Aildenafil citrate crystal form C and preparation method and application thereof
A citric acid and amorphous technology, which is applied in the field of citrate aldenafil crystalline form C and its preparation and use, and can solve the problems that do not involve the citrate aldenafil crystalline form and its preparation method, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
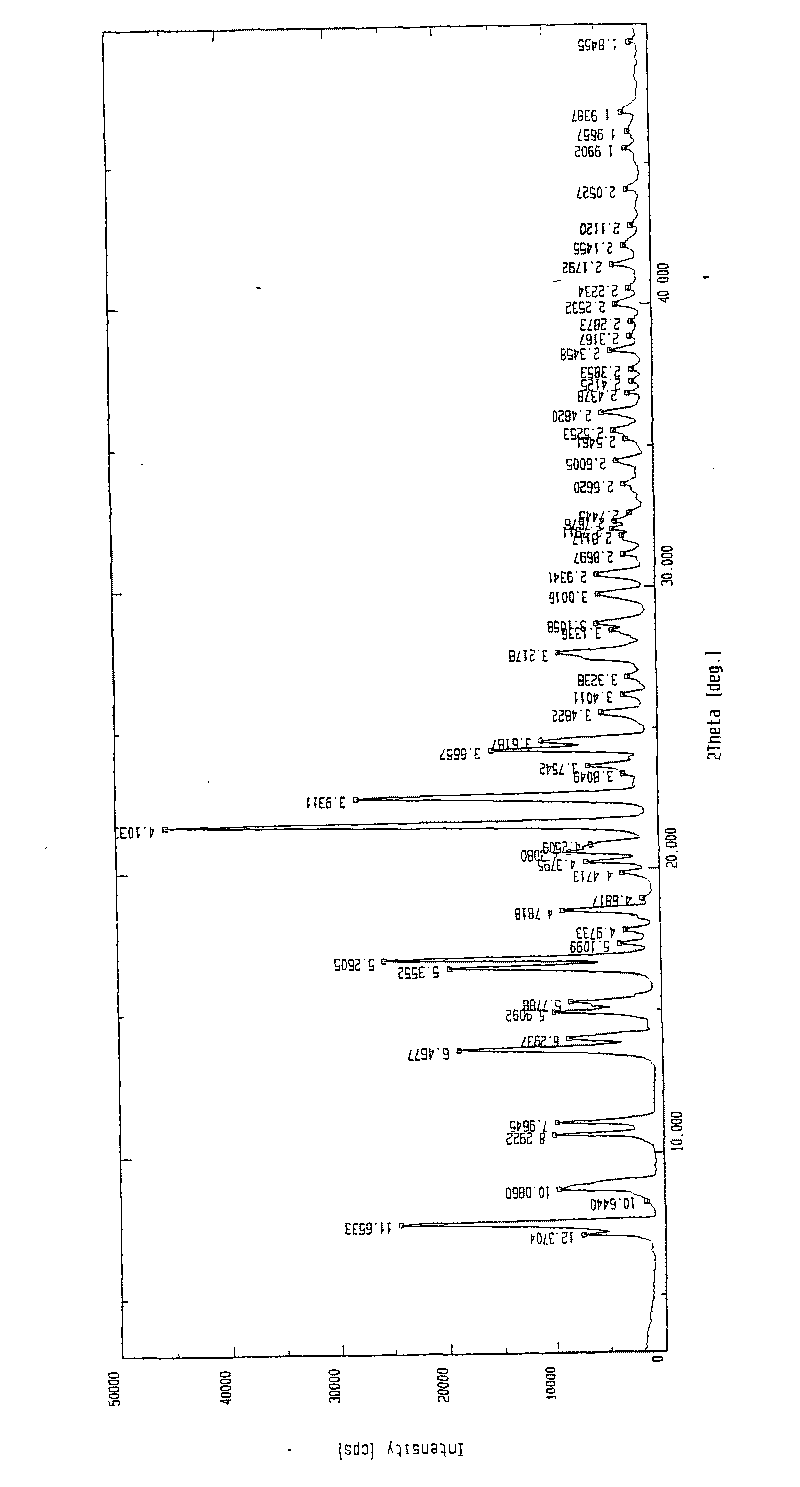
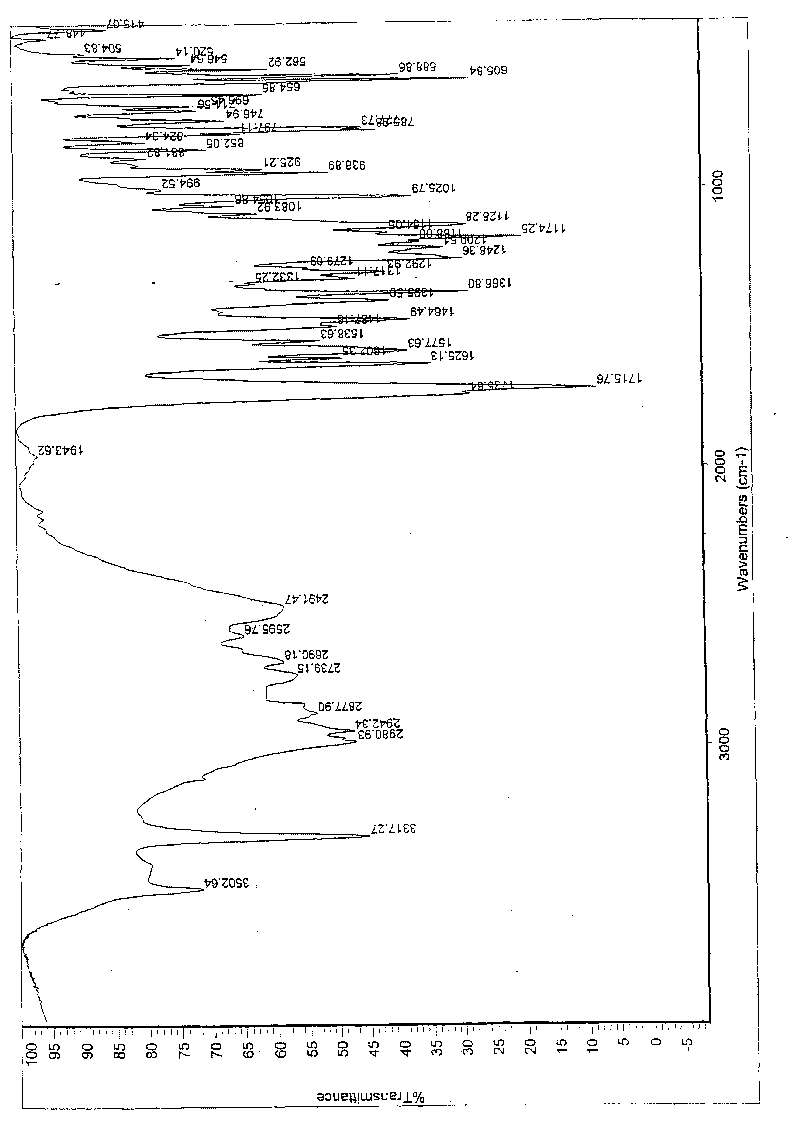
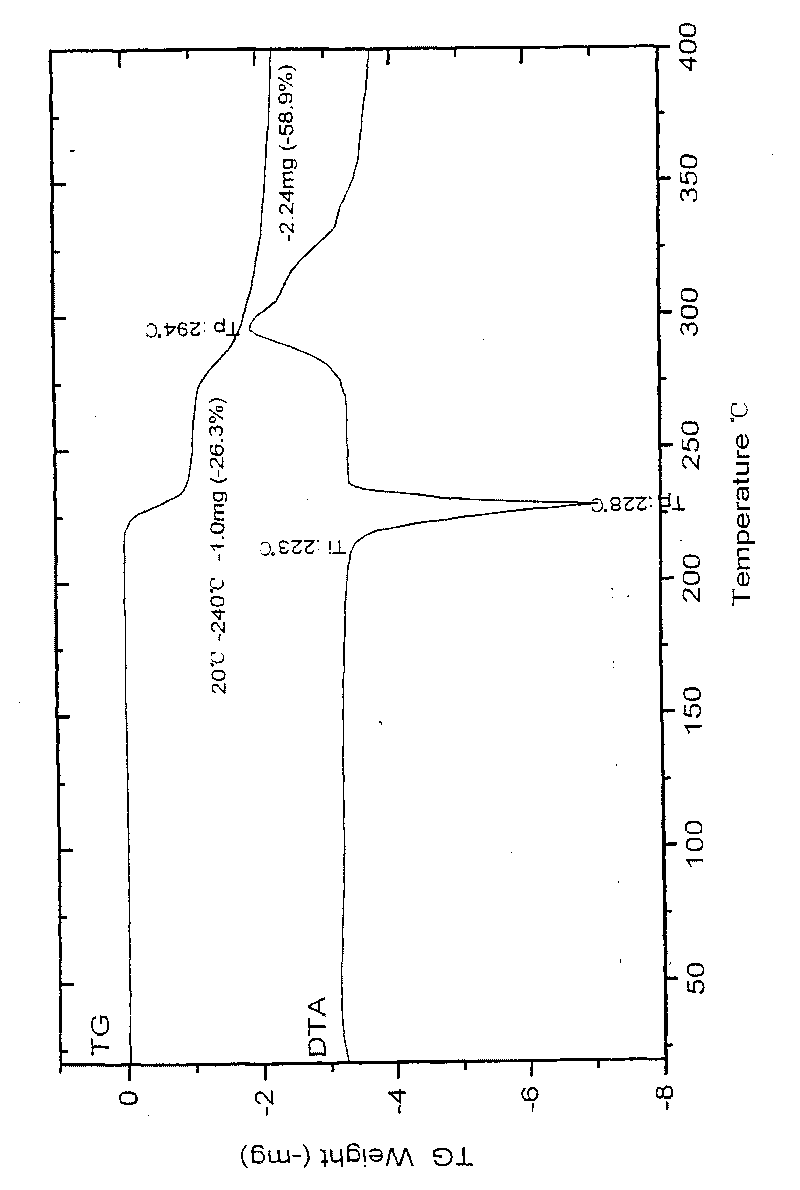
[0058] In a 1000ml reaction bottle, add 20 grams of edenafil citrate, 440ml of distilled water / acetone mixture (4.5:5.5, volume ratio), start stirring, heat up to reflux temperature, and filter while hot after 15 minutes. The filtrate was lowered to 28°C-32°C, kept warm and stirred for 45 minutes; then cooled naturally to 23±3°C, continued to stir for 10 hours, precipitated crystals, filtered, and