). The crude product was separated and purified by silica gel column chromatography (petroleum ether / ethyl acetate (v / v)=20 / 1) to obtain the title compound as red Brown oil (2.8 g, 68.0%).
[0295] MS(ESI,pos.ion)m / z:265.0[M+Na] + ;
[0296]1 H NMR (400MHz, CDCl3) δ (ppm) 7.80 (d, J = 8.3Hz, 2H), 7.36 (d, J = 8.1Hz, 2H), 5.17–5.06 (m, 1H), 3.92–3.78 (m, 4H), 2.46(s,3H), 2.13–2.04(m,2H).
[0297] Step 2) Synthesis of (R)-3-((tetrahydrofuran-3-yl)oxy)benzaldehy...
Embodiment 3
[0315] Example 3 (S, E)-1,3-diethyl-7-methyl-8-(3-((tetrahydrofuran-3-yl)oxy)styryl)-1H-purine-2,6 Synthesis of (3H,7H)-diketones
[0316]
[0317] Step 1) Synthesis of (R)-tetrahydrofuran-3-yl 4-methylbenzenesulfonate
[0318] The title compound of this step was prepared by referring to the method described in Step 1 of Example 1, that is, p-toluenesulfonyl chloride (3.0 g, 16 mmol), (R)-tetrahydrofuran-3-ol (1.5 g, 17.0 mmol) and triethylamine ( 3.5mL, 24.9mmol) was prepared by reaction in dichloromethane (20mL). The crude product was separated and purified by silica gel column chromatography (petroleum ether / ethyl acetate (v / v)=20 / 1) to obtain the title compound as reddish brown Oil (2.2 g, 58.0%).
[0319] MS(ESI,pos.ion)m / z:265.2[M+Na] + ;
[0320] 1 H NMR (400MHz, CDCl 3 )δ (ppm) 7.80 (d, J = 8.3Hz, 2H), 7.36 (d, J = 8.1Hz, 2H), 5.17–5.06 (m, 1H), 3.89–3.81 (m, 4H), 2.46 (s ,3H),2.13–2.04(m,2H).
[0321] Step 2) Synthesis of (S)-3-((tetrahydrofuran-3-yl)ox...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More