Preparation of thiamphenicol
A thiamphenicol and sulfone-based technology is applied in the field of preparation of thiamphenicol, and can solve the problems of high production cost, low yield, long process route and the like
Inactive Publication Date: 2008-06-18
柯保桂
View PDF0 Cites 5 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0002] Its synthetic method such as US: 2816915 adopts p-toluenesulfonyl chloride through reduction, methylation, bromination, hydrolysis, copper salt, esterification, resolution, reduction, acylation, etc., and has long process route, low yield, Disadvantages such as high production costs
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment Construction
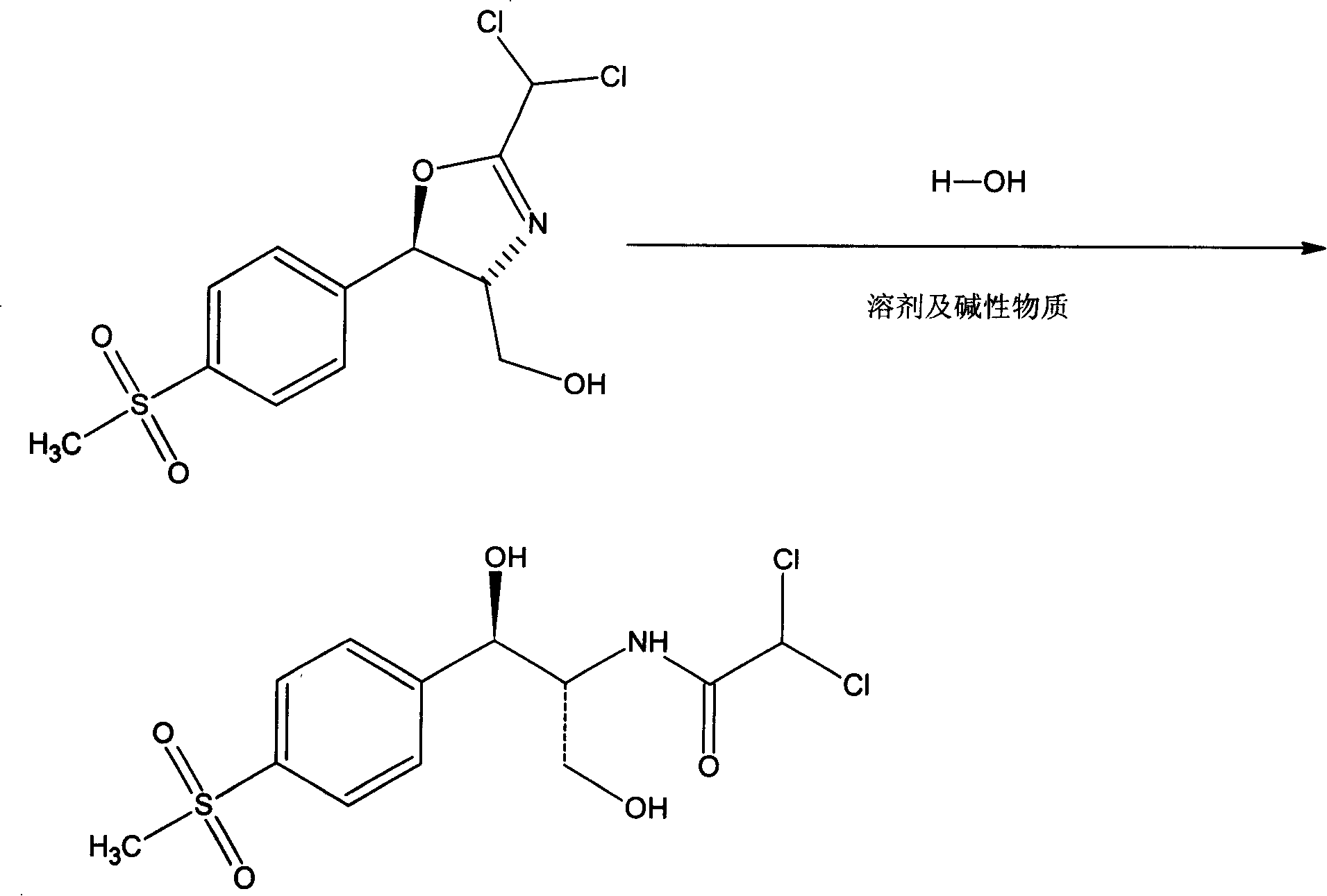
[0009] Add 100 g of D-threo-2-(dichloromethyl)-4,5-dihydro-5-[p-(thymphenyl)phenyl]-4-oxazole methanol to 280 g of isopropanol and water Add about 45g of sodium acetate to the solution of 360g to adjust the pH to 7-8, raise the temperature to 85°C, reflux for 4 hours, and distill under reduced pressure below 60°C. After the paste-like crude product is precipitated, cool to below 10°C, filter, and wash with water Dried three times to obtain 101 g of thiamphenicol with a yield of 95.9%, which was consistent with the standard product through infrared spectrum detection, and the content was determined to be 99.5% by high performance liquid chromatography.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention relates to a preparation method of thiamphenicol by one step synthesis from hydrolysis of D-thero-2-(dichloromethyl)-4,5-dihydro-[p-methylsulfonyl phenyl]-4-oxazole methyl alcohol.
Description
technical field [0001] The invention belongs to the field of heterocyclic compounds. Thiamphenicol is a broad-spectrum antibiotic and can be used to treat infections caused by Gram-positive, Gram-negative and rickettsia. The invention relates to the preparation of thiamphenicol method. Background technique [0002] Its synthesis method such as US: 2816915 is prepared by using p-toluenesulfonyl chloride through reduction, methylation, bromination, hydrolysis, copper salt, esterification, resolution, reduction, acylation, etc. It has long process route, low yield, Disadvantages such as high production costs. Contents of the invention [0003] Aiming at the above-mentioned shortcomings, we have devoted ourselves to research and invented D-threo-2-(dichloromethyl)-4,5-dihydro-5-[p-(thysulfonyl)phenyl]-4-oxazolemethanol A preparation method for synthesizing thiamphenicol in one step by hydrolysis. Due to the mass production of dichloroacetonitrile, the main raw material for ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C07C317/32C07C315/04
Inventor 柯保桂
Owner 柯保桂