Compositions contain (A) haloalkyl-nicotinic
acid derivative(s) (I),
phthalic acid diamide derivative(s) (II) or anthranilamide derivative(s) (III) and (B) safener(s) selected from about 60 specific compounds (e.g. cloquintocet-mexyl, isoxadifen-ethyl, fenclorim or mefenpyr-diethyl) and 4 general classes of
azole,
quinoline, alkanamide or N-(phenylsulfonyl)-
benzamide compounds (IVa)-(IVd). Compositions comprise: (1) at least one haloalkyl-nicotinic
acid derivative of formula (I),
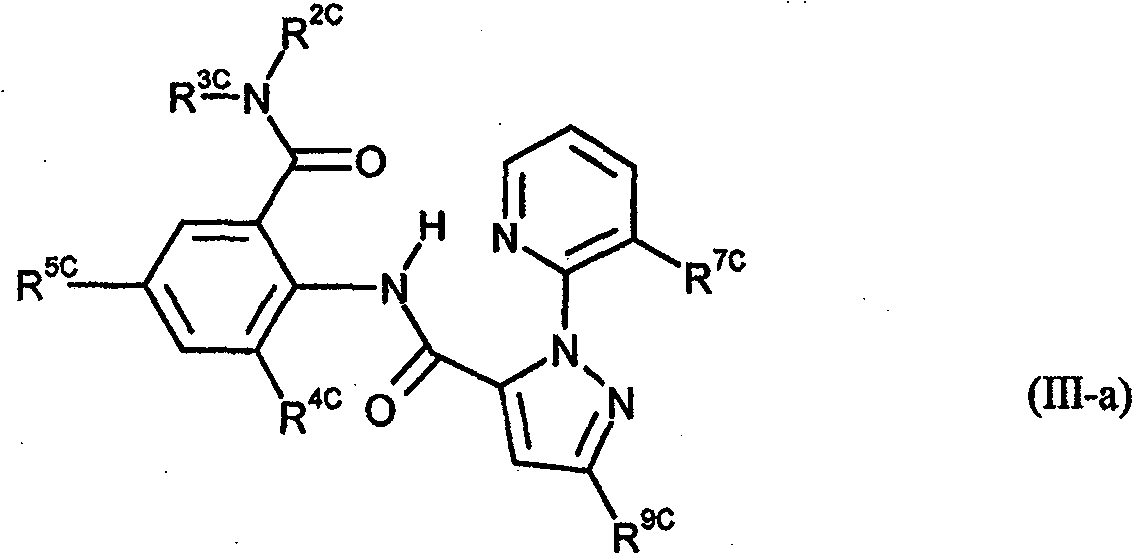
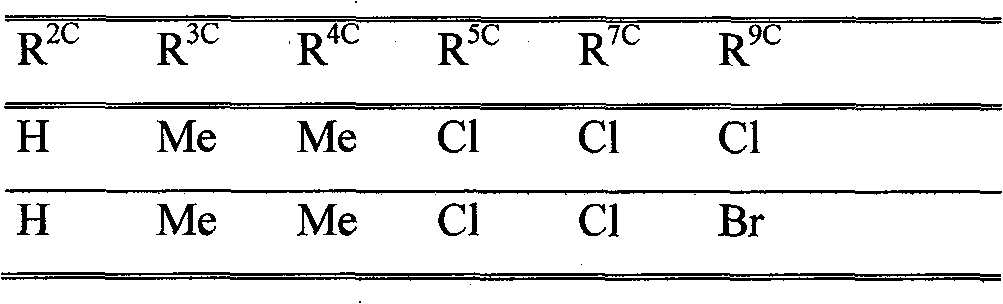
phthalic acid diamide derivative of formula (II) or anthranilamide derivative of formula (III) (optionally in N-
oxide or salt form) as
active agent (A); and (2) at least one safener (B) selected from e.g.
azole derivatives of formula (IV-a),
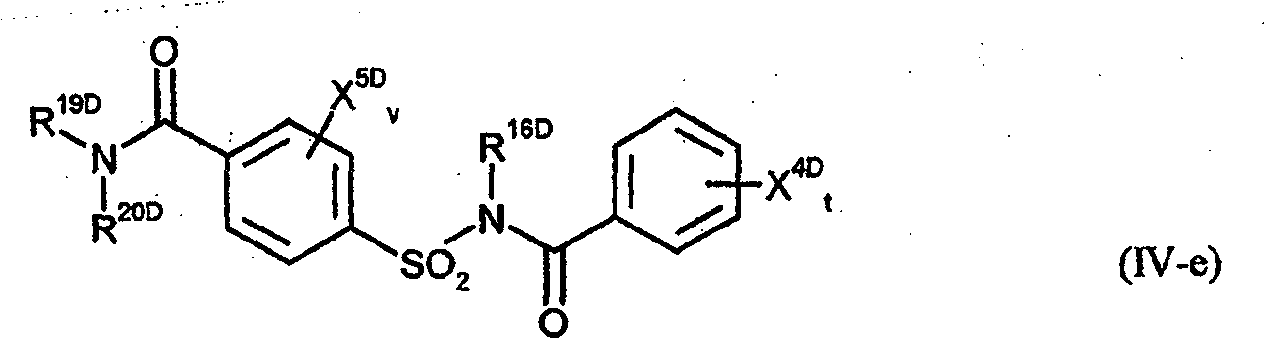
quinoline derivatives of formula (IV-b), alkanamides of formula R 10d>-CO-NR 11d>R 12d> (IV-c) and N-(phenylsulfonyl)-
benzamide compounds of formula (IV-d). A a>C(W a>)-NR 2a>R 3a>, C(W a>)-N=SR 9a>R 10a> or Het; R 1a>haloalkyl; R 2a>, R 3a>e.g. U-cycloalkyl (optionally substituted (os) by R 4a> or Q a>), C(X a>)-Y a>; or CH 2-heterocyclyl (os by R 5a>); or NR 2a>R 3a>e.g. heteroaryl os by R 4a>, U' or Q a>; Q a>hydrazono (os); R 4a>e.g. S(O) n-U'; X a>, W a>O or S; Y a>e.g. R 6a> or NR 7a>R 8a>; R 6a>e.g. -U-cycloalkyl (os); R 7a>e.g. U-cycloalkyl (os); R 8a>e.g.
aryl (os by R 5a>); R 9a>, R 10a>e.g. C(X a>)-Y a>; or R 9a>R 10a>heterocycle; Het : e.g. heterocycle os by R 4a>, U' or Q a>; X b>e.g. haloalkoxy; R 1b>-R 3b>e.g. M 1b>-Q b>; or R 1b>+R 2b>e.g. complete ring; M 1b>e.g. os alkylene; Q b>e.g. T b>-R 4b>; T b>e.g. S(O) m or N(R 5b>); R 4b>e.g. T-heteroaryl (os); R 5b>e.g. CO-O-T-Ph (os); k : 1-4; m : 0-2; L 1b>, L 3b>e.g. S(O) m-U; L 2b>e.g. M 2b>-R 6b>; or L 1b>+L 3b>e.g. complete ring; M 2b>e.g. S(O) m; R 6b>e.g. heteroaryl (os); A 1c>, A 2c>e.g. S; X c>e.g. CR 10c>; R 1c>e.g. V (os by R 6c> or R 11c>); R 2c>e.g. H; R 3c>e.g. R 11c> or heteroaryl (os by 1-3 W c> or R 12c>)); or R 2c>+R 3c>complete ring M c>; R 4c>e.g. OT' (os); R 5c>, R 8c>e.g. G c>, J c> or S(O) p-Ph (all os by e.g. V (os by e.g. Ph (os by W or R 12c>))); G c>e.g. carbocycle; J c>e.g. heteroaryl (os); R 6c>e.g. C(E c>)-R 19c>, OP(Q c>)(OR 19c>) 2, SO 2L c>R 18c> or L c>SO 2L c>R 19c>; E 1c>e.g. NR 19c>; R 7c>e.g. SO 2T'; R 9c>e.g. halo; R 10c>e.g. T'; R 11>e.g. phenylsulfenyl (os by W c>, S(O) n(R 16c>) 2 or S(O) nNR 13c>SO 2L c>R 14c>); L c>e.g. NR 18c>; R 12c>e.g. B(OR 17c>) 2; Q c>O or S; R 13c>e.g. V (os); R 14c>e.g. Cyc (os by e.g. R 6>); R 15c>U' (os by e.g. SOT' or Ph (os by 1-3 W c> or R 12c>); R 16c>e.g. haloalkyl; or N(R 15c>) 2 or N(R 16c>) 2M c>; R 17c>e.g. T; R 18c>e.g. U'; or NR 13c>R 18c>M c>; R 19c>e.g. U (os) M c>e.g. ring W c>e.g. T; n : 0 or 1; p : 0-2; A 12>group of formula (a)-(c); r, s : 0-5; A 11d>e.g. N; A 2d>e.g. alkanediyl (os); R 8d>e.g. NT 2; R 9d>e.g. NT 2; R 10d>T (os); R 11d>, R 12d>e.g. V (os); or R 11d>+R 12d>e.g. alkanediyl; R 13d>, R 15d>e.g. halo; R 14d>e.g. U (os); X 1d>e.g. T'; X 2d>, X 3d>e.g. H; Q : N(R 17d>)-COR 18d> or CO-NR 19d>R 20d>; t : 0-5; v : 0-4; R 16d>, R 17d>e.g. T; R 18d>e.g. NT 2 (os); R 19d>e.g. U (os); R 20d>e.g. phenyl (os by e.g.T' or OT'); or R 19d>+R 20d>e.g. 3-6C alkanediyl ( os); X 4d>, X 5d>e.g. T'; T : 1-4C
alkyl, U : 1-6C
alkyl; T' : T or 1-4C haloalkyl; U' : U or 1-6C haloalkyl; V : 2-6C alkenyl or 2-6C alkynyl; Cyc : 3-6C cycloalkyl; and with provisos. Full Definitions are given in Definitions (Full Definitions) Field. An independent claim is included for controlling insects and / or acarina, involving applying the composition to insects and / or acarina and / or their
habitat. [Image] [Image] [Image] [Image] - ACTIVITY : Insecticide;
acaricide; safener; nematocide. In pre-tests for control of
Aphis gosspii aphids on cotton plants, mortality after 2 days was 0% using 120 g / ha N-cyanomethyl-4-
trifluoromethyl-
nicotinamide (flonicamid) (A'), 0% using 50 g / ha isoxadifen-ethyl (B') and 50% using a combination of 120 g / ha (A') and 50 g / ha (B'). -
MECHANISM OF ACTION : None given.
 Login to View More
Login to View More  Login to View More
Login to View More