Applications of azelaic acid, adjuvants and vaccines containing azelaic acid
An azelaic acid and vaccine technology, applied in the field of immunology, can solve the problems of high price, serious side reactions, and insignificant immune response effects, and achieve the effects of low cost, small toxic and side effects, and enhancing humoral immune responses.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0025] The hepatitis A vaccine containing azelaic acid adjuvant provided in this example is: the adjuvant obtained after dissolving 0.5 mg azelaic acid with 5 μ L of 75% ethanol, and every single HAV antigen (that is, the single injection used in animal experiments) is added to the adjuvant as usual. dose), and finally add physiological saline to 300 μL, mix evenly, and obtain hepatitis A vaccine containing azelaic acid adjuvant.
[0026] Wherein, azelaic acid is a commercially available product, and its molecular formula is C 9 h 16 o 4 , with a molecular weight of about 188.22, was purchased from adamas-beta in Switzerland; HAV antigen was a commercially available HAV antigen solution with a titer of 256EU / mL 18EU, which was purchased from the Institute of Medical Biology, Chinese Academy of Medical Sciences and Peking Union Medical College.
[0027] Gained immunity test and the effect of the hepatitis A vaccine containing azelaic acid adjuvant are as follows:
[0028] A....
Embodiment 2
[0044] The Hepatitis A vaccine with azelaic acid adjuvant provided in this example is: after dissolving 1 mg azelaic acid in 10 μL of 75% ethanol, the adjuvant is routinely added to each single HAV antigen (that is, the single injection dose used in animal experiments) , and finally add physiological saline to 300 μL, mix evenly, and obtain hepatitis A vaccine containing azelaic acid adjuvant.
[0045] The immune test of the hepatitis A vaccine containing azelaic acid adjuvant obtained in this example is the same as in Example 1, and the results are shown in Table 2.
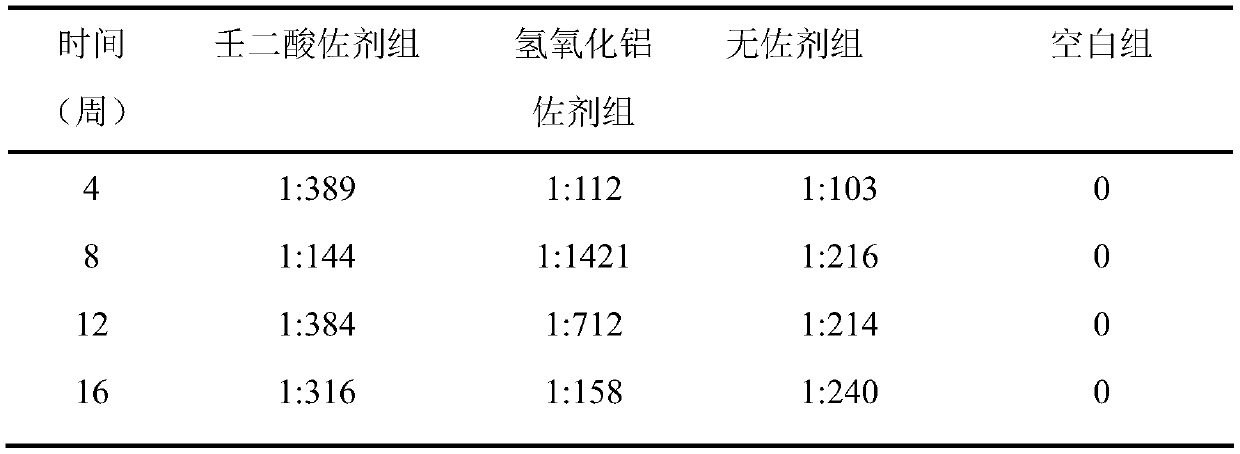
[0046] Table 2 shows the serum anti-HAV IgG antibody levels of mice in each experimental group within 16 weeks after using the adjuvant provided in Example 2.
[0047] Table 2
[0048]
[0049] It can be seen from the data analysis that except for the blank group, anti-HAVIgG was detected in all experimental groups after 4 weeks of immunization.
Embodiment 3
[0051] The Hepatitis A vaccine with azelaic acid adjuvant provided in this example is: Dissolve 2 mg of azelaic acid in 20 μL of 75% ethanol and then add the adjuvant to each single portion of HAV antigen (i.e. the single injection dose used in animal experiments) as usual , and finally add physiological saline to 300 μL, mix evenly, and obtain hepatitis A vaccine containing azelaic acid adjuvant.
[0052] The immune test of the hepatitis A vaccine containing azelaic acid adjuvant obtained in this example is the same as in Example 1, and the results are shown in Table 3.
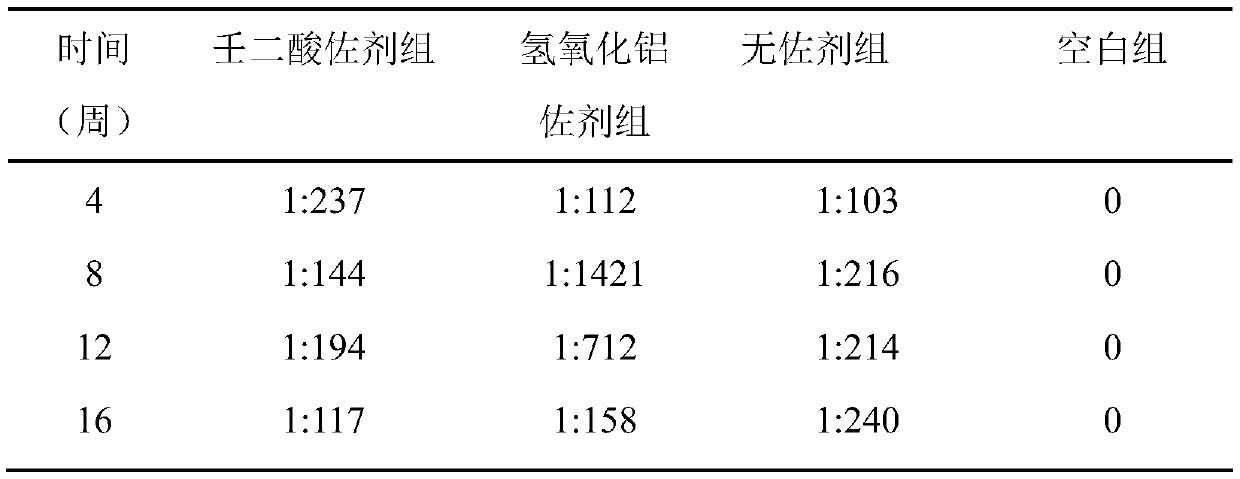
[0053] Table 3 shows the serum anti-HAV IgG antibody levels of mice in each experimental group within 16 weeks after using the adjuvant provided in Example 3.
[0054] table 3
[0055]
[0056]
[0057] It can be seen from the data analysis that except for the blank control group, the mice in each group produced anti-HAV IgG antibodies 4 weeks, 8 weeks, 12 weeks, and 16 weeks after immunization. At t...
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| boiling point | aaaaa | aaaaa |
| flash point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap