Treatment of MCI and Alzheimer's disease
A composition and drug technology, applied in the direction of botanical equipment and methods, pharmaceutical formulations, medical preparations containing active ingredients, etc., can solve problems such as neurodegeneration that does not show
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0253] Example 1: Neurotoxicity of the PDS / TTR complex
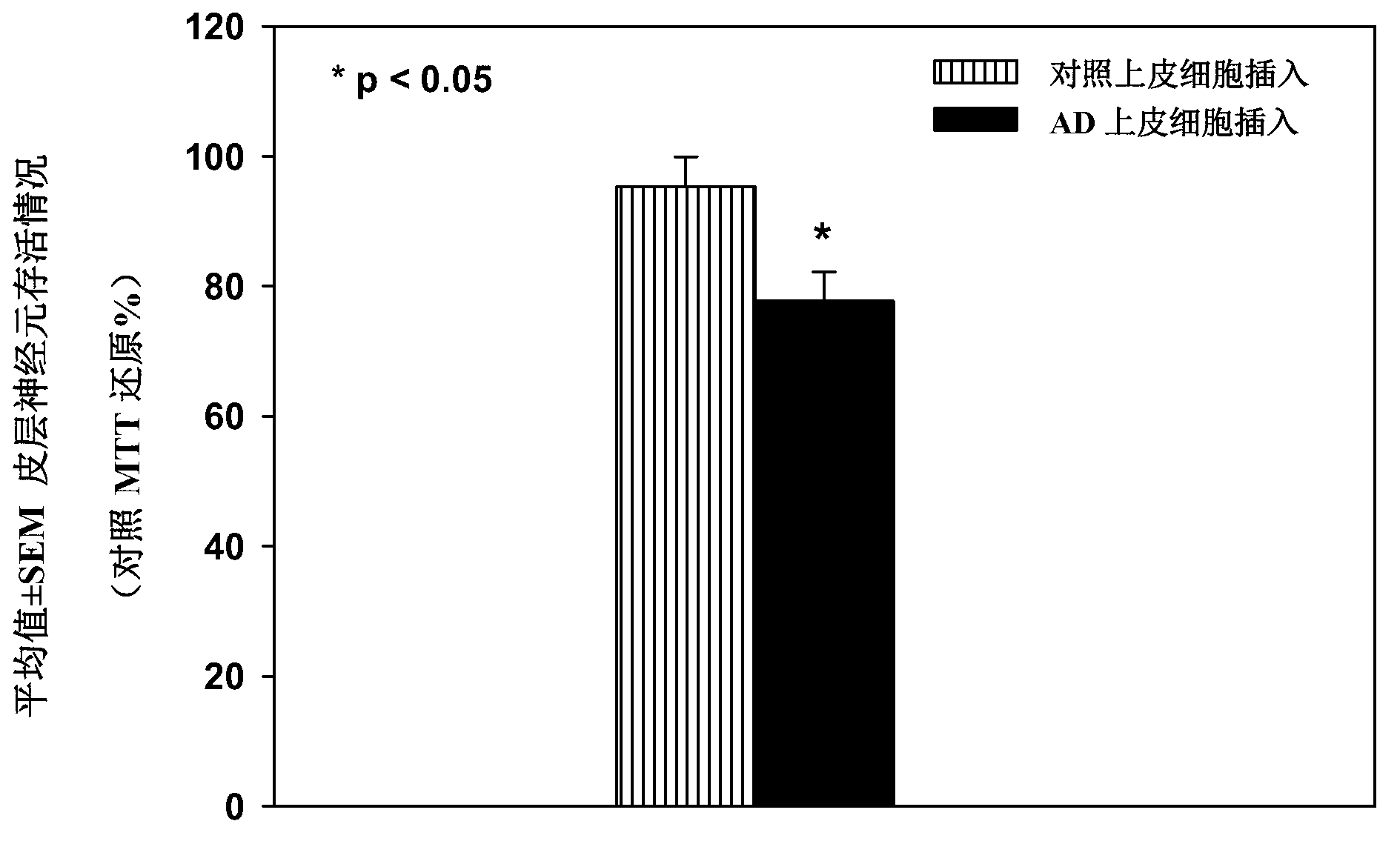
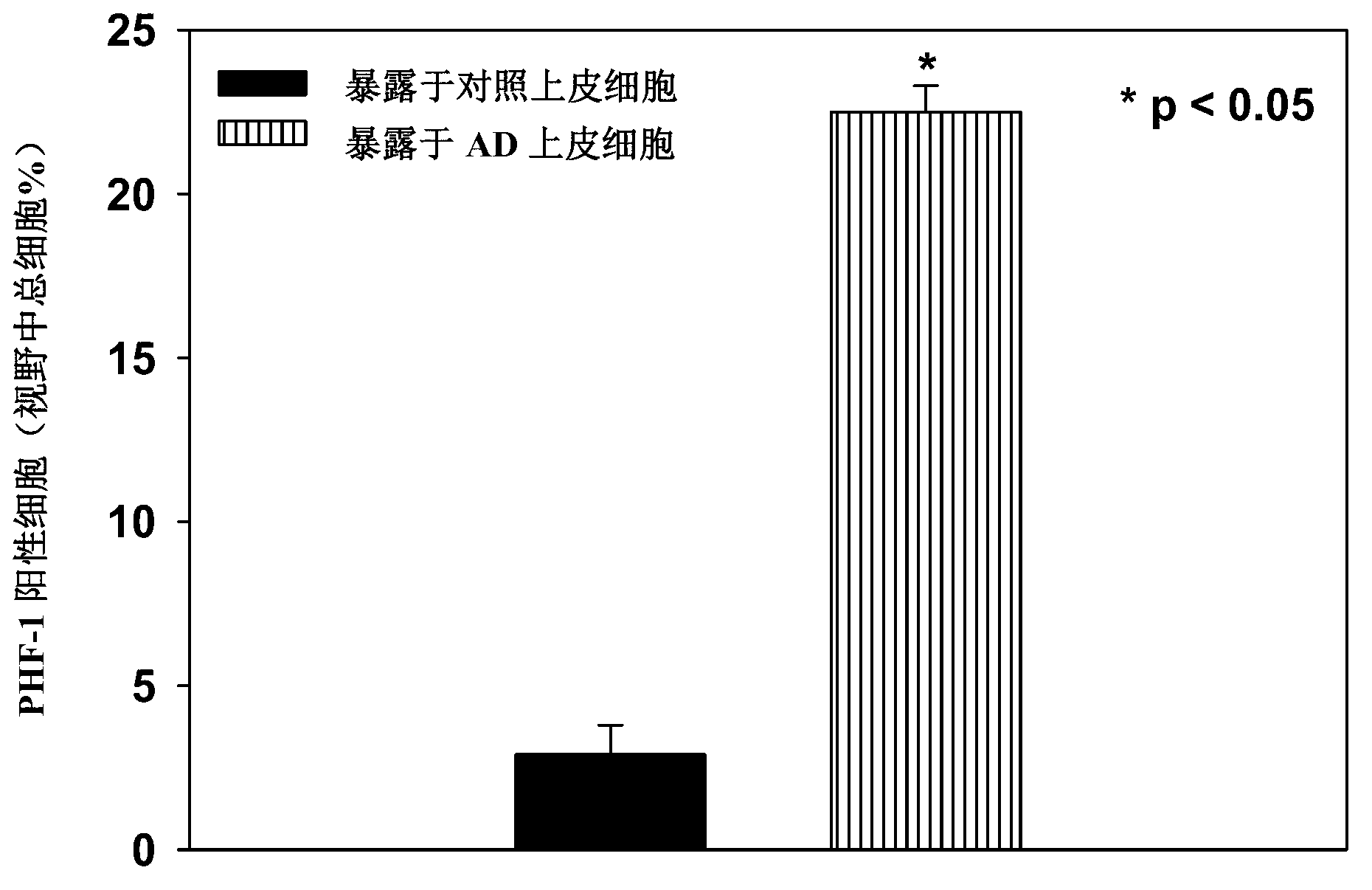
[0254] Examination of the cell culture medium of epithelial cells from Alzheimer's patients showed elevated levels of the PDS / TTR complex compared with control cells, suggesting that the PDS / TTR complex could act as a An effective biomarker for the early diagnosis of MM. See US Patent Application Publication No. 20080026405, the disclosure of which is incorporated herein by reference. This example shows that the PDS / TTR complex is neurotoxic in addition to being a biomarker of disease.
