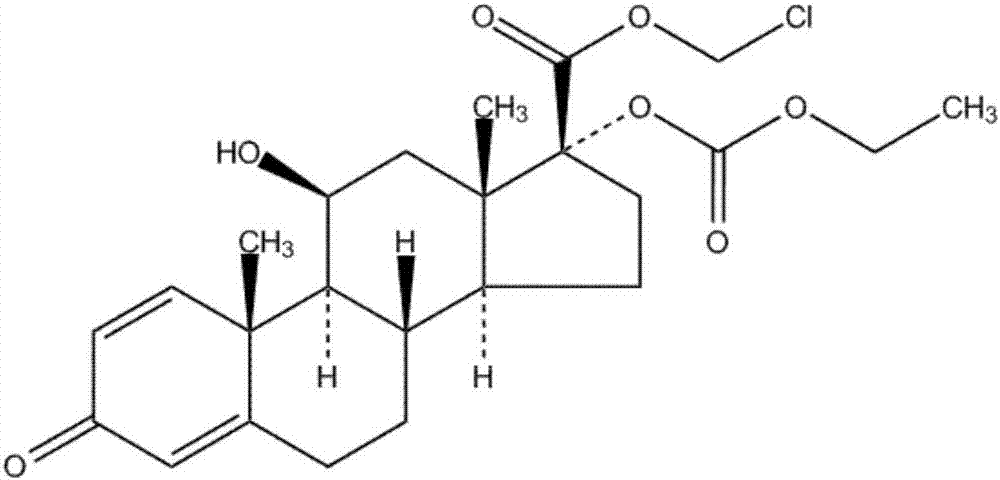
Loteprednol preparation method and ophthalmic composition of loteprednol
A technology of loteprednol and dichloromethane, which is applied in the field of medicine, can solve the problems of micronization of raw materials for preparation technology and aggregation of eye drop particles, and refrigerated storage cannot be ruled out.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
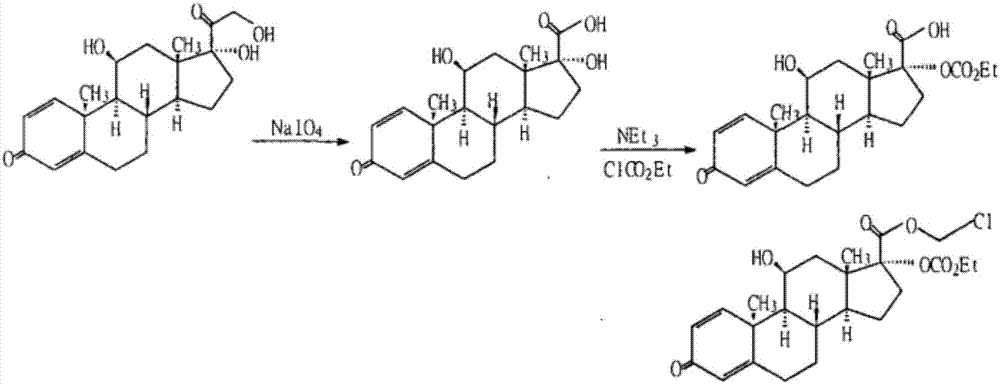
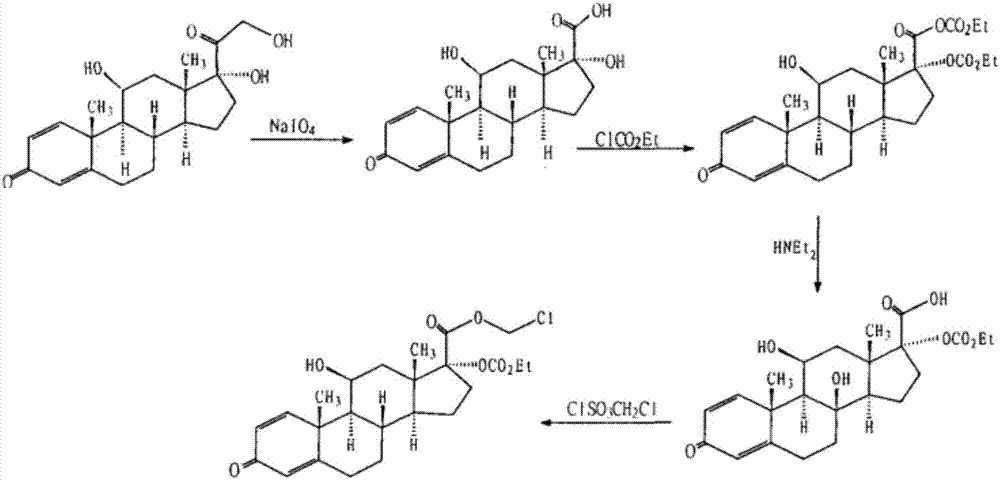
[0084] Embodiment 1: prepare loteprednol
[0085] Step 1. Oxidation
[0086]
[0087] In the mixed solution containing 120 milliliters of tetrahydrofuran and 30 milliliters of methanol, add 14.9 grams (40 mmol) of prednisolone, stir to dissolve, add dropwise warm sodium periodate solution (25.7 grams of sodium periodate are dissolved in 100 ml of water), after the dropwise addition, the reaction mixture was stirred at room temperature for 2 hours, concentrated under reduced pressure, distilled off tetrahydrofuran and methanol, cooled and suction filtered, washed with water, and dried to obtain 11α, 17β-dihydroxy-3-oxoandroster -1,4-diene-17β-carboxylic acid (Intermediate I) 13 grams, off-white powder, melting point 258-260°C, yield 90.8%.
[0088] Step 2. Condensation
[0089]
[0090] 10 grams of Intermediate I (28.8 mmol) were added to 100 milliliters of water containing 24.2 grams of sodium bicarbonate, stirred and dissolved, then 100 milliliters of dichloromethan...
Embodiment 2
[0099] Embodiment 2: prepare loteprednol
[0100] Step 1. Oxidation
[0101]
