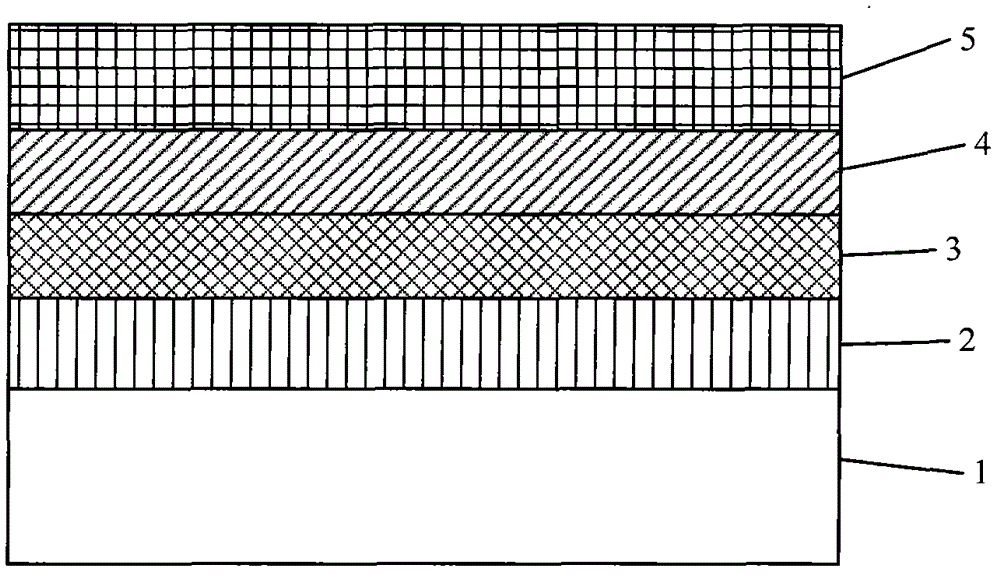
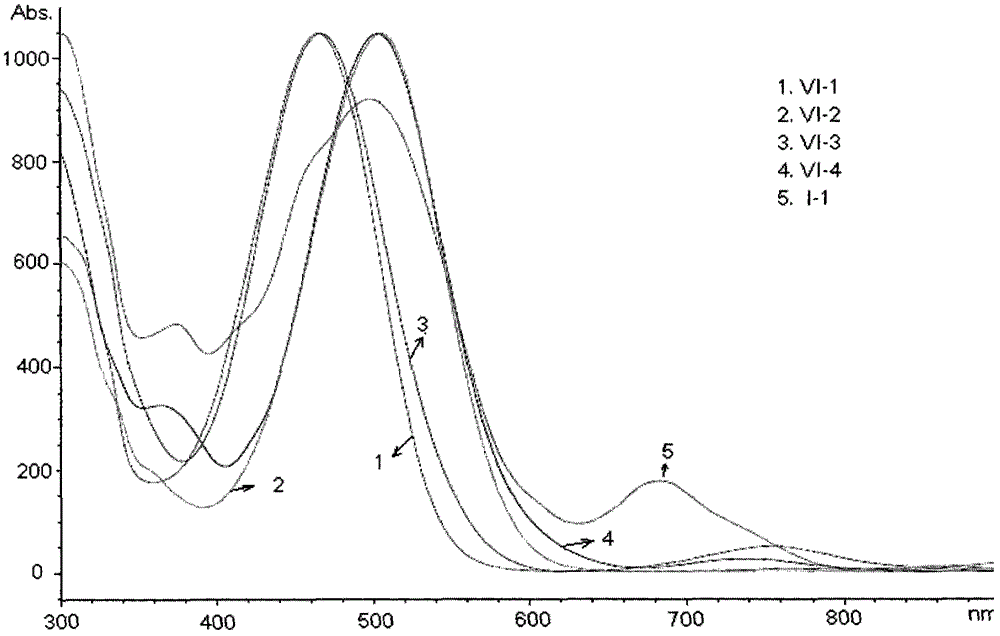
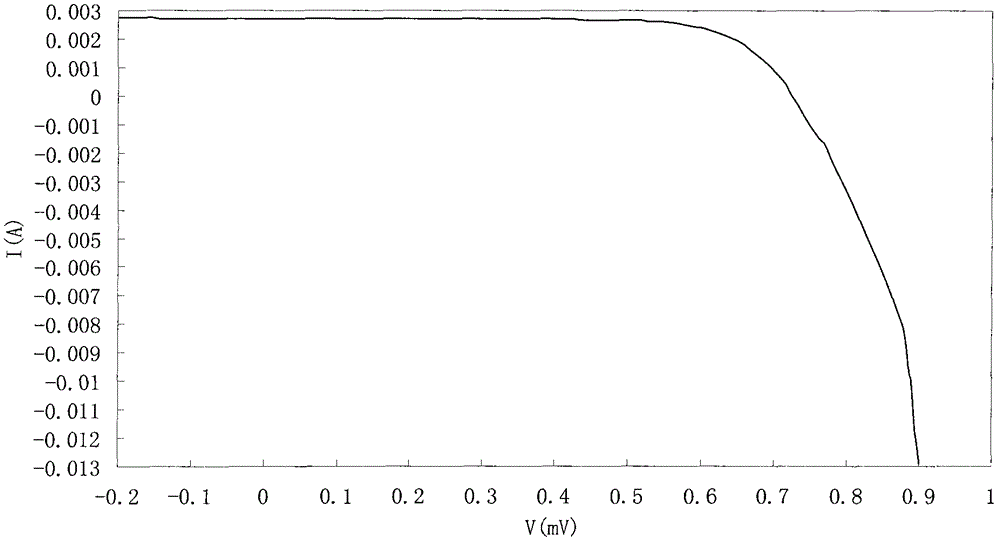
A kind of benzo nitrogen-containing heterocyclic compound, its preparation method, intermediate and application
A compound and nitrogen heterocyclic technology, applied in the application field of photosensitization medium, can solve the problems that solar cells have not been reported yet
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0117] 1) Synthesis of (E)-4-(4-(benzothiazol-2-yl)distyryl)-N,N-dimethylaniline
[0118]
[0119] Under the conditions of ice bath and nitrogen protection, sodium hydrogen (60%, 2.6 g, 64 mmol) was slowly added to 4-(2-benzothiazolyl)phenylmethyl phosphate diethyl ester M-4 (11.5 g, 32 mmol) in tetrahydrofuran (200 mL) with constant stirring. The mixture was heated to 80°C and held at this temperature for 30 minutes, then cooled to room temperature. Compound 4-N,N-dimethyl-benzaldehyde (4.8 g, 32 mmol, dissolved in 20 ml THF) was slowly added into the above reaction system, and then heated to 50° C. and kept stirring for 16 hours. The reactant was slowly poured into 500 ml of ice water, extracted twice with 250 ml of DCM, the organic phase was dried over anhydrous sodium sulfate and concentrated, and the residue was purified through silica gel with ethyl acetate petroleum ether to obtain the target product as a yellow solid 7.4 g, yield: 65%. 1 H-NMR (400MHz, CHCl 3 -d...
Embodiment 2
[0127] 2-((E)-5-((E)-3-(4-(benzothiazol-2-yl)phenyl)-2-(4-(dimethylamino)phenyl)allylidene) -Synthesis of 4-rhodanine-3-acetic acid
[0128]