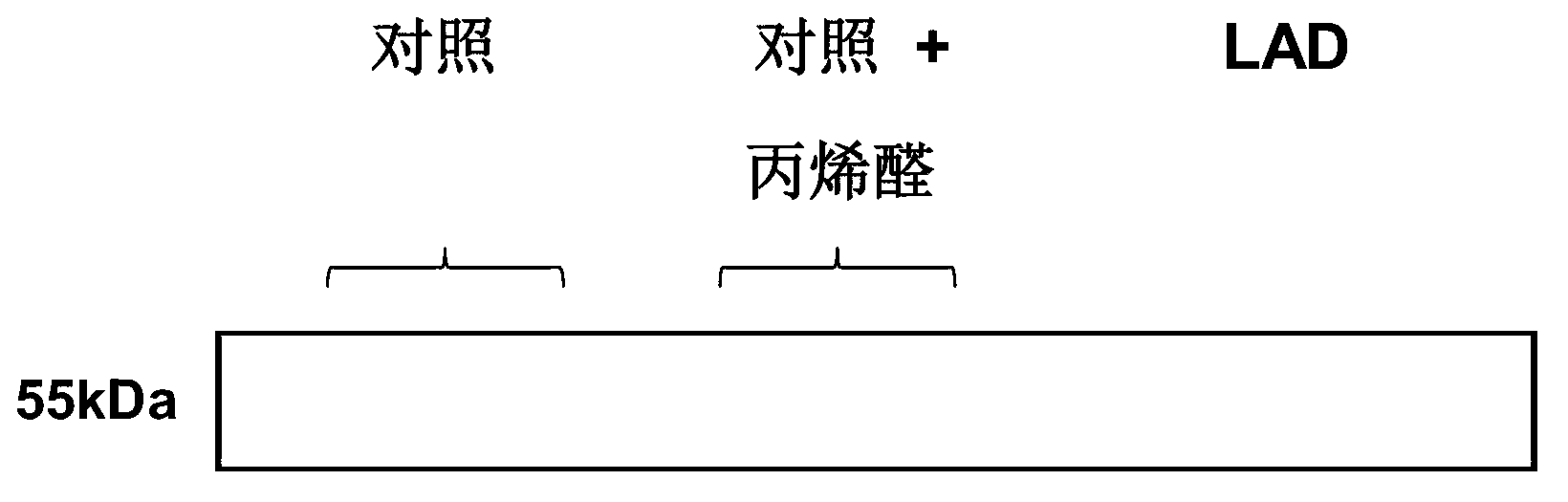
[0255] First, it was found that acrolein (a by-product of lipid peroxidation, α,β-unsaturated tricarbonaldehyde) caused normal control epithelial cells to express PDS / TTR complexes in the medium at levels comparable to those from Alzheimer's The epithelial cells of patients with Mersen's disease are comparable. In these experiments, primary cultures of choroidal plexus epithelial cells were established rapidly after necropsy using e...
Embodiment 2
[0260] Example 2: Inhibition of PDS / TTR Expression by Nifedipine, Nifedipine Analog Mixture and / or T3 / T4
[0261]The assay described in Example 1 provides a tool for identifying potential therapeutic agents capable of protecting neuronal cells against the PDS / TTR complex. The inventors observed that compounds such as nifedipine (1,4-dihydro-2,6-dimethyl-4-(2-nitrophenyl)-3,5-pyridinedicarboxylate, CAS# 21829-25-4 (Sigma Aldrich)), a calcium channel blocker for the treatment of hypertension; or nifedipine analogues such as oxidized derivatives of nifedipine (2,6-dimethyl-4-(2 -nitrophenyl)-3,5-pyridinedicarboxylate, CAS#67035-22-7 (Sigma Aldrich)) or nitroso derivatives of nifedipine (2,6-dimethyl- Dimethyl 4-(2-nitrosophenyl)-3,5-pyridinedicarboxylate, CAS#50428-14-3 (Sigma Aldrich)), alone or in combination, potently inhibits Expression of the PDS / TTR complex. In addition, T3 and T4 were evaluated and found to be effective in inhibiting the expression of the PDS / TTR comp...
Embodiment 3
[0266] Example 3: Nifedipine analogs inhibit the production of inflammatory cytokines
[0267] It has been reported that inflammatory response elements (cytokines) are increased in Alzheimer's patients. The present inventors tested the nifedipine mixture and its respective analogues in astrocytoma cultures. Human astrocytoma cells were 2.5X 10 5 Cells / well were seeded in 6-well culture plates and cultured for 24 hours. The cultures were then changed to serum-free selective MEM medium and treated with a mixture of nifedipine and its respective analogs for 24 hours. Three 6-well plates were used per treatment. After treatment, the medium in each well was collected and the levels of IL-1β, IL-6, TNF-α and TGF-β were detected using commercial ELISA. For example results see Figure 7 . Such as Figure 7 As shown, the levels of secreted IL-1, IL-6, and TNF-α in the medium were significantly decreased after treatment with the nifedipine mixture or oxidized nifedipine, sugges...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More