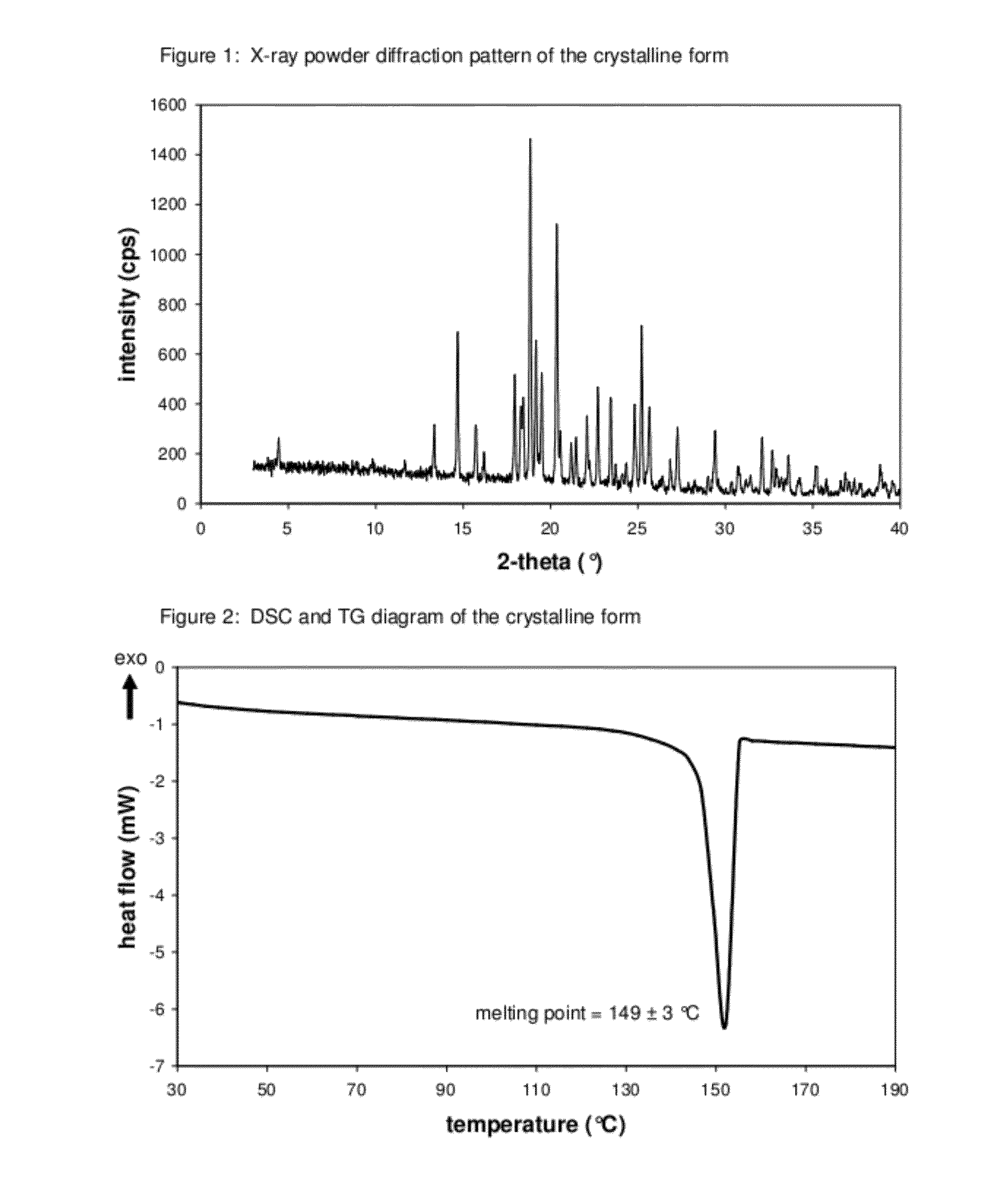
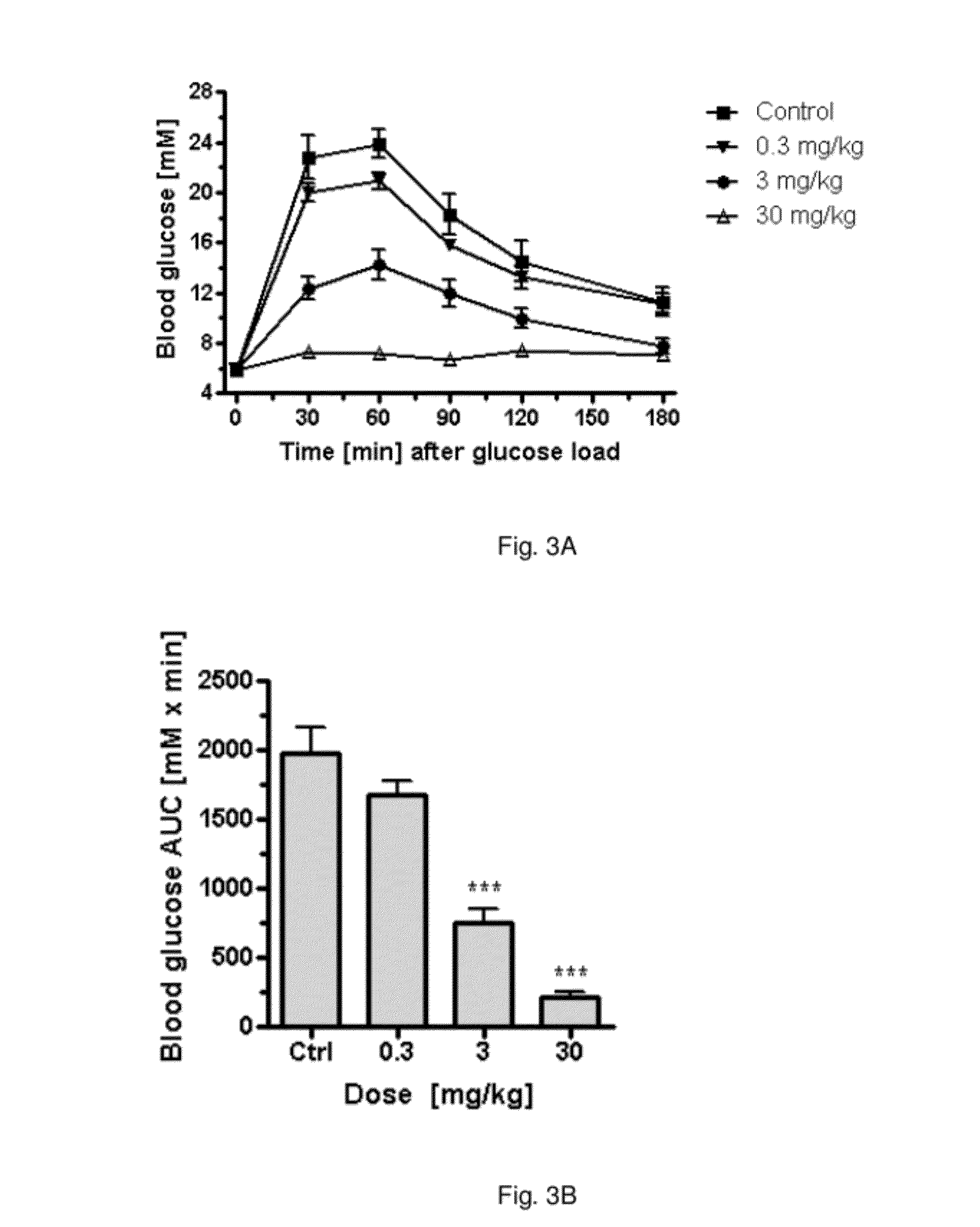
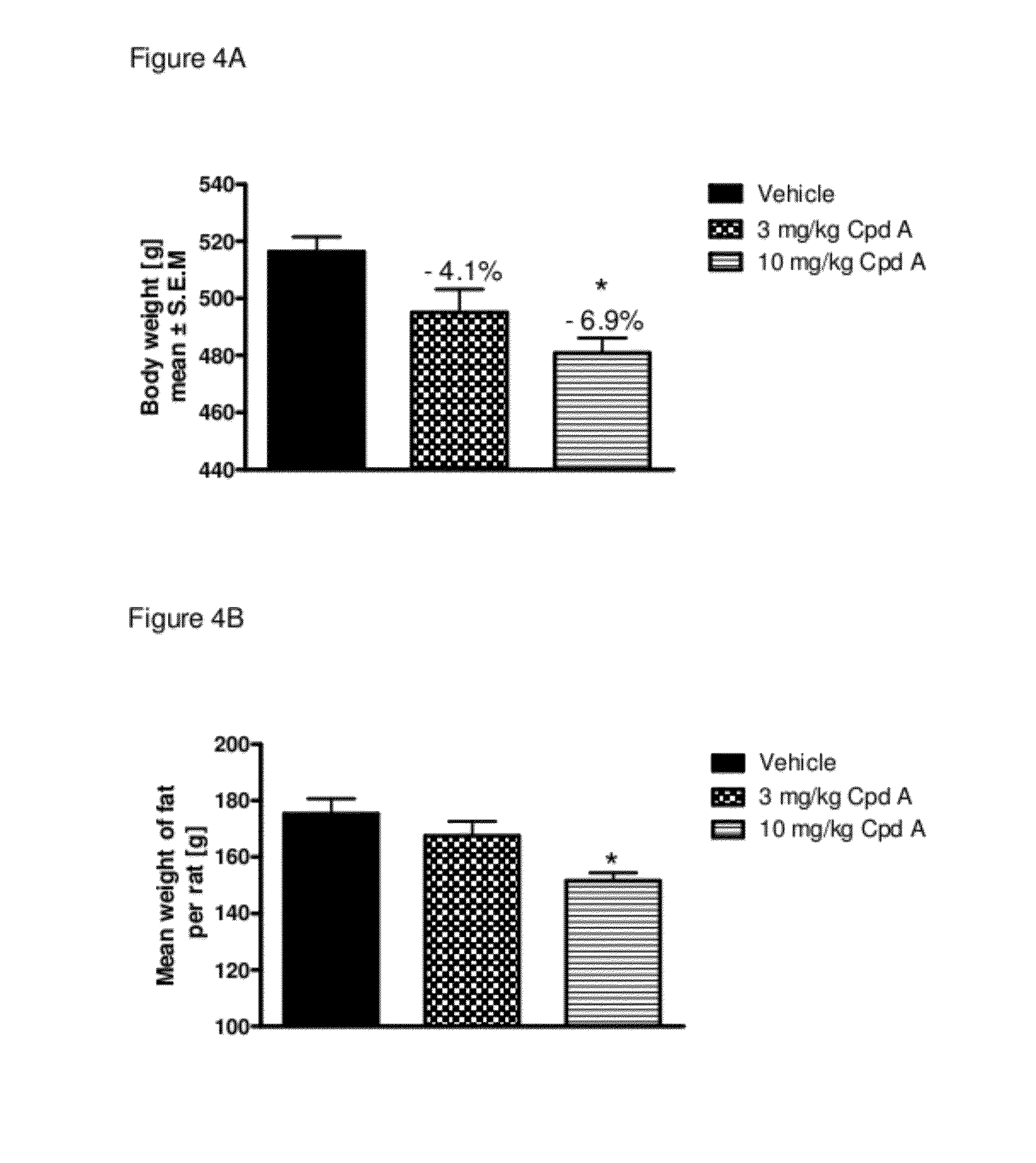
Pharmaceutical composition, methods for treating and uses thereof
a technology of pharmaceutical compositions and compositions, applied in the field of pharmaceutical compositions, can solve the problems of affecting the treatment effect, so as to improve the glycemic control, prevent delay or treat the treatment, and slow the progression of metabolic disorders
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 10a
Prevention of NODAT and / or PTMS, and NODAT / PTMS Associated Complications
[0294]Treatment of patients after organ transplantation with the pharmaceutical composition according to the invention prevents the development of NODAT and / or PTMS, and associated complications. The efficacy of the treatment can be investigated in a comparative clinical study in which patients before or immediately after transplantation are treated over a lengthy period (e.g. 1-5 years) with either a pharmaceutical composition according to this intervention or with a placebo or with a non-drug therapy or other medicaments. During and at the end of the therapy, the incidence of NODAT, PTMS, micro- and macrovascular complications, graft rejection, infection and death will be assessed. A significant reduction in the number of patients experiencing these complications demonstrates the efficacy in preventing development of NODAT, PTMS, and associated complications.
example 10b
Treatment of NODAT and / or PTMS with Prevention, Delay or Reduction of Associated Complications
[0295]Treatment of patients with NODAT and / or PTMS with the pharmaceutical composition according to the invention prevents, delays or reduces the development of NODAT / PTMS associated complications. The efficacy of the treatment can be investigated in a comparative clinical study in which patients with NODAT and / or PTMS are treated over a lengthy period (e.g. 1-5 years) with either a pharmaceutical composition according to this intervention or with a placebo or with a non-drug therapy or other medicaments. During and at the end of the therapy, the incidence of micro- and macrovascular complications, graft rejection, infection and death will be assessed. A significant reduction in the number of patients experiencing these complications demonstrates the efficacy in preventing, delaying or reducing the development of NODAT and / or PTMS associated complications.
example 11a
Treatment of Gestational Diabetes
[0296]In clinical studies running for a shorter period (e.g. 2-4 weeks) the success of the treatment is checked by determining the fasting glucose values and / or the glucose values after a meal or after a loading test (oral glucose tolerance test or food tolerance test after a defined meal) at the end of the therapeutic period of the study and comparing them with the values before the start of the study and / or with those of a placebo group. In addition, the fructosamine value can be determined before and after treatment and compared with the initial value and / or a placebo value. A significant fall in the fasting or non-fasting glucose levels demonstrates the pharmaceutical composition according to the invention.
[0297]In longer-running studies (12 weeks or more) the success of the treatment is checked by determining the HbA1c value (compared with initial value and placebo group). A significant change in the HbA1c value compared with the starting value ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| diastolic blood pressure | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More