Inhibition of histone acetyltransferases by CTK7A and methods thereof
A technology of acetyltransferase and histone, which is applied in the field of inhibiting histone acetyltransferase and histone hyperacetylation, and can solve the problem that the effect of HAT inhibitors has not been tested
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0096] A. Preparation of Benzohydrazide Curcumin (CTK7): (insoluble in water)
[0097] Acetic 4-hydrazinobenzoic acid (2 ml) was added to a solution of curcumin in methanol (10 mg, 0.027 Mmol). After 24 h of incubation, the solvent was evaporated in vacuo. The remaining acid (20 mg, 0.135 mmol), and catalytic amount of triethylamine (18.8 μL, 0.135 mmol) were analyzed using TLC (CHCl 3 / MeOH=4:1; R f =0.5) was purified by repeated recrystallization and filtration. This yielded CTK7 (7.18 mg, 55%) as a dark orange powder, which was 1 NMR, melting point test, solubility test and ESI-MS were analyzed.
[0098] Preparation of B.4-(3,5-bis(3-methoxy-5-styrene oxide)-4,5-dihydro-1H-pyrazol-1-yl)sodium benzoate (CTK7A): (solution in water)
[0099] Sodium ethoxide (10 ml) was added to a solution of CTK7 in methanol (50 mg, 0.1031 mmol), and the reaction mixture was stirred at room temperature for 90 minutes. The solvent was evaporated in vacuo, and the residue was washed with...
Embodiment 1
[0102] Histone hyperacetylation at H3K14 is associated with overexpression of NPM1 and GAPDH in oral cancer
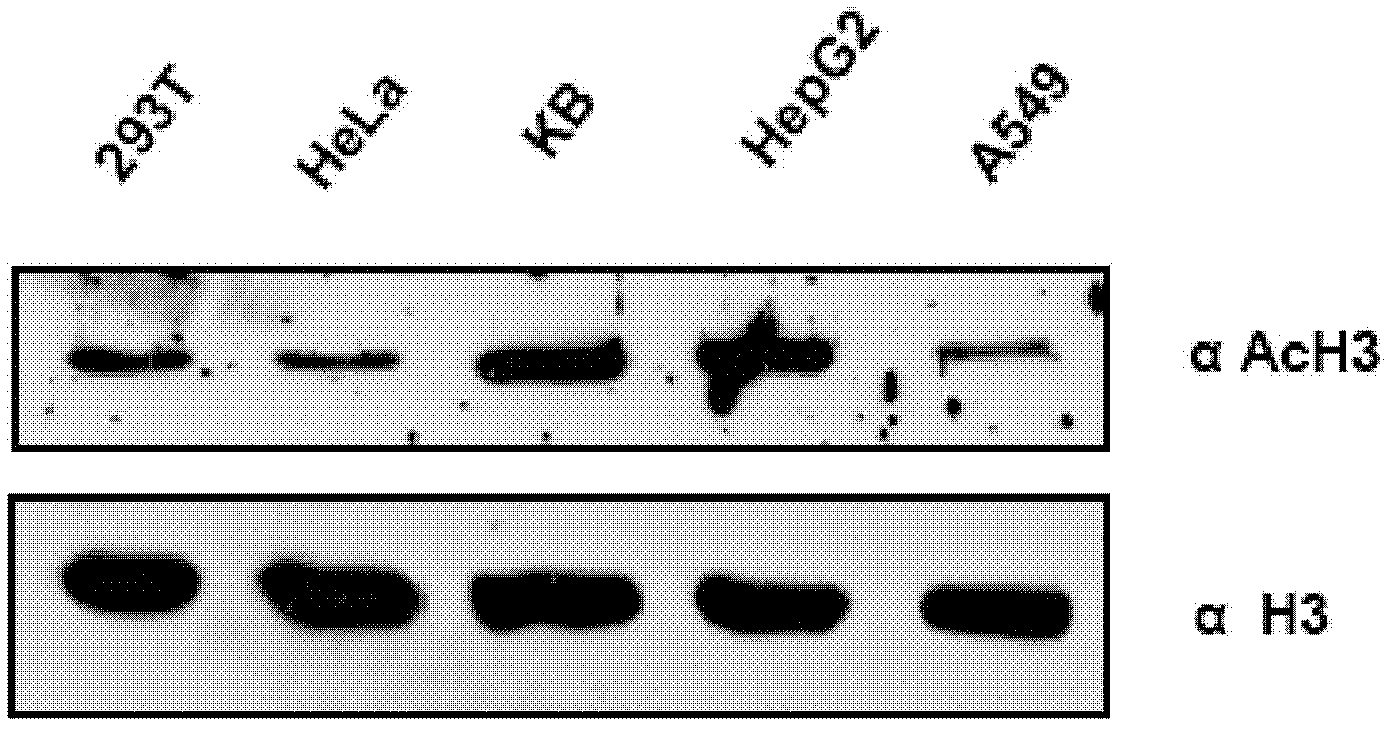
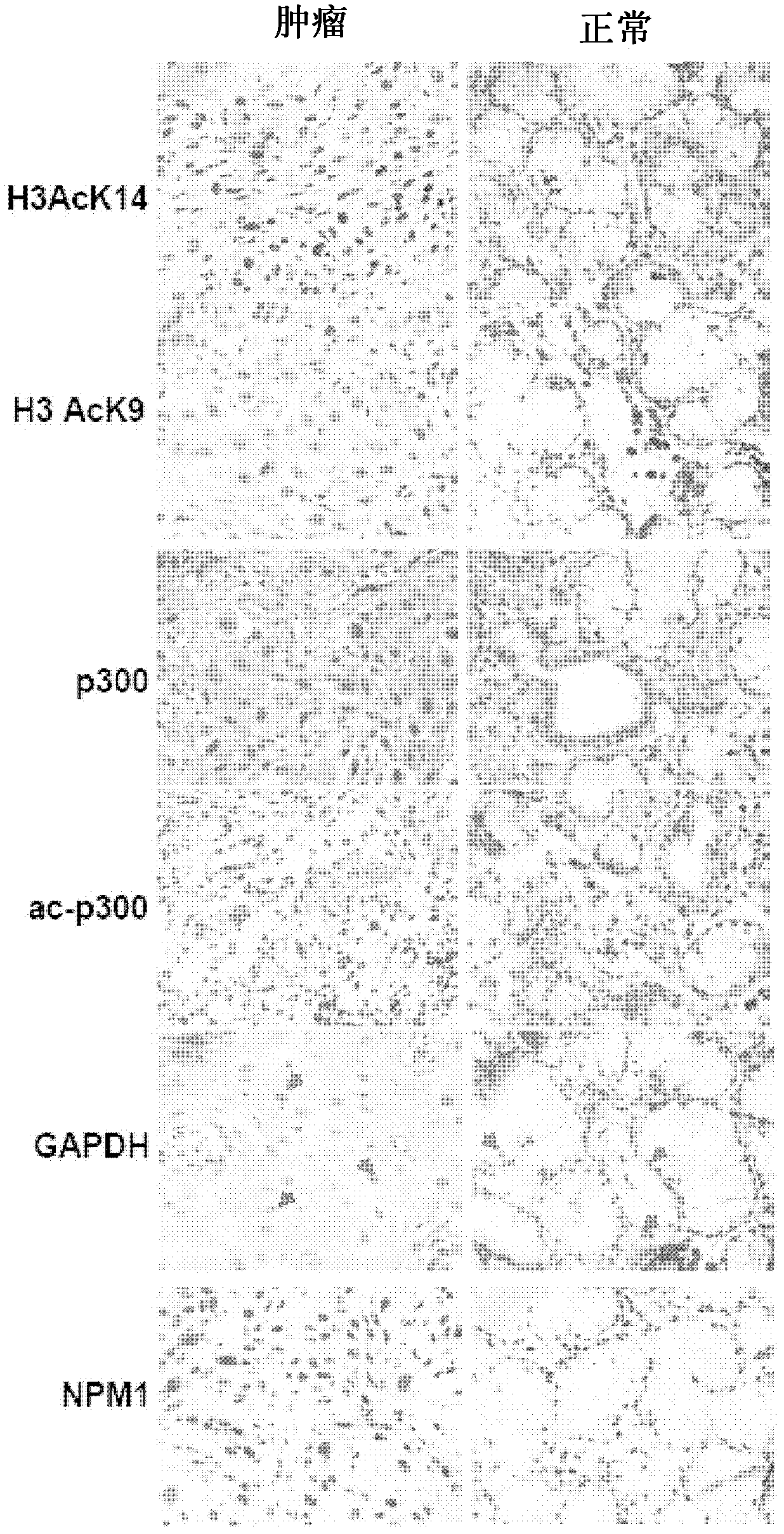
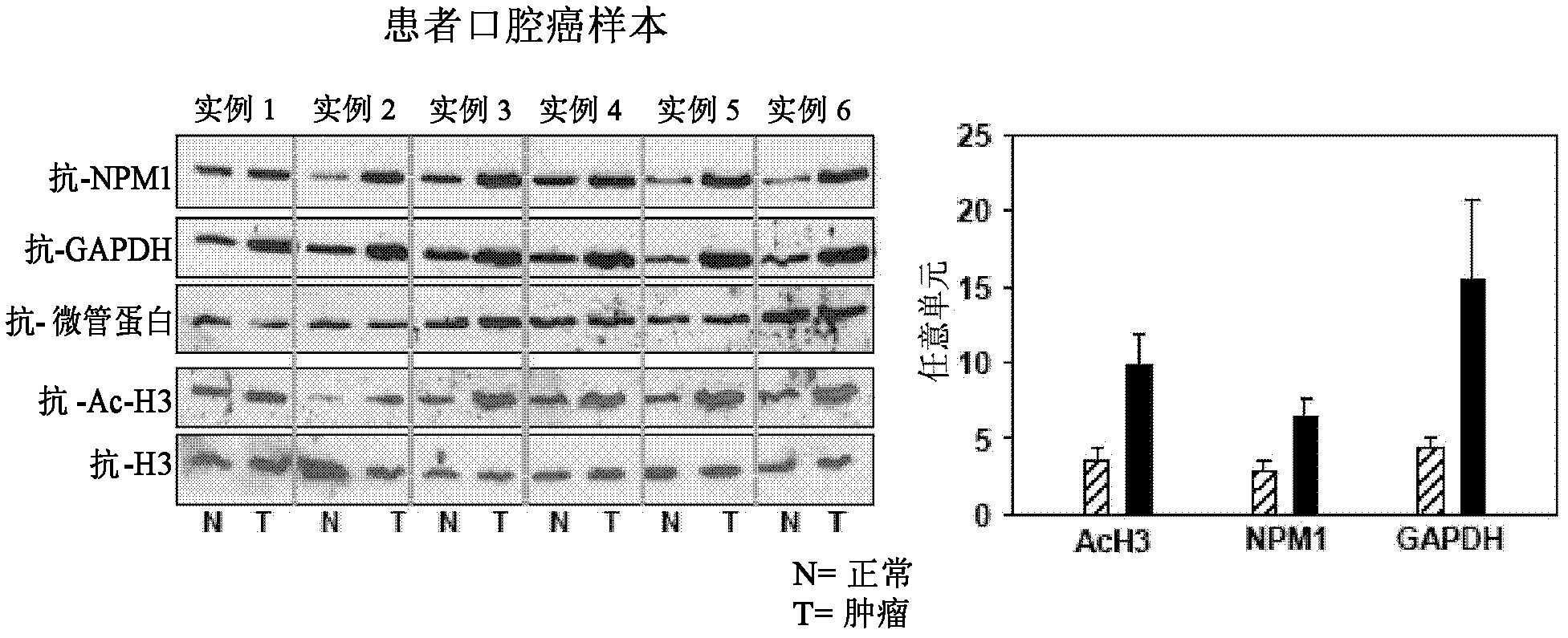
[0103] To investigate histone acetylation in different cancers, first, histones were isolated from different cell lines and analyzed by immunoblotting using an anti-acetylated histone H3 (anti-H3AcK9AcK14) antibody. Histones were observed to be significantly hyperacetylated in oral (KB) and liver (HepG2) cancer cell lines ( figure 1 ). Histones were isolated from cells of different cell lines as indicated, and histone acetylation was analyzed by immunoblotting using an anti-acetylated H3 (anti-H3AcK9AcK14) antibody. Anti-H3 was used as a loading control. Although hyperacetylation of histones in HCC has been reported recently 24 , but for oral cancer cell lines: the almost equivalent enhanced acetylation of histone H3 is quite interesting. These results led us to discover the acetylation levels of histone H3 in tissues from oral cancer patient samples. By applying ...
Embodiment 2
[0107] NO-induced H3K14 acetylation is associated with overexpression of NPM1 and GAPDH via p300 autoacetylation
[0108] The free radical gas, NO, is produced by the nitric oxide synthase (NOS) family. NO is a pleiotropic signaling molecule identified as a mediator of many physiological and pathological conditions 41 . Since the increase in NO production was found to be accompanied by a concomitant upregulation of inflammatory (mainly NFk-B responsive) genes in oral cancer 42,43 , thus hypothesizing that NO signaling may be related to autoacetylation of p300, overexpression of GAPDH and NPM1, and hyperacetylation of histones. observed that iNOS levels were indeed significantly elevated in tumor tissue samples ( Figure 3a ). It was also found that COX2 levels were higher in these tumor tissue samples ( Figure 2a ). A recent report proposes that NO-dependent and nuclear-localized GAPDH enhances p300 autoacetylation and thus its catalytic activity 37 . GAPDH has been f...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More