[0129] Under nitrogen protection, the above compound V-1 (100 mg, 0.26 mmol), rhodanine-3-acetic acid (100 mg, 0.52 mmol), ammonium acetate (8 mg, 0.1 mmol), and acetic acid ( 5 ml) were mixed and the reaction was stirred at 80°C for 16 hours. After the reaction was completed, it was cooled to room temperature, and a solid precipitated out. After filtering, the filter cake was washed with a small amount of acetic acid, water (20 ml) and a small amount of ether, and after drying, 40 mg of a dark red solid (melting point: >250° C.) was obtained, with a yield of 28%.
[0130] 1 H-NMR (400MHz, DMSO-d 6 )δ: 2.90(s, 6h), 4.62(s, 2H), 6.55(d, 2h), 6.94(d, 2h), 7.52(m, 3h), 7.61(m, 2h), 7.78(s, 1h ), 8.13(d, 1h), 8.22(d, 1h), 8.33(d, 2h).ESI-MS(M+H + ): 558.1
Embodiment 3
[0132] 1) Synthesis of (E)-4-(4-(benzoxazol-2-yl)distyryl)-N,N-dimethylaniline
[0133]
[0134] Under the conditions of ice bath and nitrogen protection, sodium hydrogen (60%, 0.8 g, 35.6 mmol) was slowly added to 4-(2-benzothiazolyl)phenylmethyl phosphate diethyl ester M-4 (4.1 g, 11.9 mmol) in tetrahydrofuran (40 mL) with constant stirring. The mixture was heated to 80°C and held at this temperature for 30 minutes, then cooled to room temperature. Slowly add compound 4-N, N-dimethyl-benzaldehyde III-1 (2.7 g, 17.8 mmol, dissolved in 40 ml of tetrahydrofuran) in the above reaction system, then heated to 50 degrees, and kept stirring for 16 Hour. The reactant was slowly poured into 200 ml of ice water, extracted twice with 150 ml of DCM, the organic phase was dried over anhydrous sodium sulfate and concentrated, and the residue was purified through silica gel with ethyl acetate petroleum ether to obtain the target product IV-1 'Yellow solid 2.7 g, yield: 68%. 1 H-NMR (...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap