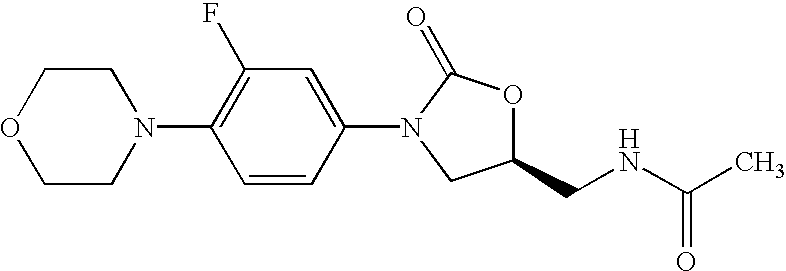
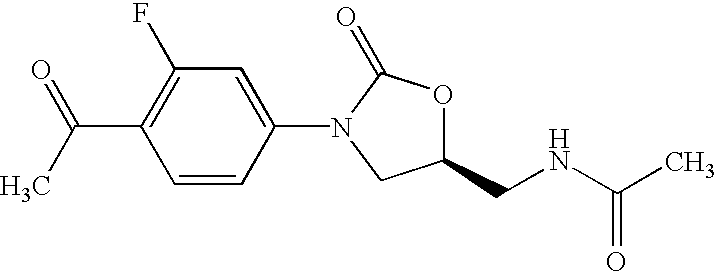
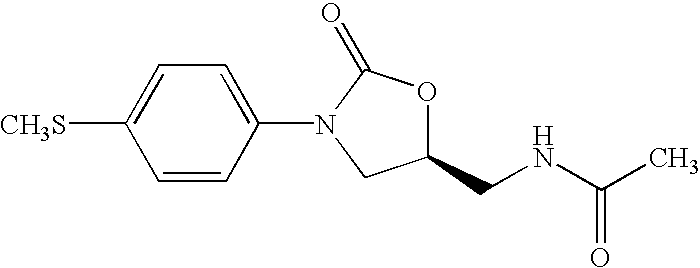
Patents
Literature
1465 results about "Oxazole" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Oxazole is the parent compound for a vast class of heterocyclic aromatic organic compounds. These are azoles with an oxygen and a nitrogen separated by one carbon. Oxazoles are aromatic compounds but less so than the thiazoles. Oxazole is a weak base; its conjugate acid has a pKₐ of 0.8, compared to 7 for imidazole.
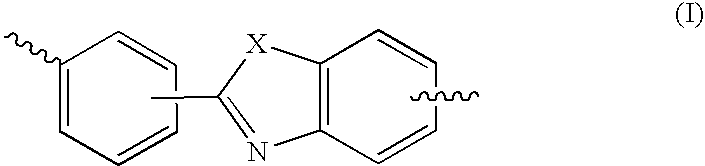
Oxazolo, thiazolo and selenazolo [4,5-c]-quinolin-4-amines and analogs thereof
Thiazolo-, oxazolo- and selenazolo[4,5-c]quinolin-4-amines and analogs thereof are described including methods of manufacture and the use of novel intermediates. The compounds are immunomodulators and induce cytokine biosynthesis, including interferon and / or tumor biosynthesis, necrosis factor, and inhibit the T-helper-type 2 immune response. The compounds are further useful in the treatment of viral and neoplastic diseases.
Owner:3M INNOVATIVE PROPERTIES CO
Thiazoles and oxazoles useful as modulators of ATP-Binding Cassette transporters
The present invention relates to modulators of ATP-Binding Cassette (“ABC”) transporters or fragments thereof, including Cystic Fibrosis Transmembrane Regulator (“CFTR”), compositions thereof, and methods therewith. The present invention also relates to methods of treating ABC transporter mediated diseases using such modulators.
Owner:VERTEX PHARMA INC
Nicotinamide acids, amides, and their mimetics active as inhibitors of PDE4 isozymes
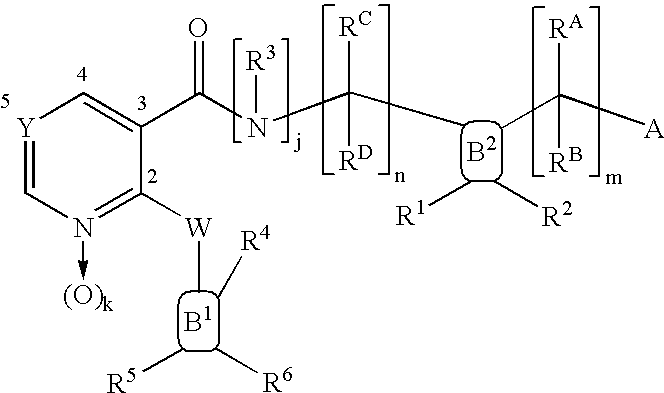
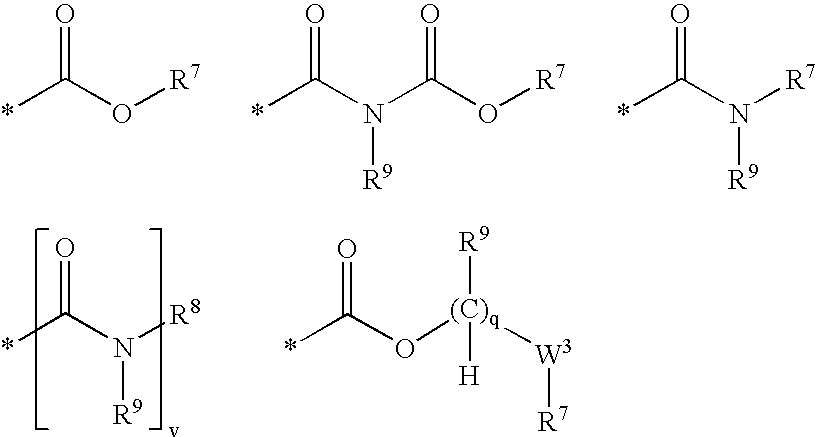
Compounds useful as inhibitors of PDE4 in the treatment of diseases regulated by the activation and degranulation of eosinophils, especially asthma, chronic bronchitis, and chronic obstructuive pulmonary disease, of the formula: wherein j is 0 or 1, k is 0 or 1, m is 0, 1, or 2; n is 1 or 2; A is selected from the partial Formulas: where q is 1, 2, or 3, W3 is -O-; -N(R9)-; or -OC(=O)-; R7 is selected from -H; -(C1-C6) alkyl, -(C2-C6) alkenyl, or -(C2-C6) alkynyl substituted by 0 to 3 substituents R10; -(CH2)u-(C3-C7) cycloalkyl where u is 0, 1 or 2, substituted by 0 to 3 R10; and phenyl or benzyl substituted by 0 to 3 R14; R8 is tetrazol-5-yl; 1,2,4-triazol-3-yl; 1,2,4-triazol-3-on-5-yl; 1,2,3-triazol-5-yl; imidazol-2-yl; imidazol-4-yl; imidazolidin-2-on-4-yl; 1,3,4-oxadiazolyl; 1,3,4-oxadiazol-2-on-5-yl; 1,2,4-oxadiazol-3-yl; 1,2,4-oxadiazol-5-on-3-yl; 1,2,4-oxadiazol-5-yl; 1,2,4-oxadiazol-3-on-5-yl; 1,2,5-thiadiazolyl; 1,3,4-thiadiazolyl; morpholinyl; parathiazinyl; oxazolyl; isoxazolyl; thiazolyl; isothiazolyl; pyrrolyl; pyrazolyl; succinimidyl; glutarimidyl; pyrrolidonyl; 2-piperidonyl; 2-pyridonyl; 4-pyridonyl; pyridazin-3-onyl; pyridyl; pyrimidinyl; pyrazinyl; pyridazinyl; indolyl; indolinyl; isoindolinyl; benzo[b]furanyl; 2,3-dihydrobenzofuranyl; 1,3-dihydroisobenzofuranyl; 2H-1-benzopyranyl; 2-H-chromenyl; chromanyl; benzothienyl; 1H-indazolyl; benzimidazolyl; benzoxazolyl; benzisoxazolyl; benzothiazolyl; benzotriazolyl; benzotriazinyl; phthalazinyl; 1,8-naphthyridinyl; quinolinyl; isoquinolinyl; quinazolinyl; quinoxalinyl; pyrazolo[3,4-d]pyrimidinyl; pyrimido[4,5-d]pyrimidinyl; imidazo[1,2-a]pyridinyl; pyridopyridinyl; pteridinyl; or 1H-purinyl; or A is selected from phosphorous and sulfur acid groups; W is -O-; -S(=O)t-, where t is 0, 1, or 2; or -N(R3)-; Y is =C(R1a)-, or -[N<custom-character file="US20020111495A1-20020815-P00900.TIF" wi="20" he="20" id="custom-character-00001" / >(O)k] where k is 0 or 1; R4, R5 and R6 are (1) -H; provided that R5 and R6 are not both -H at the same time, -F; -Cl; -(C2-C4) alkynyl; -R16; -OR16; -S(=O)pR16; -C(=O)R16, -C(=O)OR16, -C(=O)OR<highlight><sup
Owner:PFIZER INC
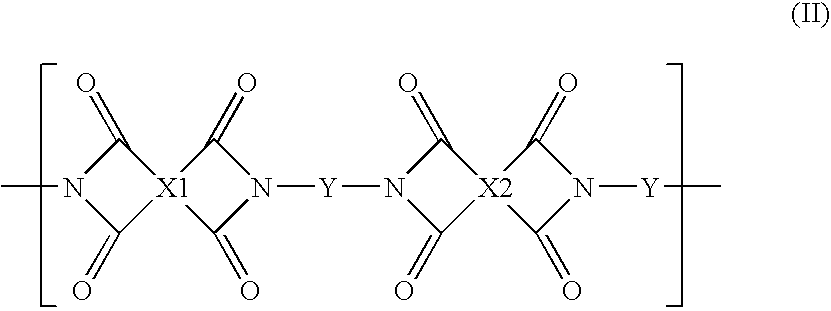
High performance cross-linked polybenzoxazole and polybenzothiazole polymer membranes
In the present invention high performance cross-linked polybenzoxazole and polybenzothiazole polymer membranes and methods for making and using these membranes have been developed. The cross-linked polybenzoxazole and polybenzothiazole polymer membranes are prepared by: 1) first synthesizing polyimide polymers comprising pendent functional groups (e.g., —OH or —SH) ortho to the heterocyclic imide nitrogen and cross-linkable functional groups in the polymer backbone; 2) fabricating polyimide membranes from these polymers; 3) converting the polyimide membranes to polybenzoxazole or polybenzothiazole membranes by heating under inert atmosphere such as nitrogen or vacuum; and 4) finally converting the membranes to high performance cross-linked polybenzoxazole or polybenzothiazole membranes by a crosslinking treatment, preferably UV radiation. The membranes can be fabricated into any convenient geometry. The high performance cross-linked polybenzoxazole and polybenzothiazole polymer membranes of the present invention are suitable for a variety of liquid, gas, and vapor separations.
Owner:UOP LLC
High performance cross-linked polybenzoxazole and polybenzothiazole polymer membranes
InactiveUS8127936B2High selectivityImprove stabilitySemi-permeable membranesMembranesImidePolymer science
In the present invention high performance cross-linked polybenzoxazole and polybenzothiazole polymer membranes and methods for making and using these membranes have been developed. The cross-linked polybenzoxazole and polybenzothiazole polymer membranes are prepared by: 1) first synthesizing polyimide polymers comprising pendent functional groups (e.g., —OH or —SH) ortho to the heterocyclic imide nitrogen and cross-linkable functional groups in the polymer backbone; 2) fabricating polyimide membranes from these polymers; 3) converting the polyimide membranes to polybenzoxazole or polybenzothiazole membranes by heating under inert atmosphere such as nitrogen or vacuum; and 4) finally converting the membranes to high performance cross-linked polybenzoxazole or polybenzothiazole membranes by a crosslinking treatment, preferably UV radiation. The membranes can be fabricated into any convenient geometry. The high performance cross-linked polybenzoxazole and polybenzothiazole polymer membranes of the present invention are suitable for a variety of liquid, gas, and vapor separations.
Owner:UOP LLC
Oxazolo, thiazolo and selenazolo [4,5-c] quinolin-4-amines and analogs thereof
Thiazolo-, oxazolo- and selenazolo[4,5-c]quinolin-4-amines and analogs thereof are described including methods of manufacture and the use of novel intermediates. The compounds are immunomodulators and induce cytokine biosynthesis, including interferon and / or tumor biosynthesis, necrosis factor, and inhibit the T-helper-type 2 immune response. The compounds are further useful in the treatment of viral and neoplastic diseases.
Owner:3M INNOVATIVE PROPERTIES CO
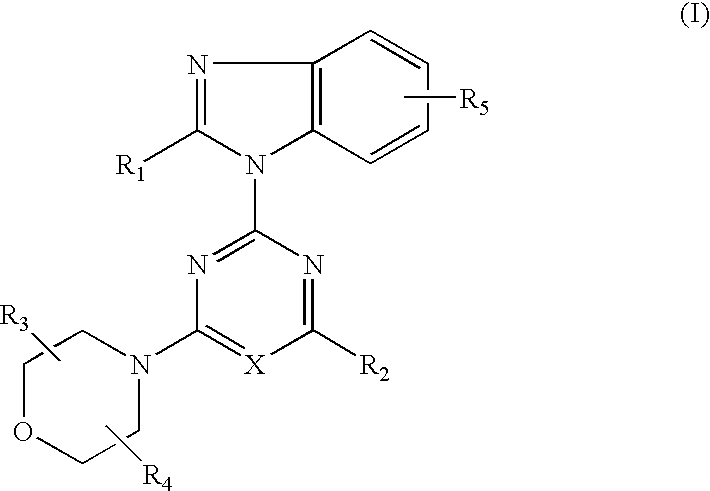
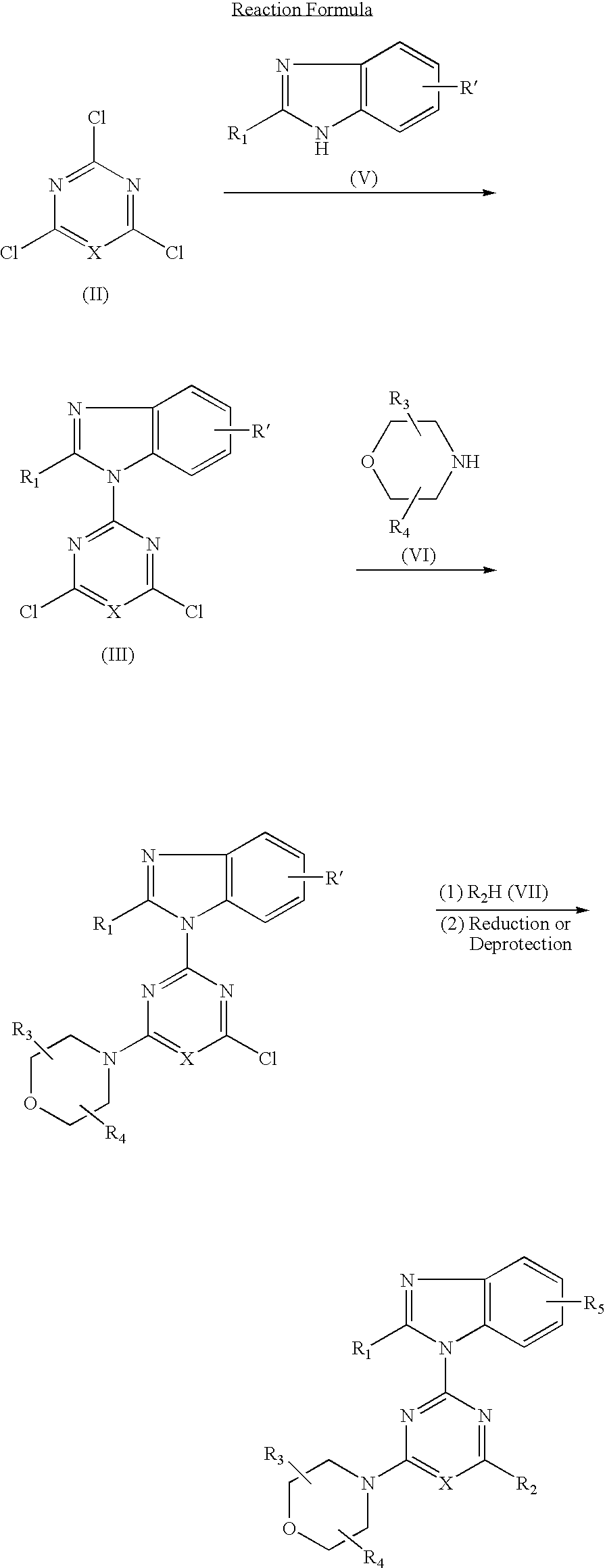
Heterocyclic compound and antitumor agent containing the same as active ingredient
The present invention relates to heterocyclic compounds represented by the formula I or pharmaceutically acceptable salts thereof and antitumor agents containing the heterocyclic compounds as effective components:wherein X represents nitrogen atom or CH; R1 represents CHnF3-n (wherein n is 1 or 2), hydroxy C1–C6 alkyl, NHR6 [wherein R6 represents hydrogen atom or COR (wherein R represents hydrogen atom, C1–C6 alkyl or C1–C6 alkoxy)]; R2 represents morpholino (which may be substituted with one to four C1–C6 alkyl), thiomorpholino, piperidino, pyrrolidinyl (which may be substituted with hydroxy C1–C6 alkyl), oxazolidinyl (which may be substituted with one or two C1–C6 alkyl) or tetrahydro-1,4-thiazin-1-oxo-4-yl; R3 and R4 each represent hydrogen atom or C1–C6 alkyl; and R5 represents hydrogen atom, amino or hydroxyl.
Owner:OHARA PHARMA
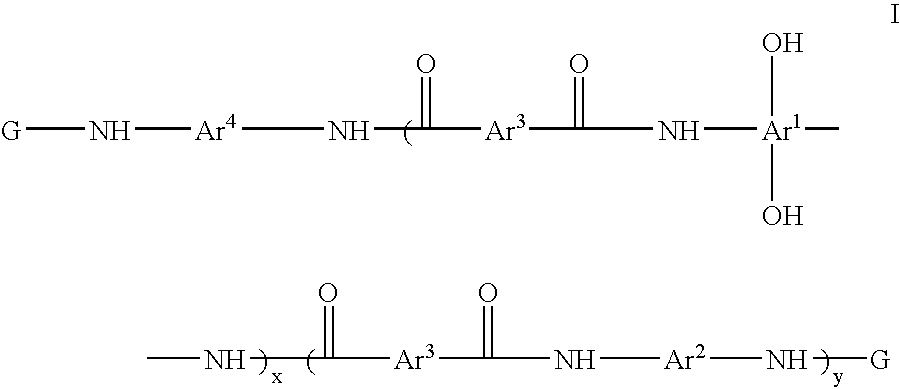
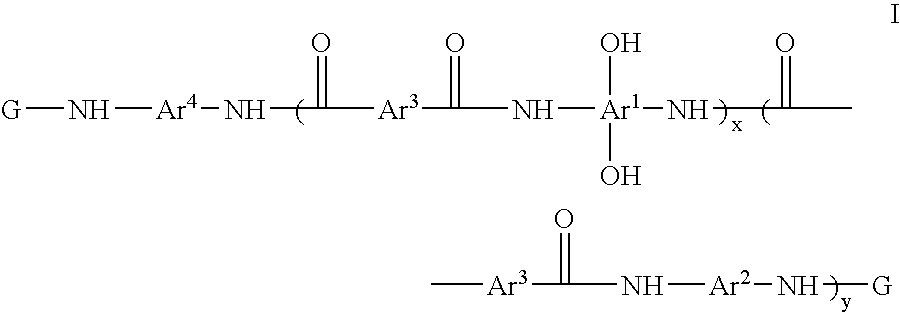
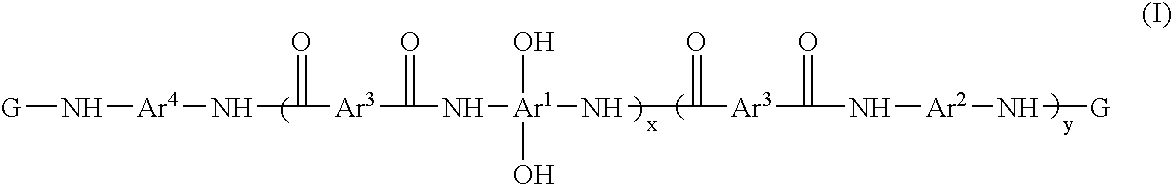
Photosensitive resin compositions
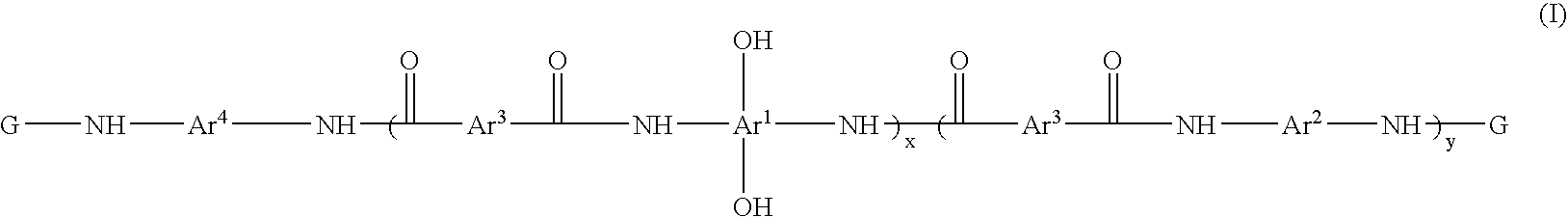
A heat resistant negative working photosensitive composition that comprises(a) one or more polybenzoxazole precursor polymers (I): wherein x is an integer from about 10 to about 1000, y is an integer from 0 to about 900 and (x+y) is about less then 1000; Ar1 is selected from the group consisting of a tetravalent aromatic group, a tetravalent heterocyclic group, or mixtures thereof; Ar2 is selected from the group consisting a divalent aromatic, a divalent heterocyclic, a divalent alicyclic, a divalent aliphatic group that may contain silicon, or mixtures thereof; Ar3 is selected from the group consisting a divalent aromatic group, a divalent aliphatic group, a divalent heterocyclic group, or mixtures thereof; Ar4 is selected from the group consisting Ar1 (OH)2 or Ar2; G is an organic group selected from the group consisting groups having a carbonyl, carbonyloxy or sulfonyl group attached directly to the terminal NH group of the polymer;(b) one or more photo-active compounds which release acid upon irradiation (PAGs);(c) a latent crosslinker which contains at least two ˜N—(CH2OR)n units wherein n=1 or 2 and R is a linear or branched C1-C8 alkyl group, with the proviso that when a glycoluril is employed as the latent crosslinker, the G group in the polybenzoxazole precursor polymer is produced from the reaction of a cyclic anhydride; and(d) at least one solvent that is not NMP.
Owner:FUJIFILM ELECTRONICS MATERIALS US
Organic electroluminescent device and method for preparing the same
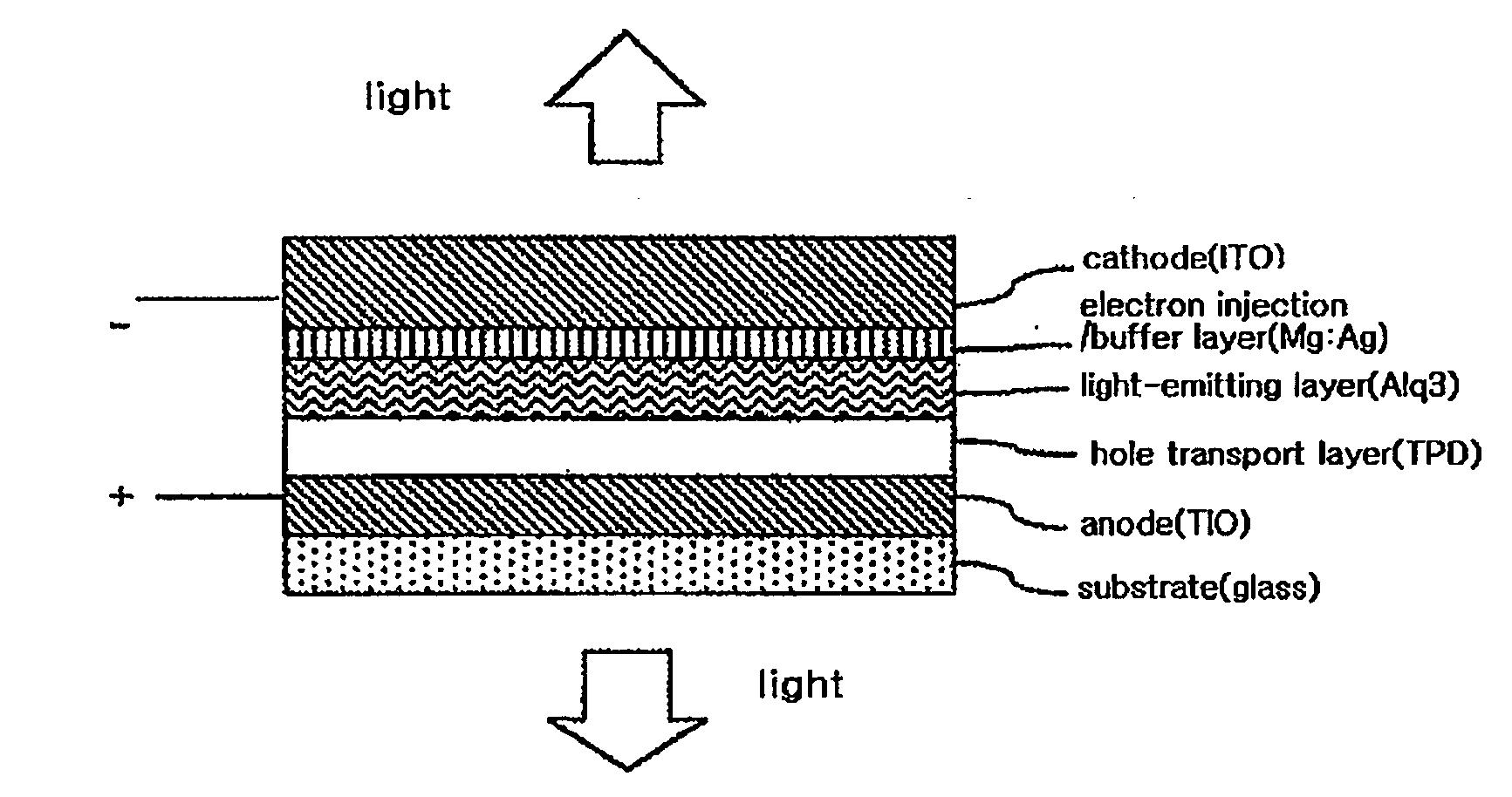
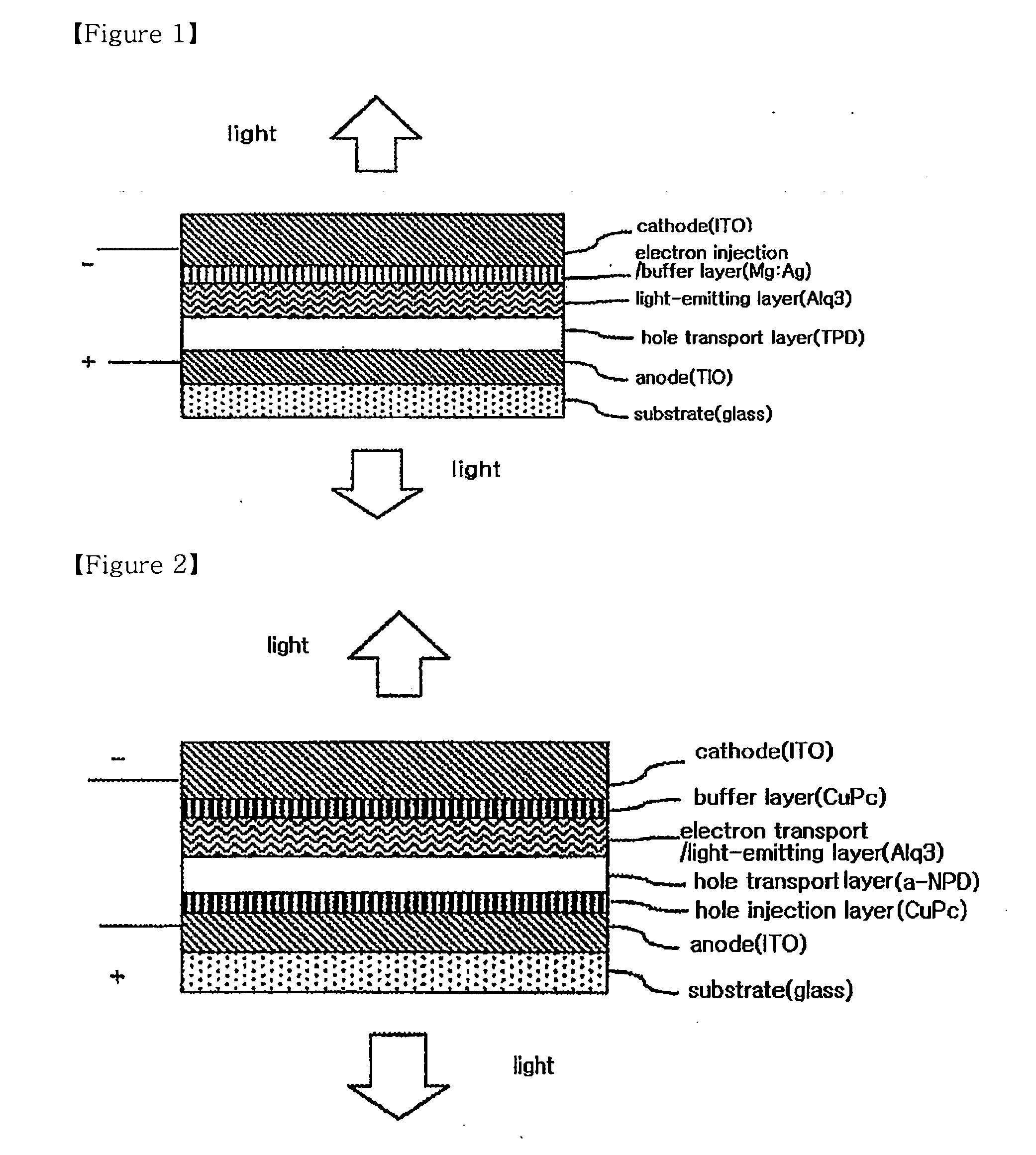
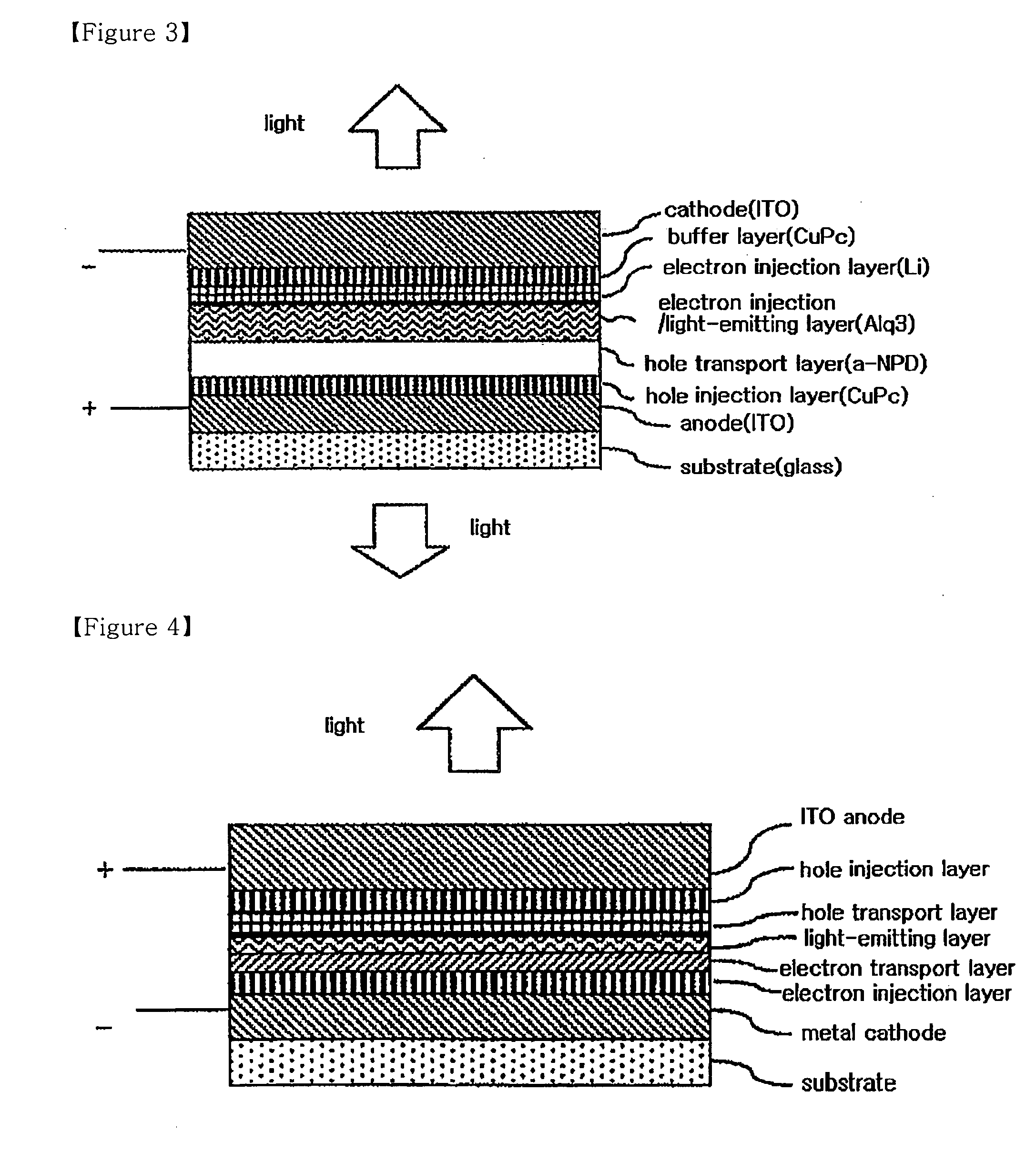
InactiveUS20080284325A1Improved electron injection characteristicAvoid damageDischarge tube luminescnet screensLamp detailsThiazoleElectron injection
The present invention relates to an organic electroluminescent device comprising a substrate, a cathode, at least two organic material layers comprising a light-emitting layer, and an anode in the sequentially laminated form, in which the organic material layers comprise an organic material layer comprising a compound having a functional group selected from the group consisting of an imidazole group, an oxazole group and a thiazole group between the cathode and the light-emitting layer. The organic electroluminescent device according to the present invention comprises an organic material layer comprising a compound having a functional group selected from the group consisting of an imidazole group, an oxazole group and a thiazole group between a cathode and a light-emitting layer, thus having an improved electron injection characteristic to provide an organic electroluminescent device of an inverted structure operating at a low voltage.
Owner:LG CHEM LTD
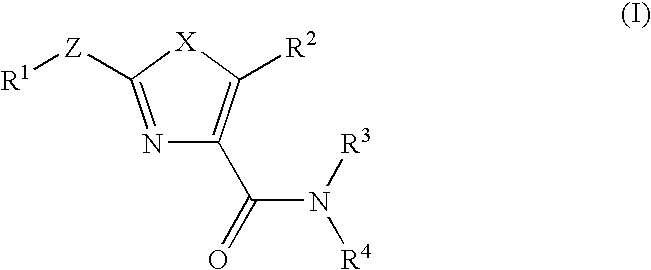
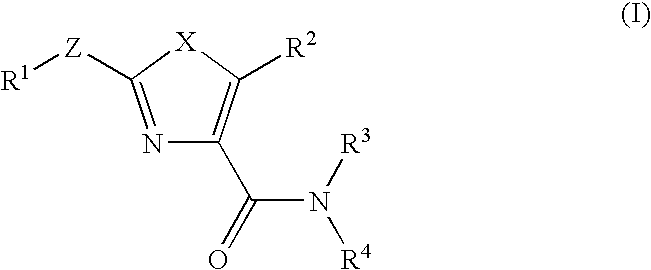
Oxazole compound and pharmaceutical composition
The present invention provides a oxazole compound represented by Formula (1), or a salt thereof:wherein R1 is an aryl group which may have one or more substituents; R2 is an aryl group or a nitrogen atom-containing heterocyclic group each of which may have one or more substituents; and W is a divalent group represented by —Y1-A1- or —Y2—C(═O)— wherein Y1 is a group such as —C(═O)—, A1 is a group such as a lower alkylene group, and Y2 is a group such as a piperazinediyl group. The oxazole compound has a specific inhibitory action against phosphodiesterase 4.
Owner:OTSUKA PHARM CO LTD
Binder composition for fuel cell, membrane-electrode assembly for fuel cell, and method for preparing the membrane-electrode assembly
InactiveUS20070020507A1Improve bindingImprove proton conductivityMaterial nanotechnologyElectrode manufacturing processesBenzoxazoleProton conductor
The present invention relates to a binder composition for a fuel cell including a proton conductor and one or more binders selected from the group consisting of poly[2,2′-(m-phenylene)-5,5′-bibenzimidazole] (PBI), poly[2,5-benzimidazole] (ABPBI), polybenzoxazole (PBO), and polybenzothiazole (PBT).
Owner:SAMSUNG SDI CO LTD
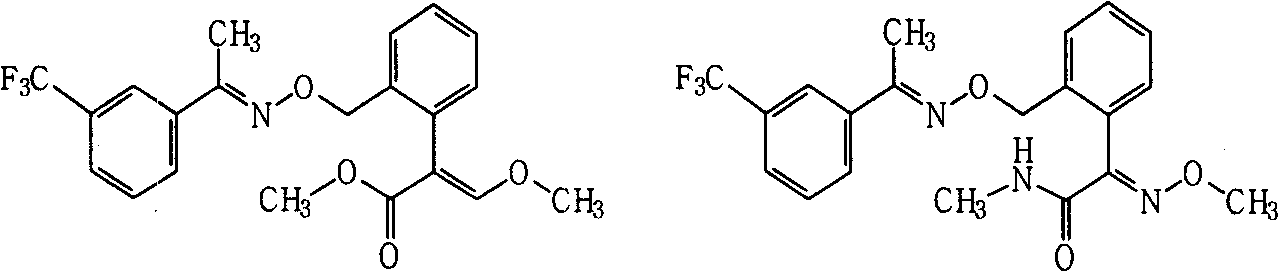
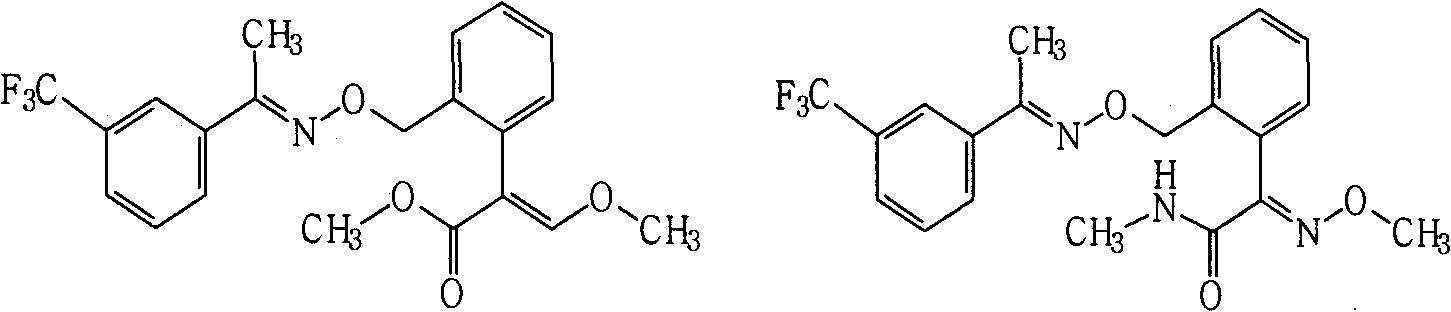
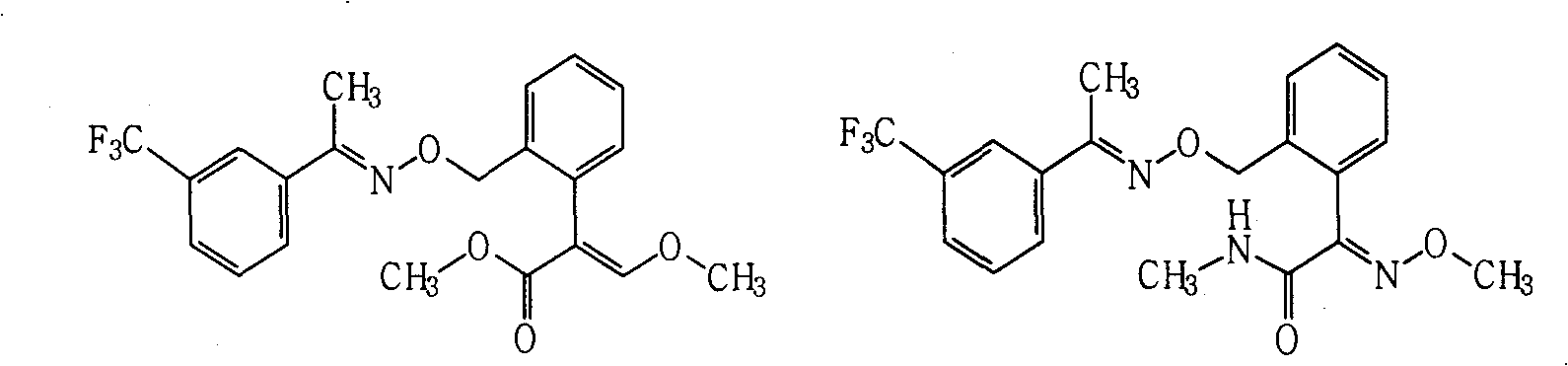
Fungicide composition
ActiveCN101637157AGood synergyImprove the effect of disease preventionBiocideFungicidesThiocarbamateCarbamate
The invention provides a fungicide composition containing an active component A and an active component B, and application thereof in preventing and controlling various agricultural fungal diseases. The active component A in the composition is one of two methoxyl acrylic ester compounds; and as shown above, the active component B of the compound A1 and the compound A2 is one of the following fungicide varieties: thiocarbamate compounds or salt thereof, aliphatic compounds or salt thereof, carbamate compounds or salt thereof, oxazole compounds or salt thereof, thiazole compounds or salt thereof, amide compounds or salt thereof, organophosphorus compounds or salt thereof, imidazole compounds or salt thereof, antibiotic compounds or salt thereof, pyridine compounds or salt thereof and triazole compounds or salt thereof.
Owner:SHENYANG SINOCHEM AGROCHEMICALS R&D CO LTD
Dibenzoxazine containing oxazole ring and preparation method thereof
InactiveCN102250117ASimple preparation processLow equipment requirementsLiquid crystal compositionsOrganic chemistrySolventOrtho-aminophenol
The invention relates to a dibenzoxazine containing an oxazole ring and a preparation method thereof. The preparation method comprises the following two steps of: 1: mixing ortho-aminophenol hydrochloride and para-hydroxybenzoic acid, adding polyphosphoric acid as a solvent, reacting at 60-180 DEG C for 24-60 hours, and washing by using deionized water, filtering and drying to obtain diphenol containing an oxazole ring structure; and 2: mixing the diphenol containing the oxazole ring structure, phenylamine and paraformaldehyde, reacting at 80-110 DEG C for 40-80 minutes, then filtering and precipitating, washing 4-8 times by using alkali liquor, and then washing, filtering and drying to obtain the product. The dibenzoxazine disclosed by the invention has the advantages of very good mechanical property because the high temperature resistant oxazole ring structure is introduced to a benzoxazine molecule structure, dielectric constant of only 1.6-2.3, simple process, lower equipment requirement and suitability for large-scale production.
Owner:EAST CHINA UNIV OF SCI & TECH
Cosmetic or dermatological light-protective formulation comprising a bisresorcinyl triazine derivative and a benzoxazole derivative
InactiveUS20050013782A1Improves UV protectionMaintain good propertiesCosmetic preparationsToilet preparationsBenzoxazoleChemical structure
The invention is a cosmetic or dermatological preparation effective as light-protective, comprising (a) at least one bis-resorcinyltriazine derivative and (b) at least one benzoxazole derivative. The invention is also a cosmetic or dermatological preparation comprising at least one bis-resorcinyltriazine derivative and at least one benzoxazole derivative of a specified chemical structure. The invention is also a method of treating or preventing cosmetic or dermatological changes in the skin, a method of tanning or accelerating tanning of the skin, and a method of protecting the skin against light-induced aging, each comprising applying the preparation to the skin. The invention also includes a wipe impregnated with the preparation.
Owner:BEIERSDORF AG
Fluorescent ion probe and its application in ion detecting
InactiveCN101153848AEasy to synthesizeSimple structureChemiluminescene/bioluminescenceLuminescent compositionsBenzoxazoleFluorophore
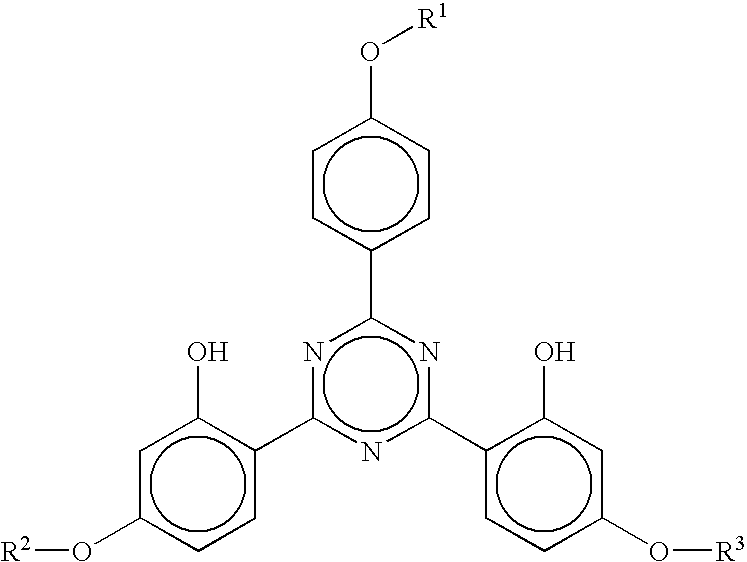
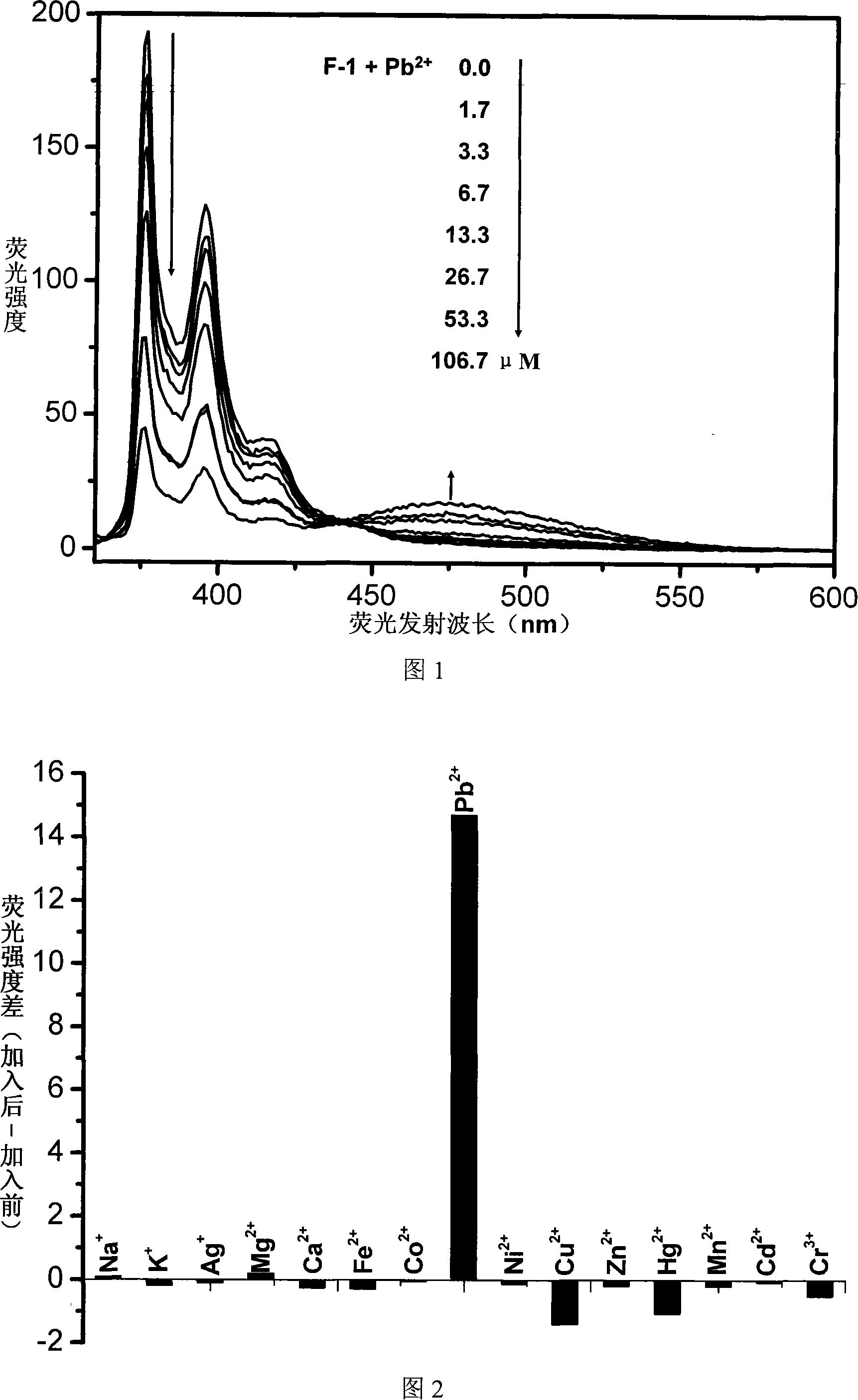
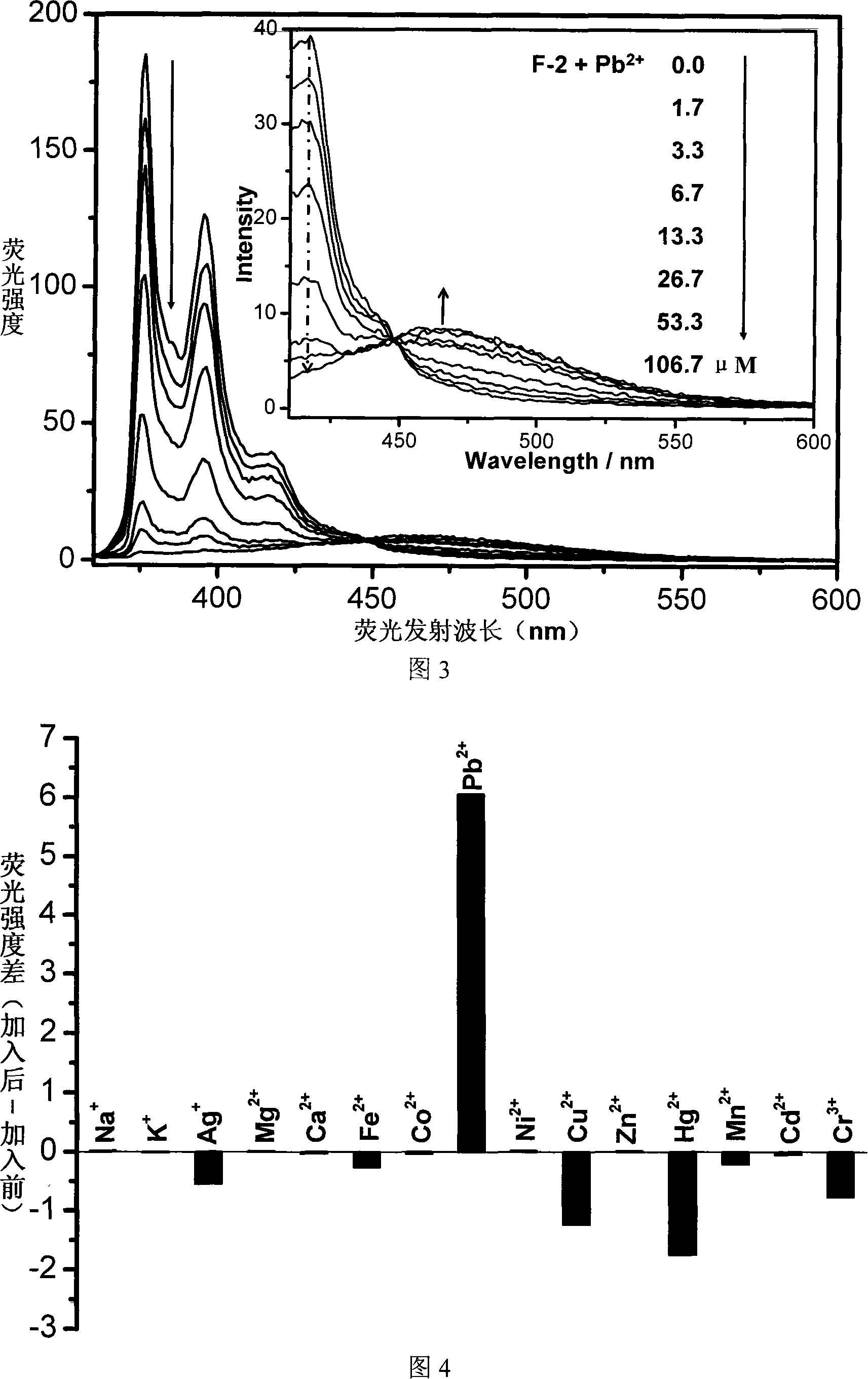
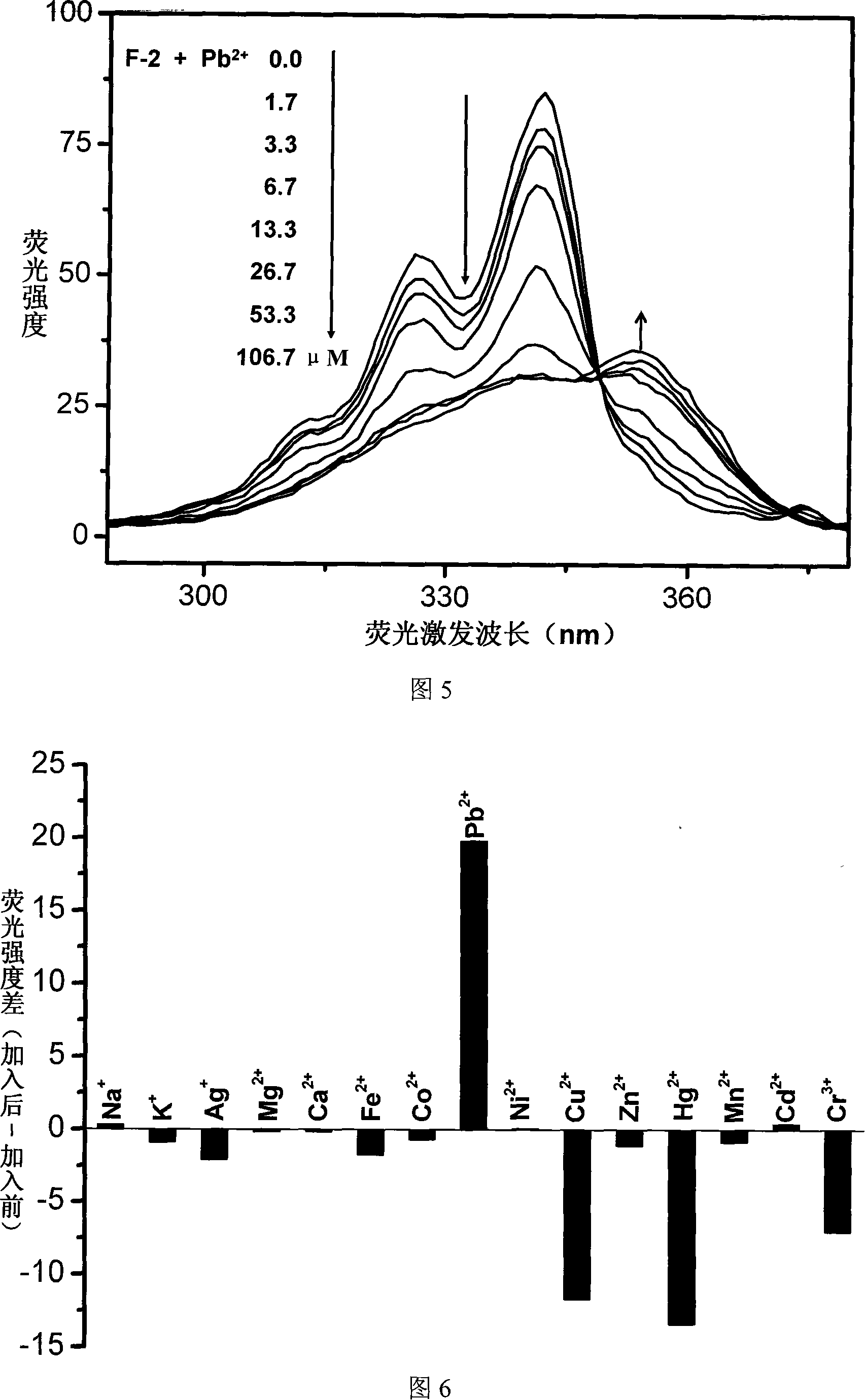
The invention relates to a fluorescent ion probe (I) with high selectivity and high sensitivity in detection and the application of the fluorescent ion probe in identifying and detecting heavy metal ion and transition metal ion, wherein, the Y is an organic conjugate group with fluorescence transmitting function such as pyrene, naphthalene, 4-amidogen-1, 8-naphthyl imide, Dan sulfonamide, anthracene, carbazole, benzimidazoins, benzoxazoles, boron fluoride bipyrrole (BODIPY), fluorescein, 3, 4, 9, 10-perylenetetracarboxylic diimide or rhodamine B; the X is acylamino group, sulfoamino group or ester group. The fluorescent ion probe uses the fluorescence peak of fluorescence chromophore aggregate as the response signal to identify metal ion, thereby effectively avoiding the quenching effect of transition metal ion and heavy metal ion on fluorophore; moreover, the fluorescent ion probe realizes selective identification of heavy metal ion and transition metal ion in various solvents and aqueous solution in particular.
Owner:JILIN UNIV
Sterilization compound containing chloroisobromine cyanuric acid
InactiveCN102823603ADelay drug resistanceExpanded bactericidal spectrumBiocideFungicidesCarbamateMorpholine
The invention discloses a sterilization compound containing chloroisobromine cyanuric acid, comprising a raw medicine formed by compounding a sterilizing agent A and a sterilizing agent B, wherein the proportion of the sterilizing agent A to the sterilizing agent B is (1-80):(1-80); the sterilizing agent A is the chloroisobromine cyanuric acid; and the sterilizing agent B is any one of the following types of the sterilizing agents: copper preparations, methoxy acrylic esters, triazoles, amides, benzimidazoles, thiocarbamates, substituted benzenes, dicarboximides, phthalimides, imidazoles, morpholines, carbamates, oxazoles and the like. The sterilization compound is formed by compounding the sterilizing agent A and the sterilizing agent B so that not only a sterilization spectrum is enlarged, but also the drug resistance of bacteria is slowed; and the sterilization compound has an obvious lasting synergistic effect.
