Stable particular pharmaceutical composition of solifenacin or salt thereof
A technology of solifenacin and composition, applied in the field of stable granular pharmaceutical composition of solifenacin or its salt
Inactive Publication Date: 2011-07-06
ASTELLAS PHARMA INC
View PDF3 Cites 3 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0012]On the other hand, Solifenacin and its salts are known to have very high solubility to various solvents and have very strong bitter taste and astringent properties
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
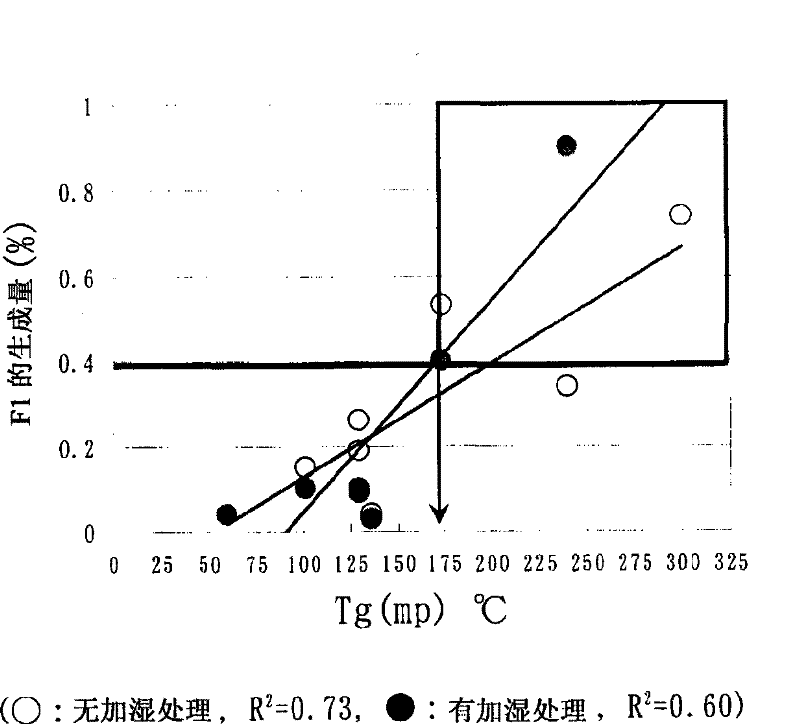
Experimental program
Comparison scheme
Effect test
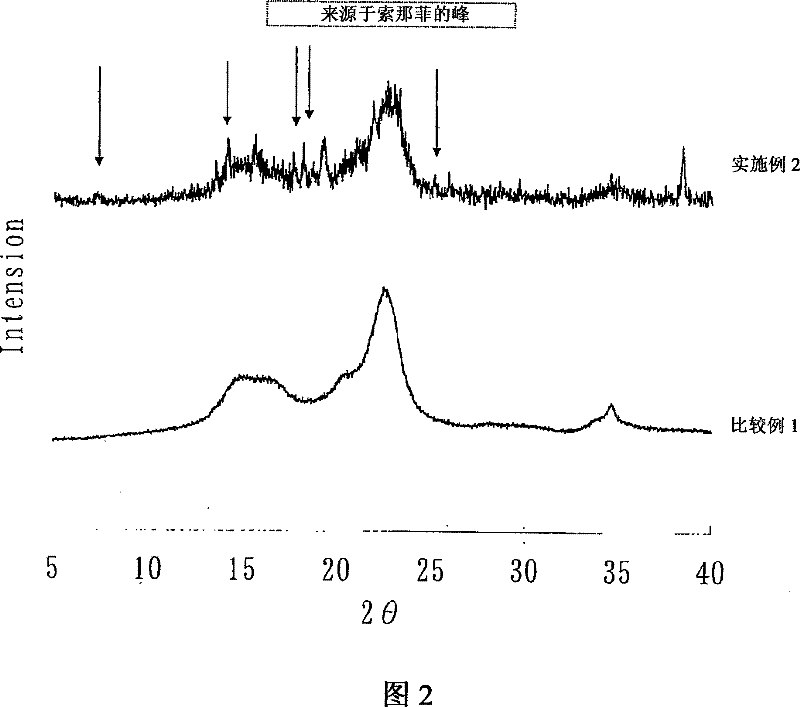
Embodiment 1
[0092] Example 2
[0093] The granular composition obtained in Example 1 was humidified at 25° C. and 75% RH for 12 hours, further dried at 30° C. and 40% RH for 3 hours, and crystallized to obtain the granular composition of the present invention.
Embodiment 2
[0095] Using PEG6000 as a binder, the material obtained by coating solifenacin succinate on crystalline cellulose core particles
Embodiment 3
[0097] Example 4
[0098] The granular composition obtained in Example 3 was humidified at 25° C. and 75% RH for 12 hours, further dried at 30° C. and 40% RH for 3 hours, and crystallized to obtain the granular composition of the present invention.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
A solid pharmaceutical preparation of solifenacin or a salt thereof, the preparation being stable and inhibited from decomposing with time when supplied to clinical fields. In a pharmaceutical preparation containing solifenacin or a salt thereof, the compound in an amorphous form was revealed to be causative of cardinal-drug decomposition with time. The composition for a solid pharmaceutical preparation of solifenacin or a salt thereof contains solifenacin or its salt each in a crystalline form, and the content of the compound in an amorphous form in the composition is within the range which exerts no influence on product stability. Also provided are: a process for producing the composition; and a medicinal composition for solid pharmaceutical preparations which contains solifenacin and an amorphization inhibitor.
Description
technical field [0001] The present invention relates to a stable granular pharmaceutical composition formed by using solifenacin or its salt and a specific binder and a manufacturing method thereof, an orally disintegrating tablet containing the granular pharmaceutical composition, and the granular medicine Method of stabilizing the composition. Background technique [0002] Solifenacin is shown in the following structural formula (I): [0003] [chemical formula 1] [0004] [0005] Structural formula (I) [0006] It is chemically known as 1-phenyl-1,2,3,4-tetrahydro-2-isoquinolinecarboxylic acid (1R,3'R)-quinuclidinyl ester. [0007] It has been reported that a series of quinucidine derivatives including solifenacin or its salts have no effect on muscarine M 3 Receptors have excellent selective antagonism and are useful in urinary disorders (e.g. neurogenic urinary frequency, neurogenic bladder, nocturia, unstable bladder, bladder contractures and chronic cysti...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Patents(China)
IPC IPC(8): A61K31/4725A61K9/14A61K47/26A61K47/32A61K47/34A61K47/36A61K47/38
CPCA61K31/4725A61K9/0056A61K9/1676A61P11/00A61P11/06A61P13/00A61P13/06A61K9/16A61K31/439A61K47/26A61K47/38
Inventor 梅岛启之大井宏志斋藤胜实武谷佑子
Owner ASTELLAS PHARMA INC