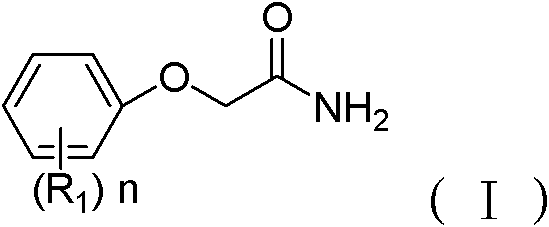
Amide compound and application thereof in preparing monoamine oxidase inhibitor
An amide compound, monoamine oxidase technology, applied in organic chemistry, drug combination, medical preparations containing active ingredients, etc., can solve the problems of psychotic episodes, side effects, liver toxicity, etc., and achieves improved selectivity, stable properties, and operation. Ease of the process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0034] Reaction formula:
[0035]
[0036] Method:
[0037] (1) Dissolving the phenolic compound (II) in DMF solvent, then adding NaOH and 2-chloroacetamide, and heating at 85°C for about 6 hours. The amount of phenolic compound: 2-chloroacetamide: NaOH is 5mmol: 5mmol: 7.2mmol, and the volume of DMF is 10ml.
[0038] (2) Dilute the amide compound constructed in step (1) with DMSO solvent to a concentration of 100 μM, pipette 4 μL of the diluent and add it to the reaction well of a 96-well plate, then add 400 μL of pH8.4 boric acid buffer and 4 μL of BSA, and add MAO -A (20mg / mL) or MAO-B (20mg / mL) 4μL, the mixture was reacted at 38°C for 3h, and then the fluorescent probe 4-methyl-7-(3-aminopropyl)-coumarin 2μL ( 10mmol / mL), then react at 38°C for 2h, and use a fully functional spectrofluorometer (λ ex / λ em =365 / 460nm) for detection, judge the level of inhibitory activity of amide compounds on monoamine oxidase according to the level of fluorescence value: the stronge...
Embodiment 2
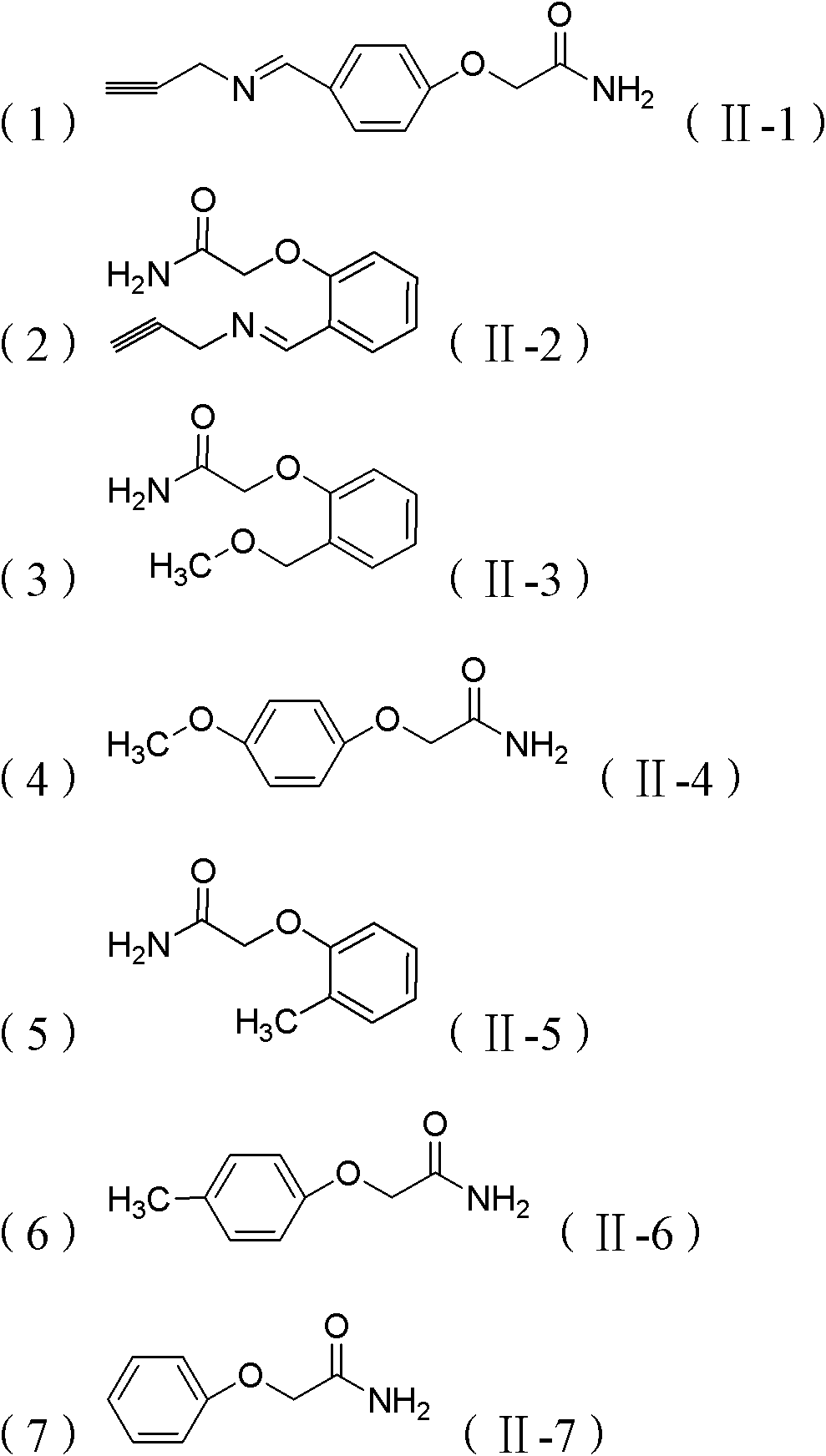
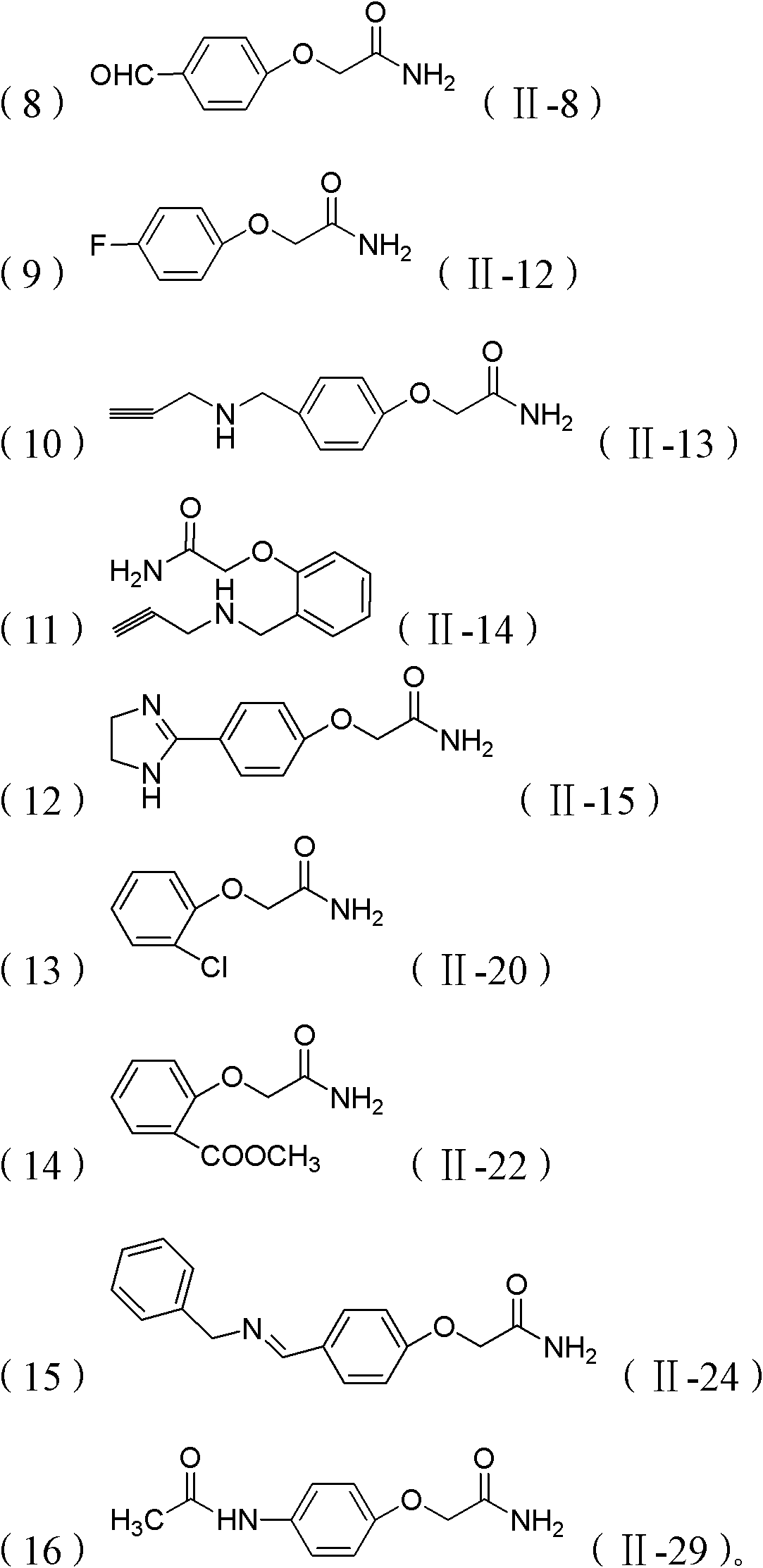
[0043]Example 2: Preparation of 2-(4-((propene-2-imino)methyl)phenoxy)acetamide (II-1)
[0044]
[0045] The synthetic route is as follows:
[0046]
[0047] 4-Hydroxybenzaldehyde (6mmol), NaOH (7.2mmol) and DMF (10ml) were added to a 50ml round-bottomed flask. After the dissolution was complete, 2-chloroacetamide (5mmol) was added and stirred at 85°C for 6h, the reaction was complete After 3*20ml dichloromethane extraction, NaHCO 3 Washed with saturated solution, dried over anhydrous magnesium sulfate, filtered, sucked dry, purified by column chromatography to obtain a white solid powder with a yield of 91%. Dissolve the obtained white powder in dichloromethane, add one equivalent of propargyl amine and react for one hour, extract with 3*20ml dichloromethane, NaHCO 3 Washed with saturated solution, dried over anhydrous magnesium sulfate, filtered, sucked to dryness, and purified by column chromatography to obtain a white solid product with a yield of 85%. Compound Ch...
Embodiment 3
[0048] Example 3: Preparation of 2-(4-((propene-2-imino)methyl)phenoxy)acetamide (II-2)
[0049]
[0050] The synthetic route is as follows:
[0051]
[0052] 2-Hydroxybenzaldehyde (6mmol), NaOH (7.2mmol) and DMF (10ml) were added to a 50ml round-bottomed flask. After the dissolution was complete, 2-chloroacetamide (5mmol) was added and stirred at 85°C for 6h, the reaction was complete After 3*20ml dichloromethane extraction, NaHCO 3 Washed with saturated solution, dried over anhydrous magnesium sulfate, filtered, sucked to dryness, and purified by column chromatography to obtain a white solid powder with a yield of 88%. Dissolve the obtained white powder in dichloromethane, add one equivalent of propargyl amine and react for one hour, extract with 3*20ml dichloromethane, NaHCO 3 Washed with saturated solution, dried over anhydrous magnesium sulfate, filtered, sucked to dryness, and purified by column chromatography to obtain a white solid product with a yield of 74%. ...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap