Lixivaptan crystal form I and preparation method and use thereof
A technology of tanned crystal form and crystal form, applied in the field of anti-heart failure drugs, can solve the problems of high price, low utilization rate and high production cost
Active Publication Date: 2014-09-24
TIANJIN INSTITUTE OF PHARMA RESEARCH
View PDF3 Cites 1 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Because raw material 2 is expensive, route 1 takes 2 as starting material, and after multi-step reaction, its utilization rate is low, and production cost is higher
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
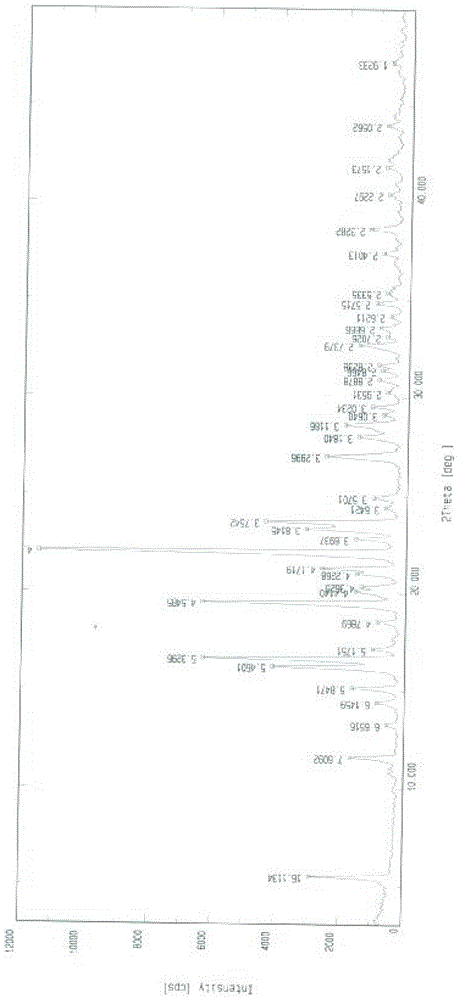
[0047] Weigh 20.0g of lixiptan, add 80mL of methanol, heat to 60°C and stir for 1 hour, naturally cool to room temperature, a solid precipitates, filter, and vacuum dry for about 24 hours to obtain white crystals of lixiptan (form I) .
Embodiment 2
[0049] Weigh 20.0g of lixiptan, add 40mL of methanol, heat to 60°C and stir for 2 hours, naturally cool to room temperature, a solid precipitates, filter, and vacuum dry for about 24 hours to obtain white crystals of lixiptan (form I) .
Embodiment 3
[0051] Weigh 20.0g of lixiptan, add 120mL of methanol, heat to 60°C and stir for 3 hours, naturally cool to room temperature, a solid precipitates, filter, and vacuum dry for about 24 hours to obtain white crystals of lixiptan (form I) .
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
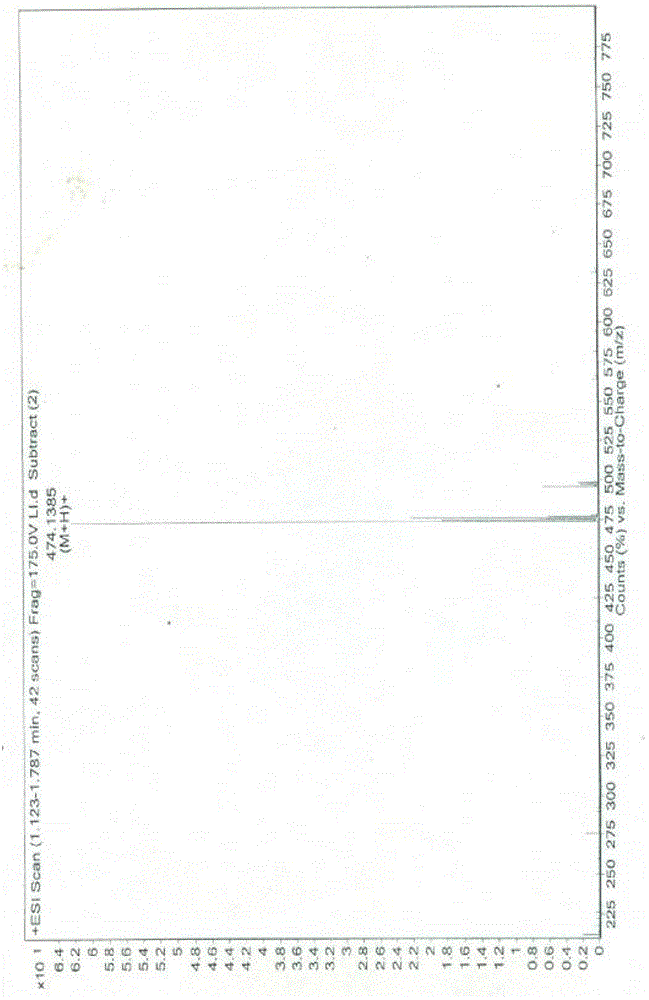
The invention belongs to the field of anti heart failure drugs, and more specifically, relates to lixivaptan crystal form I and a preparation method and use of a pharmaceutical composition containing the lixivaptan crystal form I in preparation of drugs for treating hyponatremia. The lixivaptan crystal form I is prepared from 2-chloro-4-nitro benzoic acid as a starting material by esterification, hydrogenation reduction, acylation, hydrolysis, acyl chlorination and other reaction steps, and the prepared lixivaptan purity is 97.5%. The lixivaptan crystal form I and its powder are characterized by X ray diffraction diagrams, and the lixivaptan crystal form I is important for obtaining of compounds with high purity, very determined crystal form and good reproducibility due to good prospects for development and pharmaceutical value of the lixivaptan crystal form I.
Description
technical field [0001] The present invention belongs to the field of anti-heart failure drugs, and more specifically relates to lixivaptan or N-[3-chloro-4-(10,11-dihydro-5H-pyrrolo) of formula (I) Crystal Form I of [2,1-c][1,4]benzodiazepine-10-ylcarbonyl)phenyl]-5-fluoro-2-methylbenzamide and its preparation method and containing it A pharmaceutical composition and its use in the manufacture of medicines for treating hyponatremia. Background technique [0002] Arginine vasopressin (AVP), also known as vasopressin and antidiuretic hormone, is a nine-peptide cyclic hormone produced by the hypothalamus and released by the pituitary gland. Diuretic, constrict blood vessels, enhance memory, participate in body temperature and immune regulation, participate in social behavior regulation and other different physiological effects. AVP receptors belong to G-protein-coupled receptors, which are divided into three subtypes according to different transmission mechanisms: Vla, Vlb an...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C07D487/04A61K31/5517A61P3/12A61P1/14A61P9/04A61P5/00
CPCC07D487/04
Inventor 刘登科白玫刘颖穆帅张大帅
Owner TIANJIN INSTITUTE OF PHARMA RESEARCH