Oral drug composite of beta-cyclodextrin edaravone inclusion and preparation method thereof
A technology of cyclodextrin and composition, which is applied in the field of pharmaceutical composition, can solve the problem of not verifying whether there is effective inclusion of hydroxypropyl-β-cyclodextrin/edaravone, and not making a solid package that is convenient to use. Composites, did not obtain practical application value and other issues
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
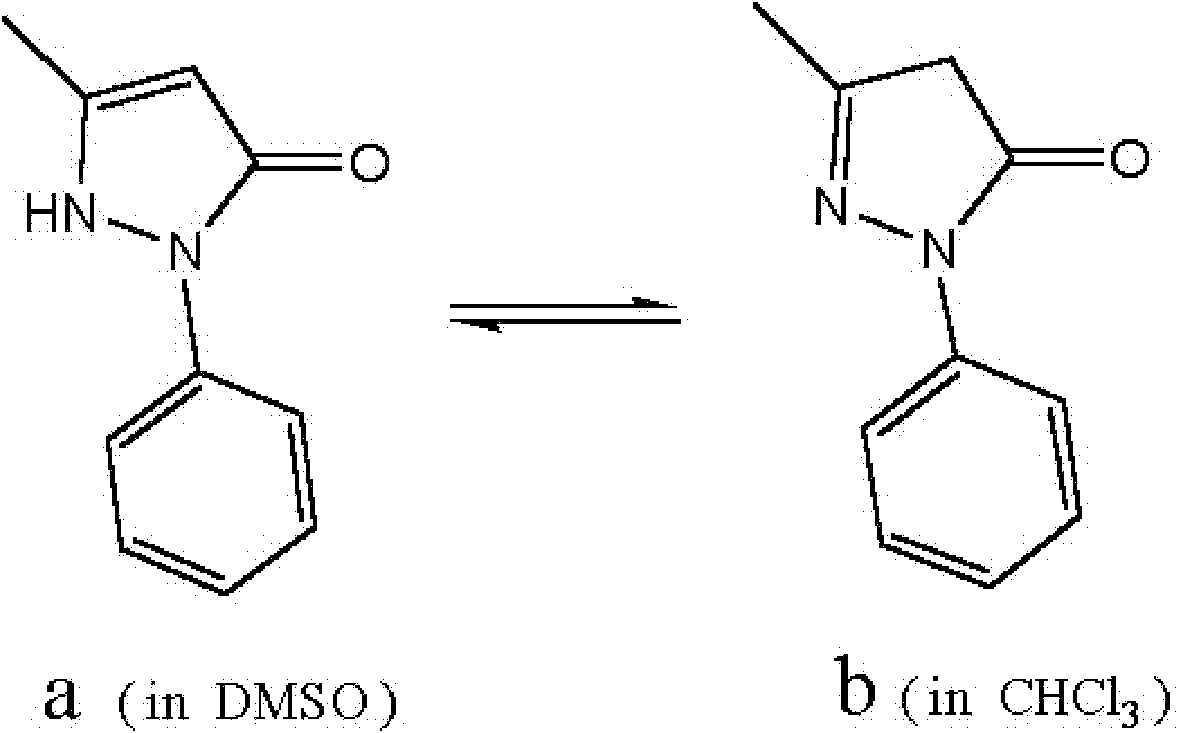
[0057] Example 1, mix 80 grams of β-cyclodextrin with 400 milliliters of pure water to make a suspension (pH=6.9), add 10 grams of powdered Edaravone, mix, fully grind and stir for 1 hour, at 50 The water was removed under reduced pressure at ℃, and then dried under reduced pressure at room temperature overnight to obtain about 90 g of white solid clathrate.
Embodiment 2
[0058] Example 2, basically the same as Example 1, but using 10 grams of β-cyclodextrin plus 240 grams of sulfobutyl-β-cyclodextrin and 400 milliliters of 0.1mol·L pH = 6.1 -1 NH 4 The Ac buffer solution was mixed to obtain about 260 g of white solid clathrates.
Embodiment 3
[0059] Example 3, basically the same as Example 1, but using 10 grams of β-cyclodextrin plus 90 grams of hydroxypropyl-β-cyclodextrin and 400 milliliters of 0.05mol·L pH = 7.5 -1 The phosphate buffer was mixed to obtain about 110 g of white solid clathrates.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More