Extracted material for forward osmosis, preparation method thereof, and forward-osmosis water desalination system using the same
A forward osmosis and chemical formula technology, applied in the direction of osmosis/dialysis water/sewage treatment, chemical instruments and methods, organic chemistry, etc., can solve the problems of reducing the operating cost of the forward osmosis system, affecting the quality of produced water, etc., and achieve the effect of low efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0089] According to one embodiment of the present disclosure, there is provided a method for preparing an extraction material, comprising: providing a first ionic compound; providing a second ionic compound; and mixing the first ionic compound and the second ionic compound to prepare the extraction material . The molar ratio of the first ionic compound to the second ionic compound is 0.05:0.95-0.95:0.05. For example, it can be about 0.15:0.85-0.85:0.15, about 0.25:0.75-0.75:0.25, about 0.33:0.67-0.67:0.33, about 0.15:0.85, 0.20:0.80, about 0.3:0.7, about 0.4:0.6, about 0.5:0.5, about 0.6:0.4, about 0.7:0.3, about 0.85:0.15, etc., but not limited thereto. The above-mentioned first ionic compound and the above-mentioned second ionic compound have the following chemical formula (I).
[0090] {K[A + (R 1 )(R 2 )(R 3 )] p}(X - ) c (Y - ) d (I)
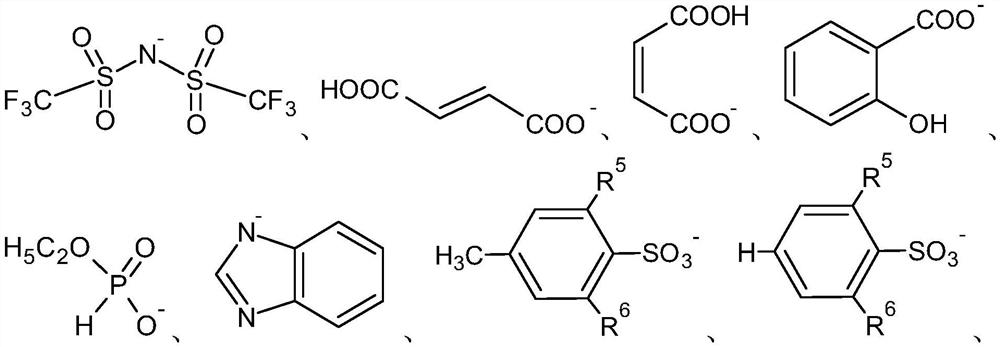
[0091] In formula (I), R 1 -R3 independently including straight or branched C 1-12 Alkyl, A includes phosphorus or nitrogen,...
Embodiment 1
[0118] Preparation of blended extract material
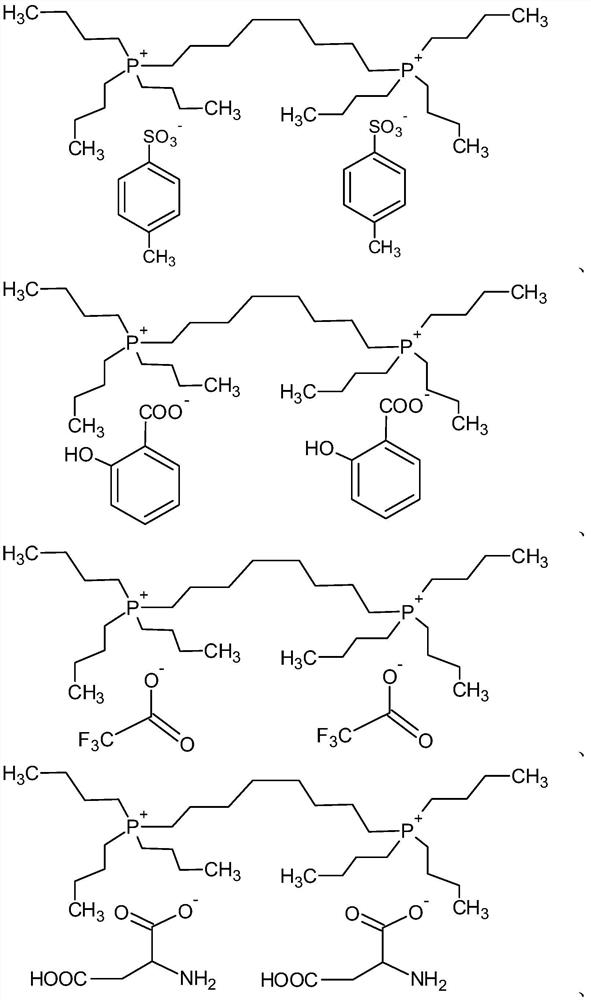
[0119] (1) Preparation of 1,8-octanediyl-bis(tri-n-butylphosphonium)bis(p-toluenesulfonate) (P2-TOS)
[0120] First, 1,8-octanediyl-bis(tri-n-butylphosphonium) dibromide (1,8-octanediyl-bis(tri-n-butylphosphonium) dibromide) (referred to as P2-Br) was synthesized according to the following steps:
[0121] Take a 500mL round bottom reaction flask, put 80g of tributylphosphine (0.4mol) and 48.9g of 1,8-dibromooctane (0.18mol), then add 150mL of acetone , stirred with a magnet at 40°C for 48 hours. After the reaction was over, the above solution was slowly dropped into 1.5L of ether. A white powder solid was obtained by filtration, and washed several times with diethyl ether. The washed white solid was dried under vacuum to obtain 117 g of the product P2-Br.
[0122] Next, the following steps were carried out to synthesize 1,8-octanediyl-bis(tri-n-butylphosphonium)bis(p-toluenesulfonate) (1,8-octanediyl-bis(tri-n-butylphosphoni...
Embodiment 2
[0132] Characterization Verification of Blended Extracted Materials
[0133] For P2-TOS, P2-TMBS, and different blending ratios of P2-TOS and P2-TMBS, the phase separation temperature test and osmotic pressure measurement were carried out respectively. The phase separation temperature test results are shown in Table 2. Table 2 compares the phase transition temperatures of P2-TOS, P2-TMBS, and extraction materials with different blending ratios of P2-TOS and P2-TMBS at different concentrations. The above materials are in a homogeneous phase at room temperature (25° C.), and phase separation begins to occur when the temperature is raised. The material P2-TOS is more hydrophilic than P2-TMBS, and requires a higher temperature to produce phase separation. For the extracted materials with different blending ratios of P2-TOS and P2-TMBS, the phase separation temperature presented is between P2-TOS and Between P2-TMBS, and with the decrease of P2-TOS mixing ratio, the phase separat...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Phase transition temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap