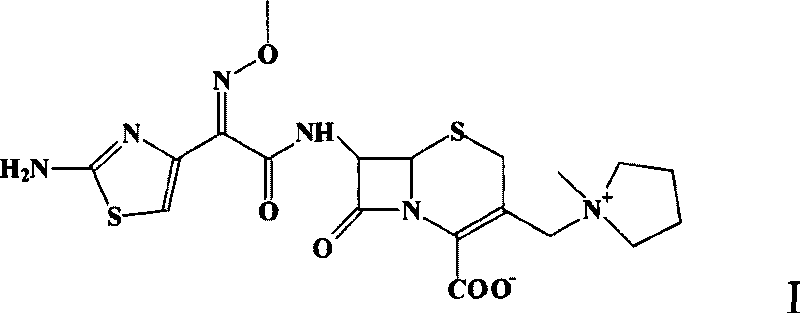
Method for preparing cefepime dihydrochloride monohydrate crystal
A technology of cefepime dihydrochloride and monohydrate, which is applied in the field of preparation of cefepime hydrochloride to control the formation of specific hydrate crystals, and can solve problems such as reducing water content, failing to meet Chinese quality standards, and unable to guarantee crystallization.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0030] Embodiment 1, the refining of cefepime dihydrochloride monohydrate
[0031]Add 150 g of cefepime dihydrochloride monohydrate (HPLC purity 98%, prepared according to the method in US4910301) to 750 mL of methanol under stirring at room temperature, add an appropriate amount of activated carbon after 10 minutes, and stir for 20 minutes. Filter and wash the carbon residue with an appropriate amount of acetone-methanol 1:1 (v / v) mixed solution. Combine the filtrates and cool to 10-20°C. Add 6500 mL of acetone to the filtrate under stirring, and complete the addition within about 1 hour. Stirring was continued for 1 hour. Suction. The filter cake was washed with acetone and dried under vacuum at 40° C. to obtain about 140 g of white crystals of cefepime dihydrochloride monohydrate. HPLC purity 99.5%, water content 3.3% (K-F method), residual methanol 0.20%, residual acetone 0.35%.
Embodiment 2
[0032] Embodiment 2, prepare cefepime dihydrochloride monohydrate by cefepime monohydrochloride
[0033] 136 g of cefepime monohydrochloride (HPLC purity 97%, prepared according to the method in US4910301) was added to 750 mL of methanol under stirring at room temperature, and 50 mL of 20% HCl was added after 10 minutes. Stir for 10 minutes, if there is any insoluble matter, filter it off. Then add 6500 mL of acetone to the solution, and add it within about 1 hour. Stirring was continued for 1 hour. Suction. The filter cake was washed with acetone and dried under vacuum at 40° C. to obtain about 130 g of white crystals of cefepime dihydrochloride monohydrate with an HPLC purity of 99%.
Embodiment 3
[0034] Embodiment 3, prepare cefepime dihydrochloride monohydrate by cefepime dihydrochloride dihydrate
[0035] Add 150g of cefepime dihydrochloride dihydrate (HPLC purity 98%, prepared according to the method in US5391729) to the mixed solution of 150mL water and 900mL methanol under stirring at room temperature, add an appropriate amount of activated carbon after 10 minutes, and stir for 30 minutes . Filter and wash the carbon residue with an appropriate amount of acetone-methanol 1:1 (v / v) mixed solution. Combine the filtrates and cool to 10-20°C. Add 8500 mL of acetone to the filtrate under stirring, and complete the addition within about 1 hour. Stirring was continued for 1 hour. Suction. The filter cake was washed with acetone and dried under vacuum at 40° C. to obtain about 130 g of white crystals of cefepime dihydrochloride monohydrate with an HPLC purity of 99.5%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More