[0102] In the mixed solution containing 120 milliliters of tetrahydrofuran and 30 milliliters of methanol, add 14.9 grams (40 mmol) of prednisolone, stir to dissolve, add dropwise warm sodium periodate solution (25.7 grams of sodium periodate are dissolved in 100 ml of water), after the dropwise addition, the reaction mixture was stirred at room temperature for 3 hours, concentrated under reduced pressure, distilled off tetrahydrofuran and methanol, cooled and suction filtered, washed with water, and dried to obtain 11α, 17β-dihydroxy-3-oxoandroster 13.3 g of -1,4-diene-17β-carboxylic acid (Intermediate I), an off-white powder with a melting point of 258-261°C.
[0103] Step 2. Condensation
[0104]
[0105] 10 grams of Intermediate I (28.8 mmol) were added to 100 milliliters of water containing 24.2 grams of sodium bicarbonate, stirred and dissolved, then 100 milliliters of dichloromethan...
Embodiment 3
[0114] Embodiment 3: prepare loteprednol
[0115] Step 1. Oxidation
[0116]
[0117] In the mixed solution containing 120 milliliters of tetrahydrofuran and 30 milliliters of methanol, add 14.9 grams (40 mmol) of prednisolone, stir to dissolve, add dropwise warm sodium periodate solution (25.7 grams of sodium periodate are dissolved in 100 ml of water), after the dropwise addition, the reaction mixture was stirred at room temperature for 5 hours, concentrated under reduced pressure, distilled off tetrahydrofuran and methanol, cooled and suction filtered, washed with water, and dried to obtain 11α, 17β-dihydroxy-3-oxoandroster 13.3 g of -1,4-diene-17β-carboxylic acid (Intermediate I), an off-white powder with a melting point of 257-260°C.
[0118] Step 2. Condensation
[0119]
[0120] 10 grams of Intermediate I (28.8 mmol) were added to 100 milliliters of water containing 24.2 grams of sodium bicarbonate, stirred and dissolved, then 100 milliliters of dichloromethan...
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More