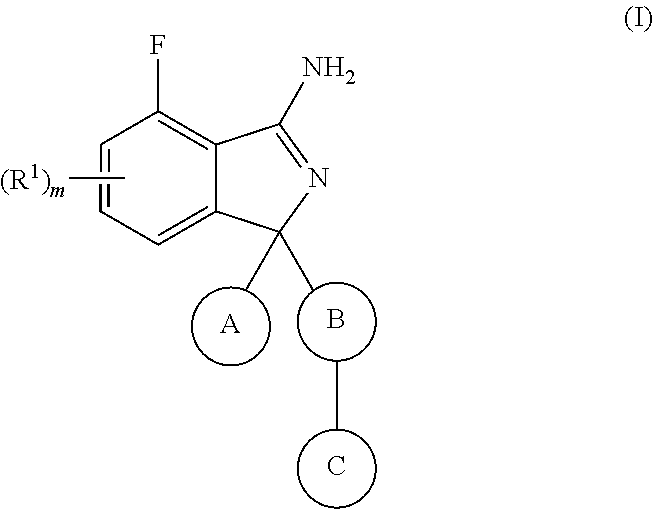
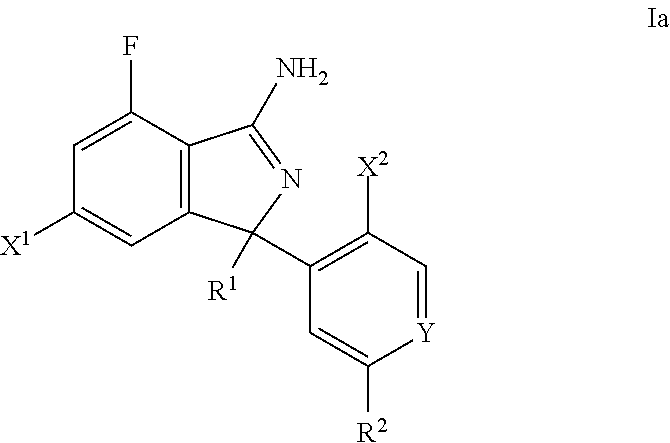
Substituted Isoindoles
a technology of substituted isoindoles and isoindoles, which is applied in the field of substituted isoindoles, can solve the problems of high prevalence of alzheimer's disease in this population, disease becomes a greater and greater problem,
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1i
5-Bromo-2-fluoro-benzoyl chloride
[0228]
[0229]To a suspension of 5-bromo-2-fluoro-benzoic acid (10.0 g, 45.7 mmol) in anhydrous dichloromethane (200 mL) was added oxalyl chloride (5.2 mL, 59.4 mmol) followed by addition of anhydrous DMF (0.2 mL). The reaction mixture was stirred at room temperature overnight, then concentrated to dryness in vacuo to give title compound in quantitative yield, which was used directly in the next step.
[0230]1H NMR (400 MHz, CDCl3) δ ppm 8.22 (dd, 1H), 7.77-7.73 (m, 1H), 714 (dd, 1H); 19F NMR (400 MHz, CDCl3) δ ppm −110.55
example 2i
2-Methoxy-isonicotinoyl chloride
[0231]
[0232]Thionyl chloride (30.0 mL, 0.39 mol) was added to a suspension of 2-methoxy-isonicotinic acid (15.0 g, 97.9 mmol) in anhydrous toluene (150 mL). The mixture was heated at 80° C. under nitrogen for 3 hours, cooled to room temperature and filtered. The filtrate was concentrated in vacuo to afford the title compound 7.7 g (46% yield).
[0233]1H NMR (400 MHz, CDCl3) δ ppm 8.36 (d, 1H), 5.30 (d, 1H), 7.38 (s, 1H), 4.00 (s, 3H).
example 3i
2-(3-Bromobenzoyl)-6-fluorobenzonitrile
[0234]
[0235]A solution of copper(I) cyanide (4.70 g, 52.50 mmol) and lithium bromide (2.63 mL, 105.00 mmol) in THF (65 mL) was added to (2-cyano-3-fluorophenyl)zinc(II) iodide (100 mL, 50 mmol) at −78° C. under an argon atmosphere. The mixture was stirred at r.t. for 1 h and then cooled to −78° C. 3-Bromobenzoyl chloride (6.94 mL, 52.50 mmol) was added dropwise and the mixture was stirred at r.t. for 4 h. NH4Cl (50 mL, sat aq) was added followed by water (50 mL). The THF was removed in vacuo and the aqueous residue was diluted with water (100 mL) and DCM (150 mL). A precipitate was filtered off and the filtrate was added to a separation funnel. The organic layer was separated and the water phase extracted with DCM (100 mL). The combined organics were washed with brine (150 mL), dried over MgSO4, concentrated and purified on a silica gel column eluted with DCM to give the title compound (13.37 g, 88% yield): MS (ES+) m / z 304, 306 [M+H]+.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More