Owner:王学权
Imazalil-containing sterilization composition
InactiveCN103918691ADelay drug resistanceExpanded bactericidal spectrumBiocideFungicidesCarbamateMorpholine
The invention discloses an imazalil-containing sterilization composition, and belongs to the technical field of pesticides. The sterilization composition comprises a raw pesticide prepared by compounding a bactericide A and a bactericide B with a ratio of 1-80:1-80; the bactericide A is imazalil or efficient imazalil; and the bactericide B is any one of the following bactericides: a copper preparation, methoxy acrylates, triazoles, amides, benzimidazoles, thiocarbamates, substituted benzenes, pyrroles, dicarboximides, phthalimides, imidazoles, morpholines, carbamates, oxazoles, antibiotics, and the like. The sterilization composition comprises the raw pesticide prepared by compounding imazalil or efficient imazalil and a bactericide B, the sterilization spectrum is expanded, the pesticide resistance of pathogens is alleviated, and the sterilization composition has obvious continuous synergic effect.
Owner:王学权
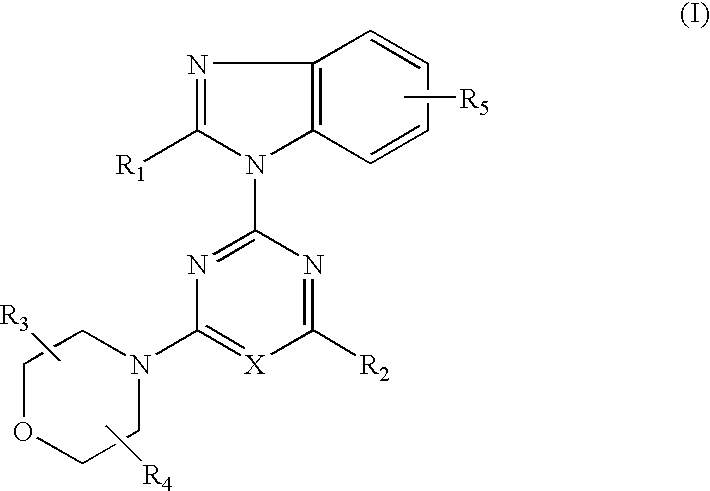
Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds
InactiveUS20090325945A1Promote apoptosisInhibiting cell cycle progressionBiocideOrganic chemistryDiseaseMelanoma
The present invention pertains to certain imidazo[4,5-b]pyridin-2-one and oxazolo[4,5 b]pyridin-2-one compounds and analogs thereof, which, inter alia, inhibit RAF (e.g., B RAF) activity, inhibit cell proliferation, treat cancer, etc., and more particularly to compounds of the formulae: wherein: J is independently —O— or —NRN1−; RN1, if present, is independently —H or a substituent; RN2 is independently —H or a substituent; Y is independently —CH═ or —N═; Q is independently —(CH2)j-M-(CH2)k— wherein: j is independently 0, 1 or 2; k is independently 0, 1, or 2; j+k is 0, 1, or 2; M is independently O—, —S—, —NH—, —NMe-, or —CH2—; each of RP1, RP2, RP5, and RP4 is independently —H or a substituent; and additionally RP1 and RP2 taken together may be CH═CH—CH═CH—; and additionally RP1 and RP5 taken together may be CH═CH—CH═CH—; L is independently: a linker group formed by a chain of 2, 3, or 4 linker moieties; each linker moiety is independently CH2—, —NRN—, —C(═X)—, or —S(═O)2—; either: exactly one linker moiety is —NRN—, or: exactly two linker moieties are —NRN—; either: exactly one linker moiety is —C(═X)—, and no linker moiety is —S(═O)2—, or: exactly one linker moiety is —S(═O)2—, and no linker moiety is —C(═X)—; no two adjacent linker moieties are —NRN—; X is independently ═O or ═S; each RN is independently —H or a substituent; A is independently: C6-14carboaryl, C5-14heteroaryl, C3-12carbocyclic, C3-12heterocyclic; and is independently unsubstituted or substituted; and pharmaceutically acceptable salts, solvates, amides, esters, ethers, N-oxides, chemically protected forms, and prodrugs thereof. The present invention also pertains to pharmaceutical compositions comprising such compounds, and the use of such compounds and compositions, both in vitro and in vivo, to inhibit RAF (e.g., B-RAF) activity, to inhibit receptor tyrosine kinase (RTK) activity, to inhibit cell proliferation, and in the treatment of diseases and conditions that are ameliorated by the inhibition of RAF, RTK, etc., proliferative conditions such as cancer (e.g., colorectal cancer, melanoma), etc.
Owner:CANCER RES TECH LTD +1
Resin composition and adhesive film for multi-layered printed wiring board
ActiveUS20050019554A1High mechanical strengthEasy to manufactureLayered product treatmentAdhesive articlesBenzoxazolePolyetherimide
Resin compositions, which contain: (a) a heat-resistant resin which is soluble in organic solvents wherein the heat-resistant resin is one or more heat-resistant resin(s) selected from the group consisting of a polyimide resin, a polyamide-imide resin, a polyamide resin, a polyether imide resin, a polybenzoxazol resin, a polybenzimidazole resin, copolymers thereof, and mixtures thereof; (b) a thermosetting resin; (c) a filler; and (d) resin having a polybutadiene structure and / or a polysiloxane structure, wherein: the heat-resistant resin (a) and the thermosetting resin (b) are present in a weight ratio of heat-resistant resin (a) to thermosetting resin (b) of from 100:1 to 1:1; the heat-resistant resin (a), thermosetting resin (b), and filler (c) are present in relative amounts such that weight ratio of the total amount of heat-resistant resin (a) and thermosetting resin (b) to filler (c) by weight is from 100:1 to 3:2; the resin having a polybutadiene structure and / or a polysiloxane structure (d) is present in an amount of 0.1 to 15 parts by weight based on 100 parts by weight of the heat-resistant resin (a); and the heat-resistant resin (a), thermosetting resin (b), filler (c), and resin having a polybutadiene structure and / or a polysiloxane structure (d) are present in a total amount of not less than 70% by weight, based on the total weight of the resin composition, are useful for preparing adhesive films, which are, in turn, useful for forming interlayer insulation layers for multi-layered printed wiring boards having an excellent mechanical strength and capable of being roughened by an oxidizing agent.
Owner:AJINOMOTO CO INC
Lithium battery including storage stabilized dioxolane-containing electrolyte
InactiveUS6255021B1Reduced self dischargeEasy to storeOrganic electrolyte cellsLi-accumulatorsOrganic solventOxygen
A nonaqueous electrolyte battery having improved storage stability is disclosed. The battery includes a positive electrode; s negative electrode in which the active material is lithium or a compound capable of absorbing and desorbing lithium; and a nonaqueous electrolyte containing an organic solvent, at least 10 wt % of which is dioxolane, a solute and a storage stabilizing additive which is an oxygen acid ester, isoxazole, oxazole or oxazoline or a derivative thereof. The additive reduces the self-discharge rate of the battery during storage.
Owner:SANYO ELECTRIC CO LTD
One component glass primer including oxazoladine
Improved long open time one component primer compositions for bonding substrates, methods and the articles made therefrom. The primer compositions include an ingredient including an oxazolidine ring or derivative or analog thereof.
Owner:DOW GLOBAL TECH LLC
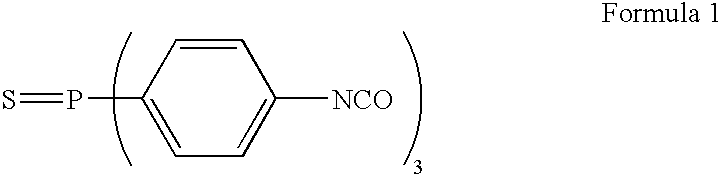
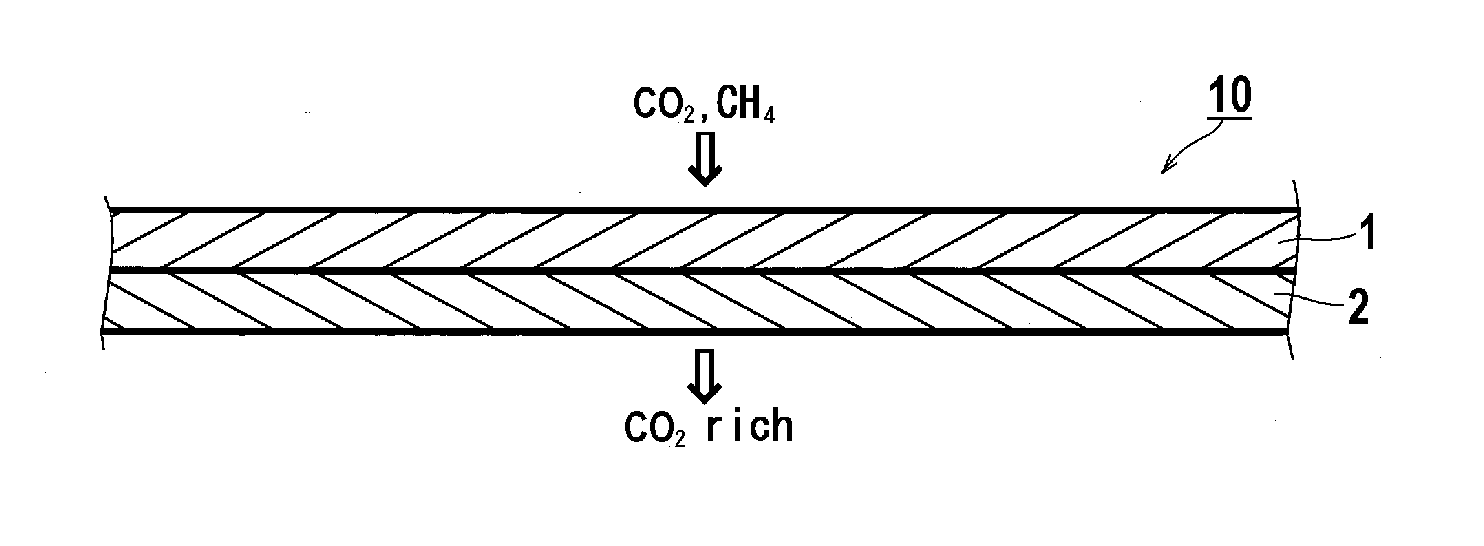
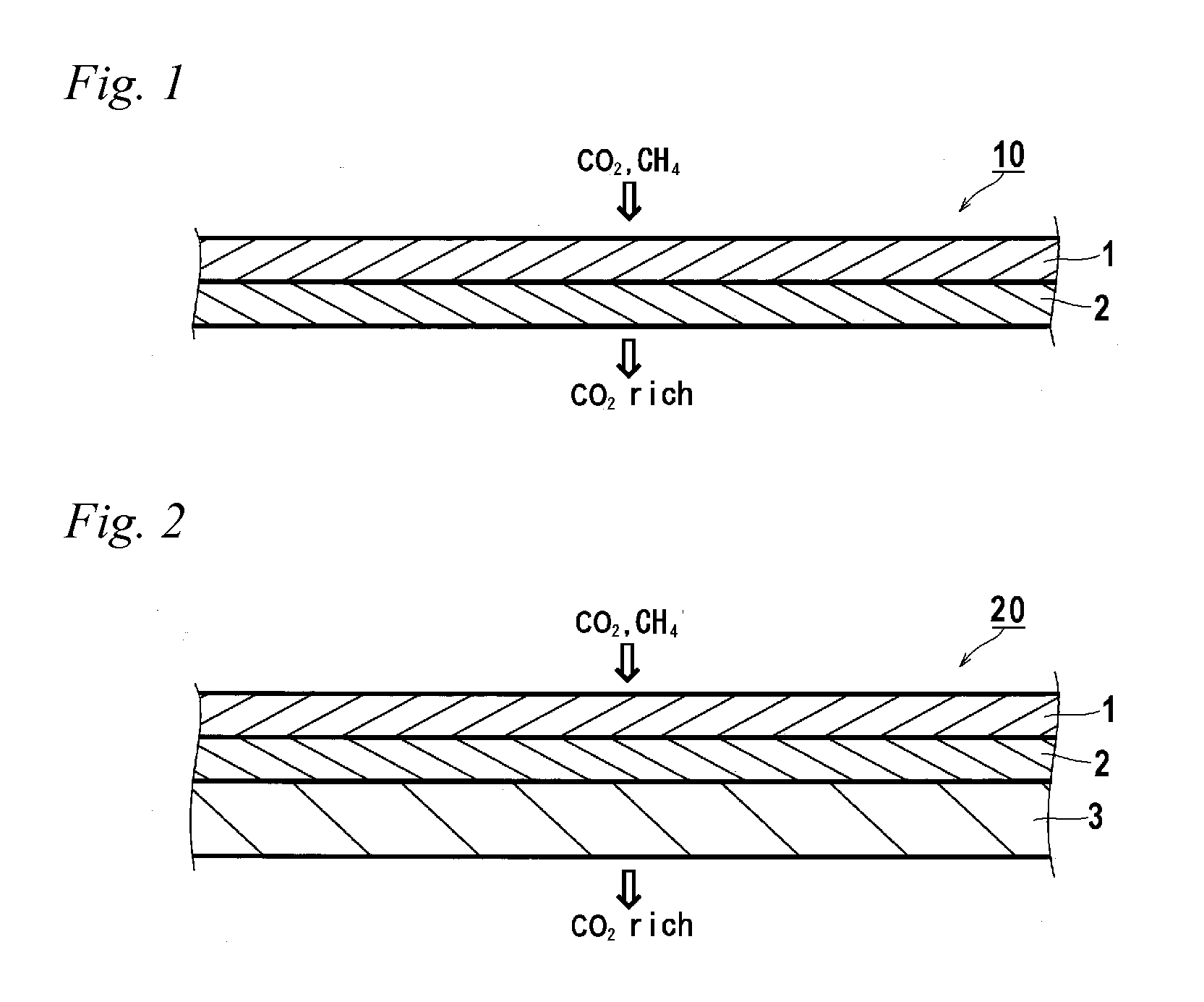
Gas separation membrane, gas separation module, gas separation apparatus, and gas separation method
InactiveUS20150258505A1Satisfactory mechanical strengthImprove permeabilitySemi-permeable membranesMembranesBenzoxazoleSolubility
A gas separation membrane having a gas separating layer containing a polybenzoxazole resin, in which the polybenzoxazole exhibits a solubility of 1% by mass or more to any one solvent selected from tetrahydrofuran, chloroform, methyl ethyl ketone, and N-methylpyrrolidone, at a temperature of 30° C., a gas separation module utilizing the gas separation membrane, a gas separation apparatus, and a gas separation method.
Owner:FUJIFILM CORP
Thiazoles and oxazoles useful as modulators of ATP-Binding Cassette transporters
The present invention relates to modulators of ATP-Binding Cassette (“ABC”) transporters or fragments thereof, including Cystic Fibrosis Transmembrane Regulator (“CFTR”), compositions thereof, and methods therewith. The present invention also relates to methods of treating ABC transporter mediated diseases using such modulators.
Owner:VERTEX PHARMA INC
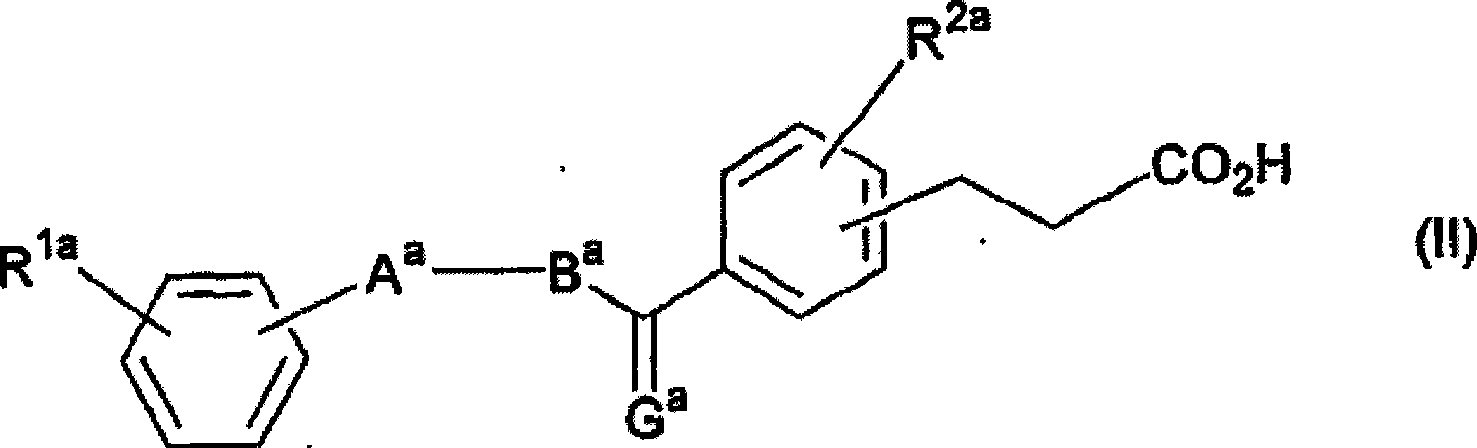
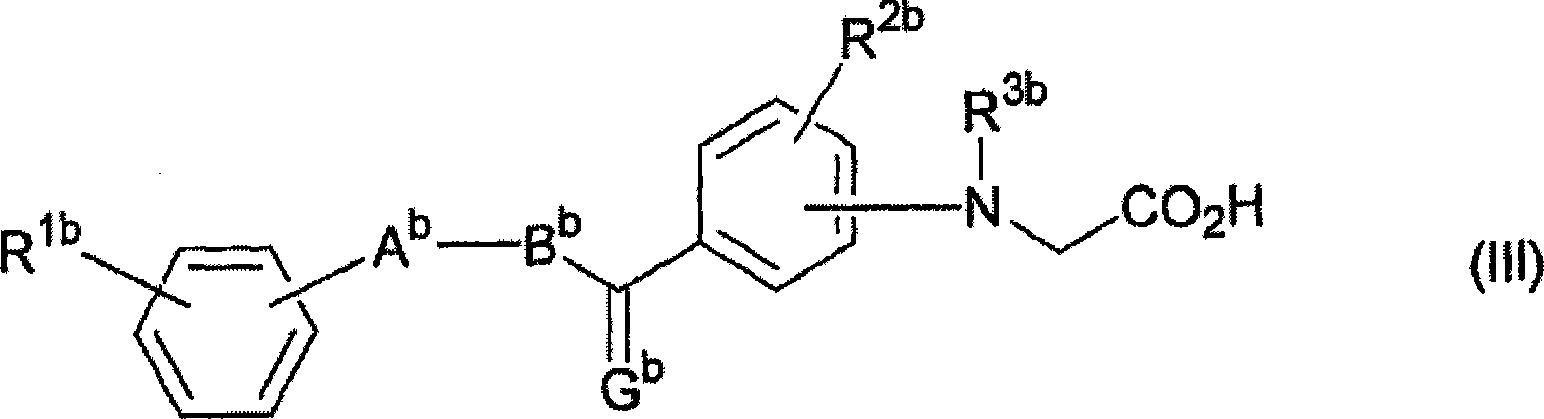
Activating agent for peroxisome proliferator activated receptor delta
A compound represented by the general formula (II) below or a salt thereof is used as an activating agent for PPAR d. (II) (In the formula, G represents O, CH2 or the like; A represents a thiazole, oxazole or thiophene which may have a substituent selected from an alkyl group having 1-8 carbon atoms, an alkoxy group having 1-8 carbon atoms, a halogen atom, an alkyl group having 1-8 carbon atoms and substituted by a halogen atom and the like; B represents an alkylene chain having 1-8 carbon atoms, and when the alkylene chain has two or more carbon atoms, it may have a double bond; and R<1a> and R<2a> respectively represent a hydrogen atom, an alkyl group having 1-8 carbon atoms, an alkoxy group having 1-8 carbon atoms, a halogen atom, an alkyl group having 1-8 carbon atoms and substituted by a halogen atom or the like.
Owner:NIPPON CHEMIPHAR CO LTD
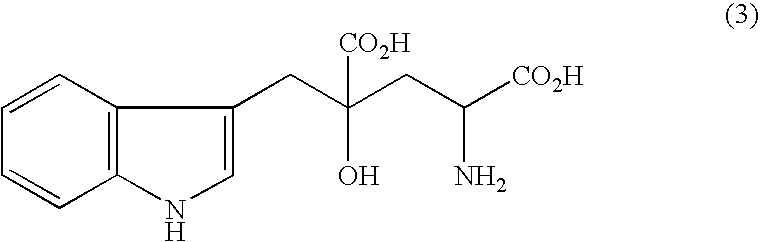
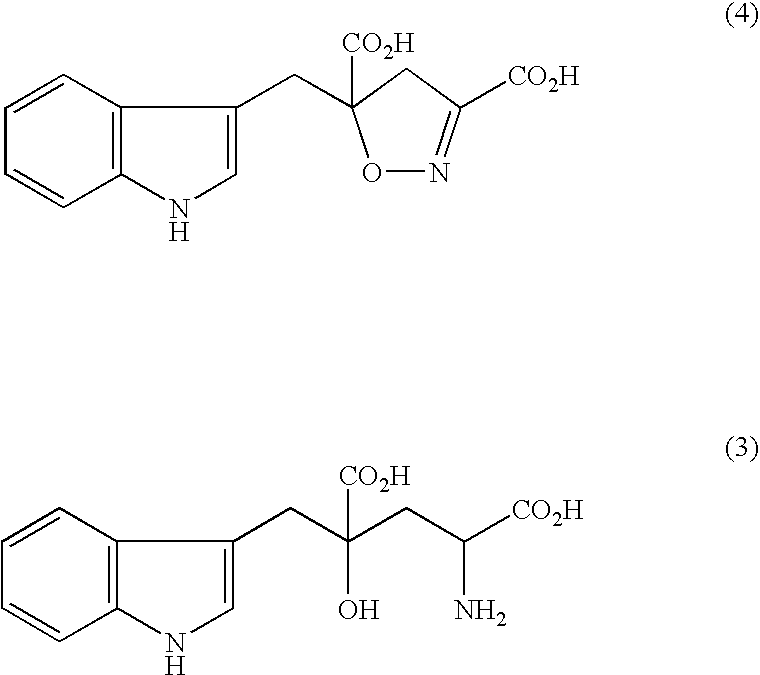
Process for producing gamma-hydroxyamino acid derivatives and monatins
InactiveUS7329427B2Safe and simple procedureEasy to getOrganic chemistry methodsFood preparationAcid derivativeDicarboxylic acid
Dihydroisoxazole derivatives are conveniently converted to γ-hydroxyamino acid derivatives which are important as various synthetic intermediates by a catalytic hydrogenation reaction. High-purity monatins which may be used as sweeteners or ingredients thereof can be obtained by subjecting a 5-indolylmethyl-4,5-dihydroisoxazole-3,5-dicarboxylic acid to catalytic hydrogenation.
Owner:AJINOMOTO CO INC
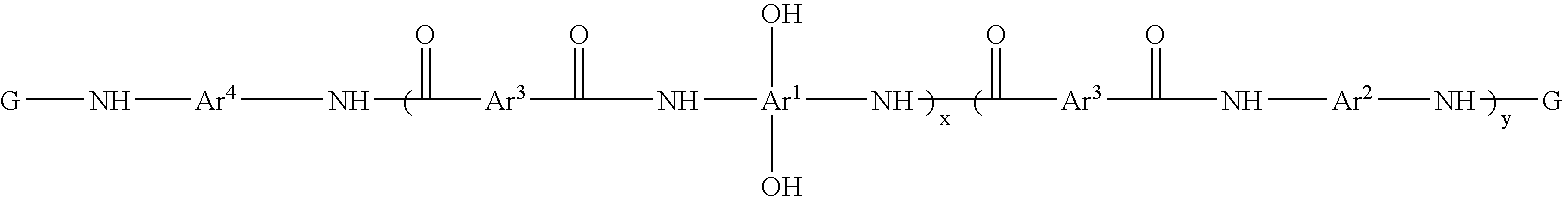
Photosensitive resin compositions
A heat resistant negative working photosensitive composition that comprises(a) one or more polybenzoxazole precursor polymers (I): wherein x is an integer from about 10 to about 1000, y is an integer from 0 to about 900 and (x+y) is about less then 1000; Ar1 is a tetravalent aromatic group, a tetravalent heterocyclic group, or mixtures thereof; Ar2 is a divalent aromatic, a divalent heterocyclic, a divalent alicyclic, or a divalent aliphatic group that may contain silicon; Ar3 is a divalent aromatic group, a divalent aliphatic group, a divalent heterocyclic group, or mixtures thereof; Ar4 is Ar1 (OH)2 or Ar2; G is a monovalent organic group a carbonyl, carbonyloxy or sulfonyl group;(b) one or more photo-active compounds which release acid upon irradiation (PAGs);(c) one or more latent crosslinkers each of which contains at least two ˜N—(CH2OR)n units (n=1 or 2, wherein R is a linear or branched C1–C8 alkyl group);(d) at least one solvent, and(e) at least one dissolution rate modifier,with the proviso that when the latent crosslinker is highly reactive, the dissolution rate modifier does not contain carboxylic acid groups.
Owner:FUJIFILM ELECTRONICS MATERIALS US
Positive photosensitive polybenzoxazole precursor compositions
ActiveUS7803510B2Reduce sidewall angleAvoid stress failureDiazo compound compositionsPhotosensitive material processingBenzoxazolePlasticizer
A heat resistant positive-working photosensitive PBO precursor composition comprising:(a) at least one polybenzoxazole precursor polymer;(a) at least one plasticizer compound;(b) at least one solvent;wherein the amount of the plasticizer present in the composition is an amount effective to reduce the sidewall angle of imaged and cured features in the coated film on the substrate to prevent stress failures in subsequent metallization of the substrate due to steep angles of the imaged features, and with the proviso that if the polybenzoxazole precursor polymer solely consists of polybenzoxazole precursor polymers that do not contain a photoactive moiety on the polymer, then(c) at least one photoactive compound is also present in the composition.
Owner:FUJIFILM ELECTRONICS MATERIALS US
2-imidazolinylaminobenzoxazole compounds useful as alpha-2 adrenoceptor agonists
InactiveUS6110952AHigh activityNo adverse side effectsBiocideSenses disorderAdrenergicAlpha 2 adrenoceptors
Owner:THE PROCTER & GAMBLE COMPANY
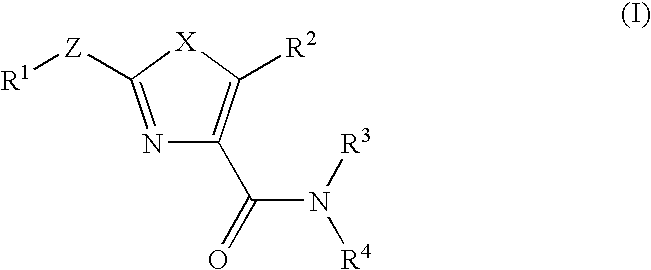
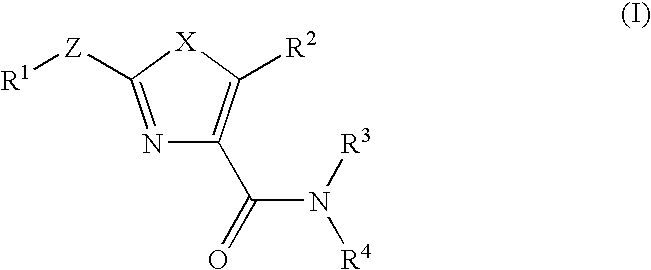
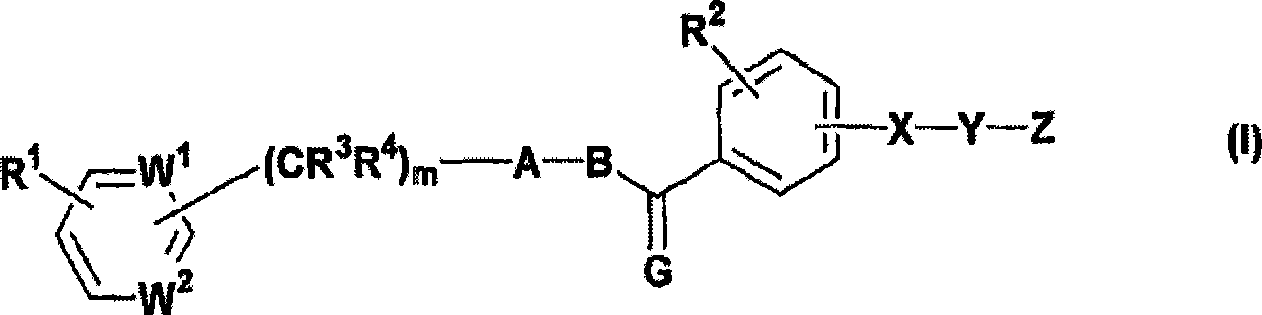
Imidazo[4, 5-B]pyridin-2-one and oxazolo[4, 5-B] pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds
The present invention pertains to certain imidazo[4,5-b]pyridin-2-one and oxazolo[4,5 b]pyridin-2-one compounds and analogs thereof, which, inter alia, inhibit RAF (e.g., B RAF) activity, inhibit cell proliferation, treat cancer, etc. The present invention also pertains to pharmaceutical compositions comprising such compounds, and the use of such compounds and compositions, both in vitro and in vivo, to inhibit RAF (e.g., B-RAF) activity, to inhibit receptor tyrosine kinase (RTK) activity, to inhibit cell proliferation, and in the treatment of diseases and conditions that are ameliorated by the inhibition of RAF, RTK, etc., proliferative conditions such as cancer (e.g., colorectal cancer, melanoma), etc.
Owner:CANCER RES TECH LTD +1
Oxazolidinone combinatorial libraries, compositions and methods of preparation
InactiveUS6562844B2Rapid productionEasy to synthesizeBiocideGroup 5/15 element organic compoundsLibrary preparationCombinatorial chemistry
Oxazolidinones and methods for their synthesis are provided. Also provided are combinatorial libraries comprising oxazolidinones, and methods to prepare the libraries. Further provided are methods of making biologically active oxazolidinones as well as pharmaceutically acceptable compositions comprising the oxazolidinones. The methods of library preparation include the attachment of oxazolidinones to a solid support. The methods of compound preparation in one embodiment involve the reaction of an iminophosphorane with a carbonyl containing polymeric support.
Owner:PHARMACIA & UPJOHN CO


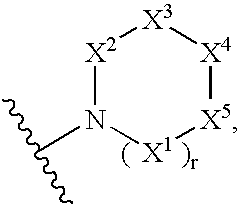
![Oxazolo, thiazolo and selenazolo [4,5-c] quinolin-4-amines and analogs thereof Oxazolo, thiazolo and selenazolo [4,5-c] quinolin-4-amines and analogs thereof](https://images-eureka.patsnap.com/patent_img/74ac572b-d35d-49e9-8ac6-b860065b1a3c/US06323200-20011127-C00001.png)
![Oxazolo, thiazolo and selenazolo [4,5-c] quinolin-4-amines and analogs thereof Oxazolo, thiazolo and selenazolo [4,5-c] quinolin-4-amines and analogs thereof](https://images-eureka.patsnap.com/patent_img/74ac572b-d35d-49e9-8ac6-b860065b1a3c/US06323200-20011127-C00002.png)
![Oxazolo, thiazolo and selenazolo [4,5-c] quinolin-4-amines and analogs thereof Oxazolo, thiazolo and selenazolo [4,5-c] quinolin-4-amines and analogs thereof](https://images-eureka.patsnap.com/patent_img/74ac572b-d35d-49e9-8ac6-b860065b1a3c/US06323200-20011127-C00003.png)
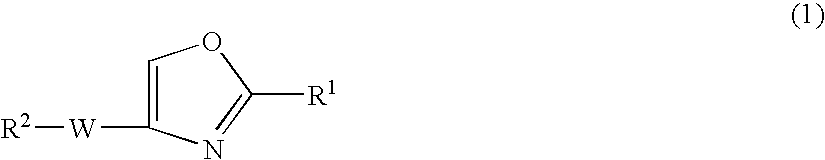
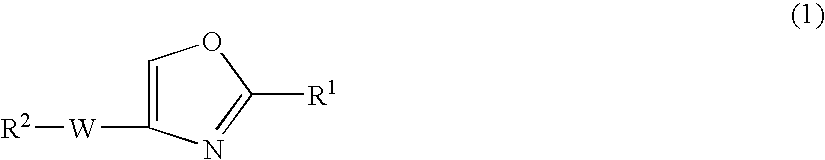
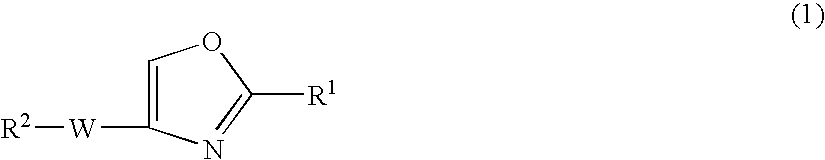



![Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds](https://images-eureka.patsnap.com/patent_img/160c93b4-7535-4186-a3b0-42b5846b0524/US20090325945A1-C00001.png)
![Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds](https://images-eureka.patsnap.com/patent_img/160c93b4-7535-4186-a3b0-42b5846b0524/US20090325945A1-C00002.png)
![Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds](https://images-eureka.patsnap.com/patent_img/160c93b4-7535-4186-a3b0-42b5846b0524/US20090325945A1-C00003.png)



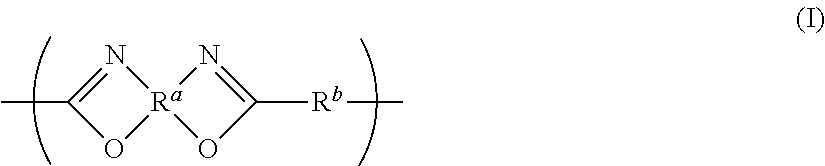
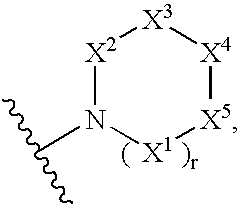
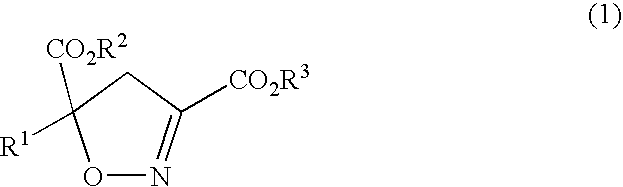
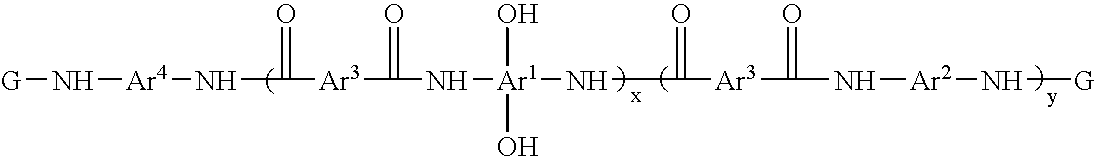
![Imidazo[4, 5-B]pyridin-2-one and oxazolo[4, 5-B] pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds Imidazo[4, 5-B]pyridin-2-one and oxazolo[4, 5-B] pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds](https://images-eureka.patsnap.com/patent_img/56ad8e42-9fd9-4d02-ba70-fb173ab812ce/US07951819-20110531-C00001.png)
![Imidazo[4, 5-B]pyridin-2-one and oxazolo[4, 5-B] pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds Imidazo[4, 5-B]pyridin-2-one and oxazolo[4, 5-B] pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds](https://images-eureka.patsnap.com/patent_img/56ad8e42-9fd9-4d02-ba70-fb173ab812ce/US07951819-20110531-C00002.png)
![Imidazo[4, 5-B]pyridin-2-one and oxazolo[4, 5-B] pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds Imidazo[4, 5-B]pyridin-2-one and oxazolo[4, 5-B] pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds](https://images-eureka.patsnap.com/patent_img/56ad8e42-9fd9-4d02-ba70-fb173ab812ce/US07951819-20110531-C00003.png)