Novel preparation method of Erlotinib hydrochloride with crystal form A
A technology of hydrochloride and crystal form, which is applied in the field of preparation of polymorphic drugs, can solve the problems of easy conversion of crystal form into other crystal forms and restrictions, and achieve the effects of good reproducibility, low cost, and increased reaction temperature
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
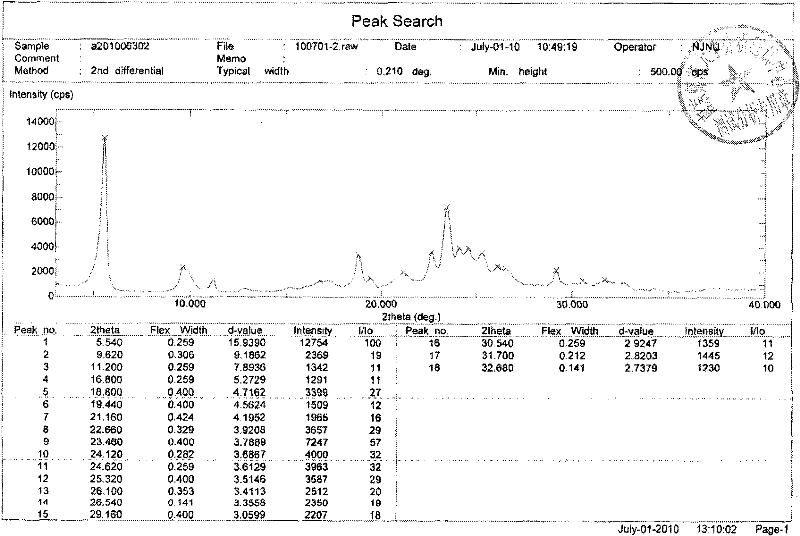
[0028] Embodiment 1: Preparation of Erlotinib Hydrochloride A Crystal Form
[0029] Weigh Erlotinib free base (393mg, 1mmol), then add it to the reaction flask, add 12mL ethyl acetate, control the temperature at 20°C, stir magnetically, start to add 1.1mL HCl ethyl acetate solution (1M) dropwise, drop after 5min After the addition was complete, the mixture was stirred for 20 min, filtered, washed with ethyl acetate (3×2 mL), and dried in vacuo at 30° C. to obtain 428 mg of crystalline form A of Erlotinib hydrochloride with a yield of 100%.
Embodiment 2
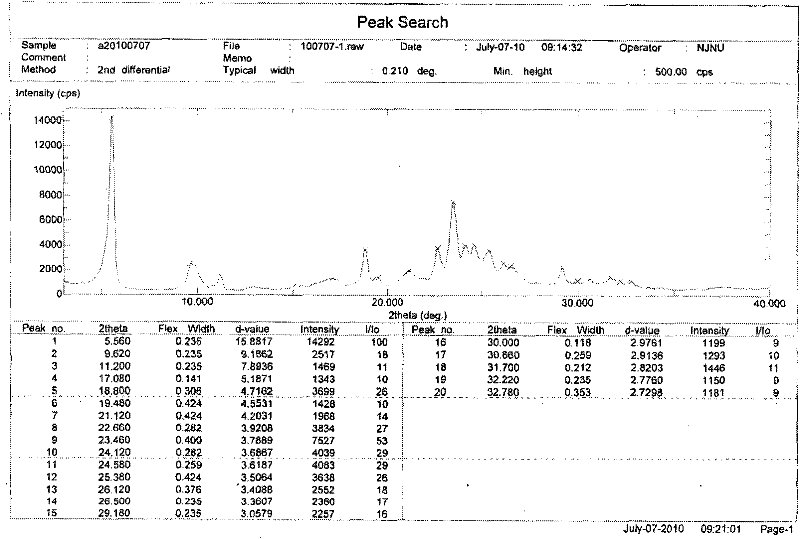
[0030] Embodiment 2: Preparation of Erlotinib Hydrochloride A Crystal Form
[0031] Weigh Erlotinib free base (393mg, 1mmol), then add it to the reaction flask, add 12mL ethyl acetate, control the temperature at 40°C, stir magnetically, start to add 1.1mL HCl ethyl acetate solution (1M) dropwise, drop after 5min After the addition was complete, the mixture was stirred for 20 min, filtered, washed with ethyl acetate (3×2 mL), and dried in vacuo at 30° C. to obtain 427 mg of crystalline form A of Erlotinib hydrochloride with a yield of 100%.
Embodiment 3
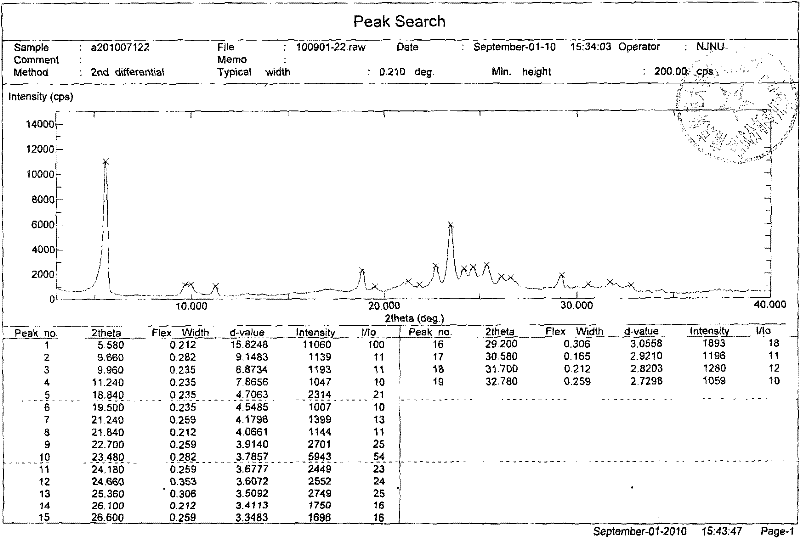
[0032] Embodiment 3: Preparation of Erlotinib Hydrochloride A Crystal Form
[0033] Weigh Erlotinib free base (393mg, 1mmol), then add it to the reaction flask, add 12mL acetone, control the temperature at 30°C, stir magnetically, start to add 1.1mL HCl isopropyl acetate solution (1M) dropwise, and add dropwise after 5min After completion, continue to stir for 20 min, filter, wash with acetone (3×2 mL), and dry in vacuo at 30° C. to obtain 426 mg of Erlotinib hydrochloride Form A with a yield of 99%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com