Oral, nose and throat two-component spray with antibacterial and antiviral functions and preparation method of spray
An anti-viral, two-component technology, applied in the direction of anti-viral agents, medical preparations with non-active ingredients, anti-bacterial drugs, etc., can solve the problems of mask shortage and increase the risk of infection, and achieve good biocompatibility, The effect of good antibacterial properties
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
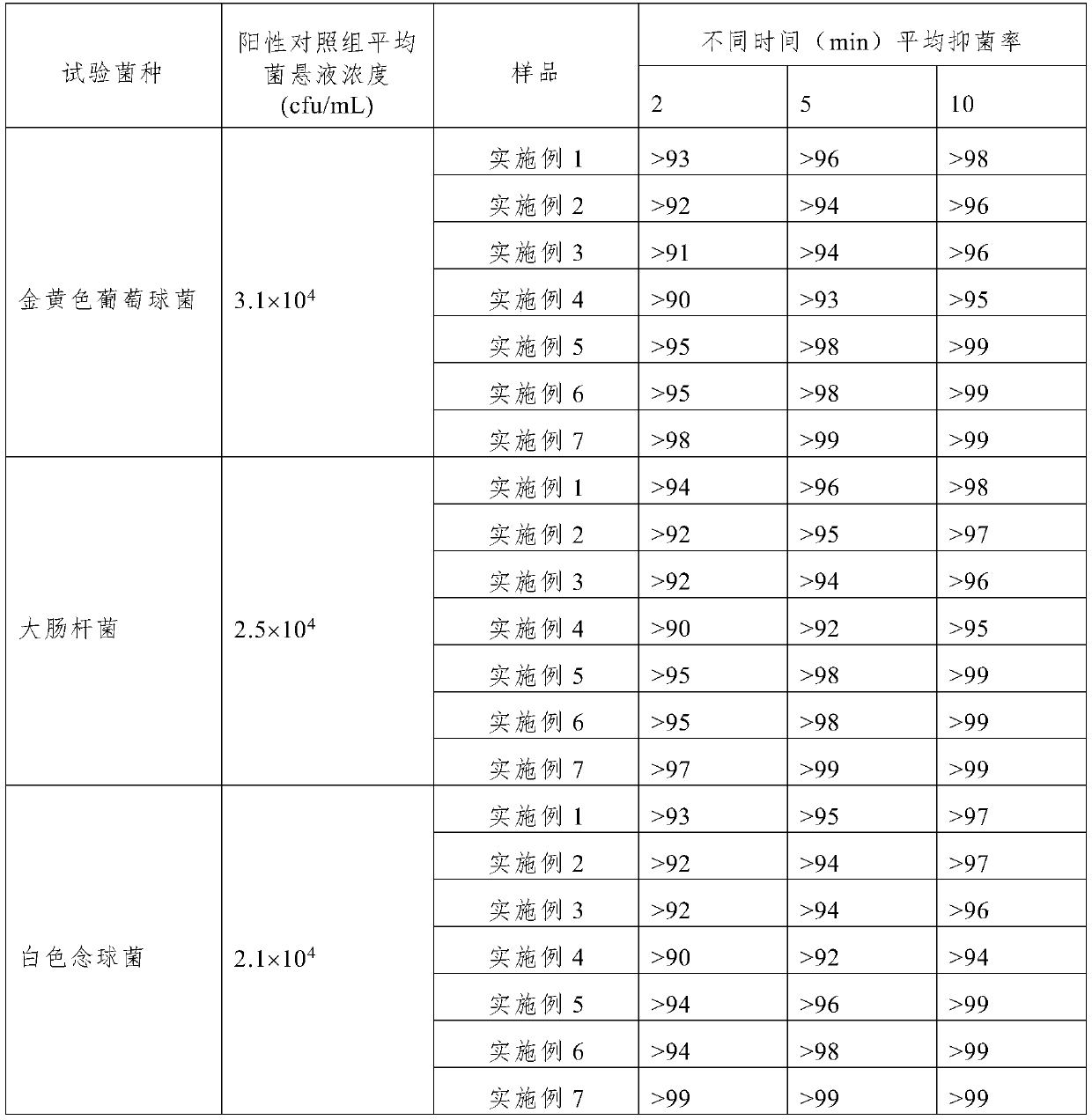
Embodiment 1
[0038] The present embodiment provides a two-component spray for the nasal cavity, including independently packaged first and second components:
[0039] The composition of the first component: 0.5% chitosan hydrochloride, 2.2% sodium chloride, 0.05% glycerin, pH regulator, purified water.
[0040] The composition of the second component: 0.5% sodium hyaluronate, 2.2% sodium chloride, 0.05% glycerin, pH regulator, purified water.
[0041] Among them, the deacetylation degree range of the water-soluble chitosan used is 85-95%, and when the concentration is 10g / L, the viscosity range is 50-100mpa·s at 20°C, which not only has good water solubility, but also has a high viscosity Suitable, easy to spray and good film forming.
[0042] The two-component spray for the nasal cavity of the present embodiment comprises the following preparation steps:
[0043] Step 1. Under GMP purification conditions, add pure water, sodium chloride and glycerin into a clean container, control the r...
Embodiment 2
[0048] This embodiment provides a two-component spray for oral cavity including the first component and the second component of independent packaging:
[0049] The composition of the first component: 0.2% gelatin, 1% sodium chloride, 1% glycerin, pH regulator, purified water.
[0050] The composition of the second component: 0.2% sodium alginate, 1% sodium chloride, 1% glycerin, pH regulator, purified water.
[0051] The two-component spray for oral cavity of the present embodiment comprises the following preparation steps:
[0052] Step 1. Under GMP purification conditions, add pure water, sodium chloride and glycerin into a clean container, control the reaction temperature at 50-60°C, add gelatin under low-speed stirring, and continue stirring until a transparent and uniform solution is formed;
[0053] Step 2. Under GMP purification conditions, add pure water, sodium chloride and glycerin into another clean container, control the reaction temperature at 50-60°C, add sodium...
Embodiment 3
[0057] This embodiment provides a two-component spray for throat including independently packaged first component and second component:
[0058] The composition of the first component: 0.2% guar gum, 1.1% sodium chloride, 2% glycerin, pH regulator, purified water.
[0059] The composition of the second component: 0.2% sodium alginate, 1.1% sodium chloride, 2% glycerin, pH regulator, purified water.
[0060] The two-component spray for throat of the present embodiment comprises the following preparation steps:
[0061] Step 1. Under GMP purification conditions, add pure water, sodium chloride and glycerin into a clean container, control the reaction temperature at 50-60°C, add guar gum under low-speed stirring, and continue stirring until a transparent and uniform solution is formed;
[0062] Step 2. Under GMP purification conditions, add pure water, sodium chloride and glycerin into another clean container, control the reaction temperature at 50-60°C, add sodium alginate unde...
PUM
| Property | Measurement | Unit |
|---|---|---|
| viscosity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap