Packaging system for pharmaceutical dispenser and associated method
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
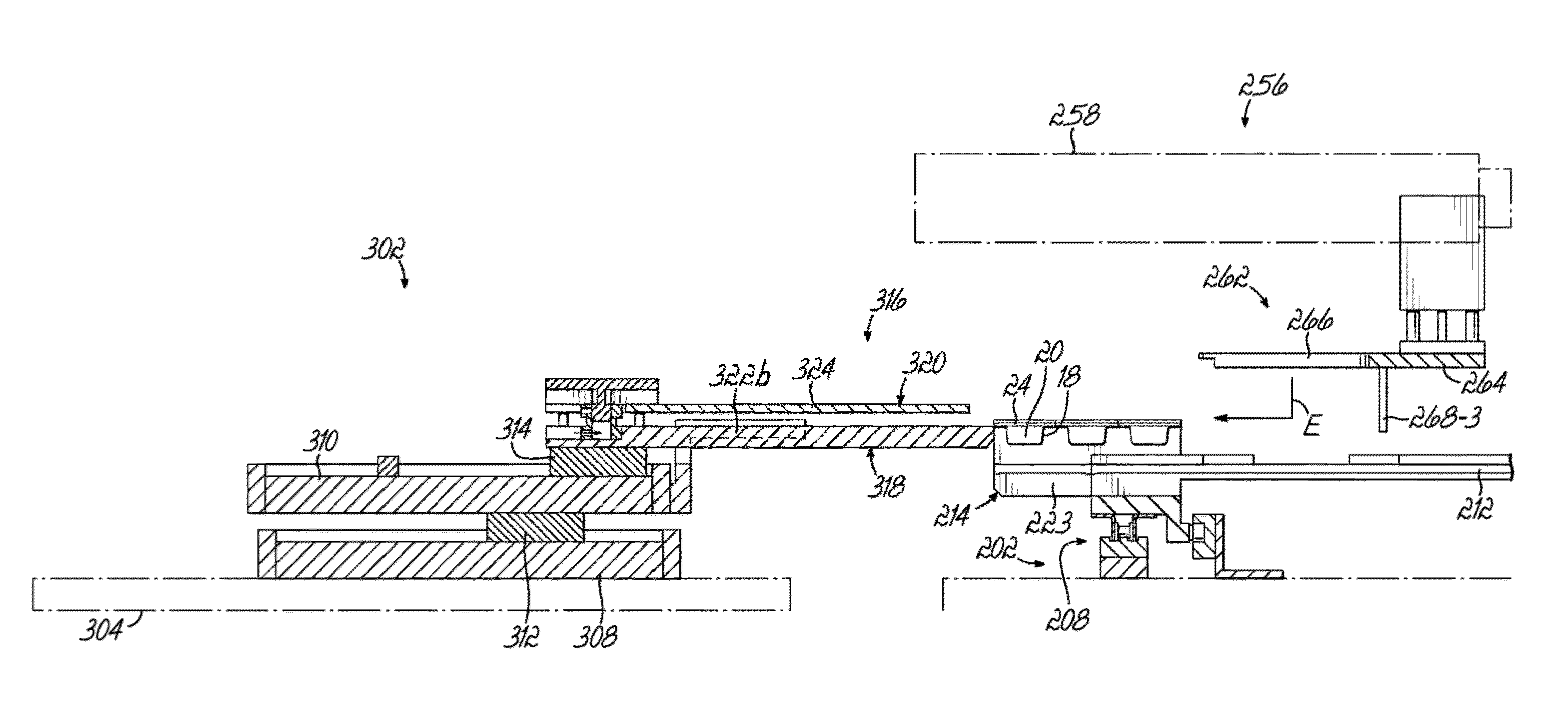
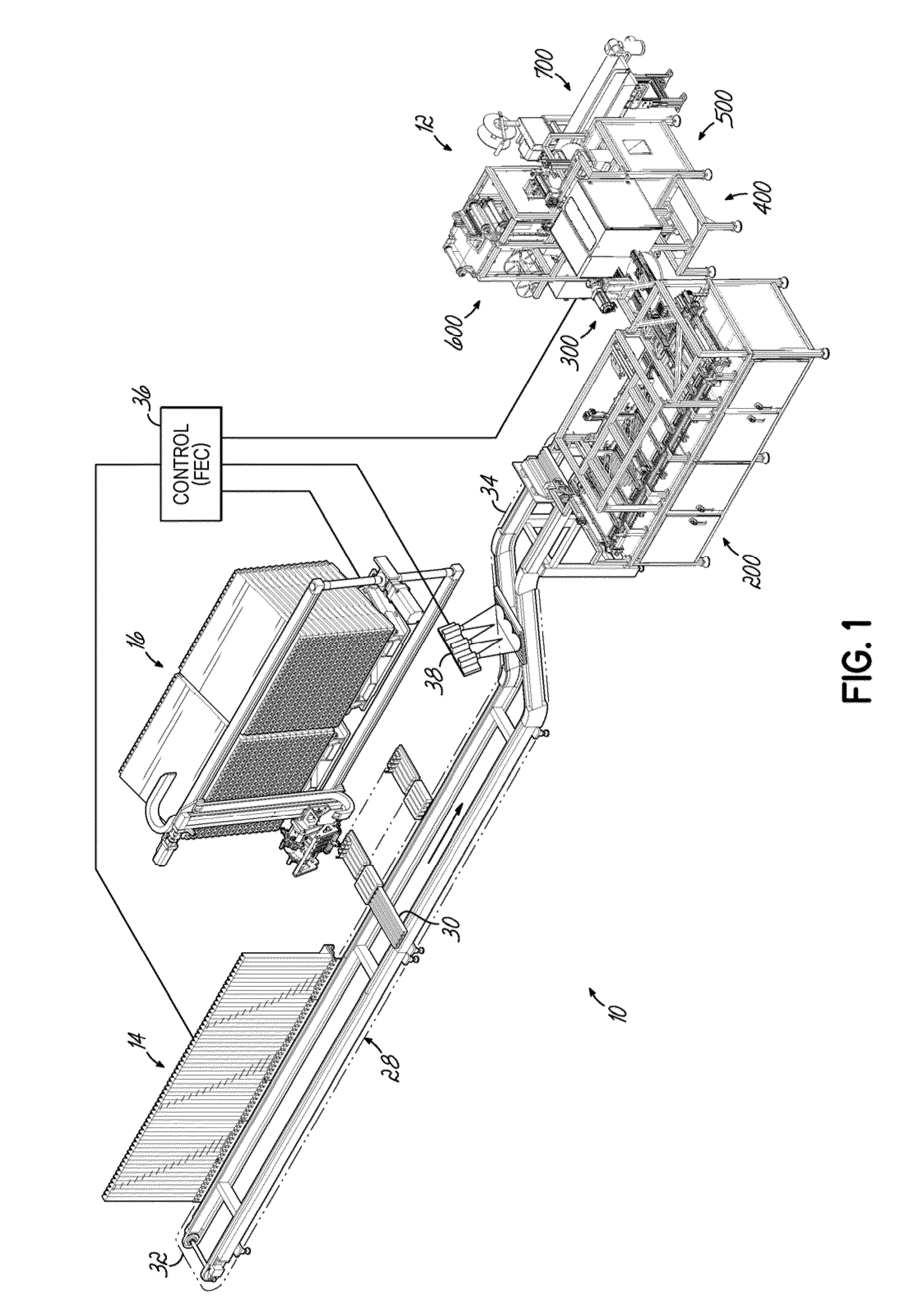
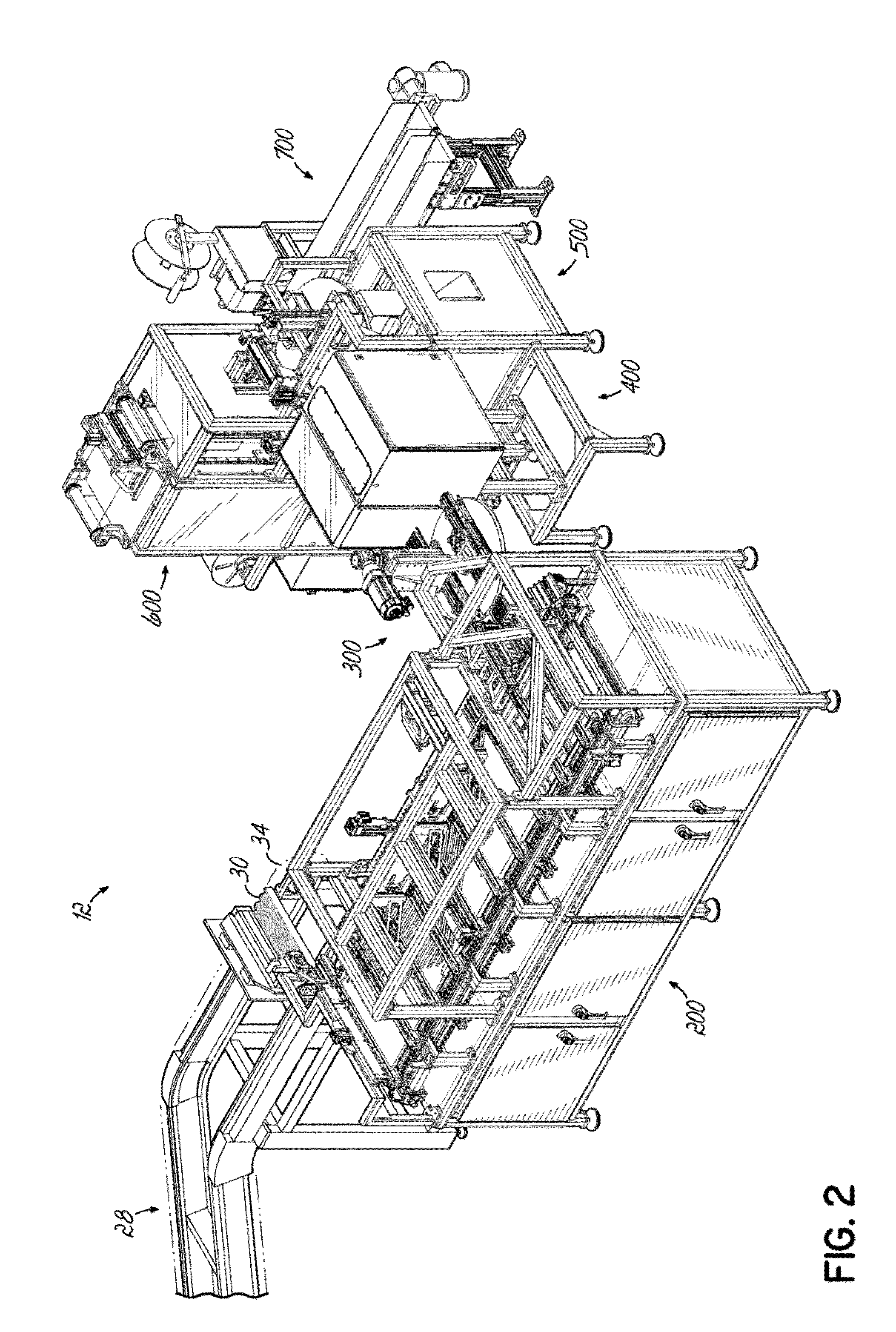
[0085]A dispensing system 10 according to one embodiment is shown in FIG. 1 and is configured to store and dispense individually packaged and labeled doses of medications / supplements, and to assemble the dispensed medications / supplements into individual medication orders, such as time-pass medication orders to be delivered to a long-term care (LTC) facility, for example. The dispensing system 10 shown and described herein is one example of such a system that can be utilized in conjunction with a packaging system 12 as shown and described herein. The dispensing system 10 of one embodiment is divided into distinct modules that are dedicated to dispensing the medications / supplements based on the demand, or order frequency, of those items. In the exemplary embodiment shown, a first module 14 is configured to dispense medications / supplements having a relatively high-demand or order frequency, and a second module 16 of the dispensing system 10 is configured to store and dispense medicatio...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More