placed in the room for 2 hours, then moved to a vacuum oven and dried in vacuum (0.08 -0.10Mpa) for 4 hours, 17.6 g of the above crystalline form C of edenamorph citrate was obtained, and the refining rate was 88%. Adopt HPLC area normalization method to measure content 99.90%, see Figure 1-Figure 3 , detected by X-ray diffractometer, infrared spectrometer and thermal analyzer, showing the characteristics of Aldenamorph Citrate Form C.
Embodiment 2
[0060] In a 2000ml reaction bottle, add 35 grams of edenafil citrate, 630ml of distilled water / acetone (6:4, volume ratio), start stirring, heat up to reflux, and filter while hot after 18 minutes. The filtrate was lowered to 30°C-32°C, kept warm and stirred for 30 minutes; then cooled naturally to 23±3°C, continued to stir for 8 hours, precipitated crystals, filtered, and placed in the room for 3 hours, then moved to a vacuum oven and dried in vacuum (0.08 -0.10Mpa) for 5 hours, 31.9 g of the above-mentioned edenamorph citrate crystal form C was obtained, and the purification rate was 91.1%. The content measured by the HPLC area normalization method is 99.90%, and after testing, it shows the characteristics of Aldenamorph citrate crystal form C.
Embodiment 3
[0062] Granules Containing Aldenafil Citrate Form C
[0063] Prescription: 50 grams of Aldenafil Citrate Form C, 650 grams of lactose, 100 grams of crospovidone, 90 grams of PEG-4000, 135 grams of hydroxypropyl methylcellulose, appropriate amount of distilled water, made into 1000 bags.
[0064] Process: PEG-4000 and aldenafil citrate crystal form C are crushed together, passed through an 80-mesh sieve, mixed with other materials, made into soft materials with distilled water, granulated, dried at low temperature, and then packed into granules.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More