Preparation method of high-optical-purity pitavastatin calcium key intermediate
A technology of pitavastatin calcium and intermediates, which is applied in the field of blood lipid-lowering drugs, can solve the problems of low yield, insufficient optical purity of pitavastatin calcium intermediates, and difficult separation and purification, and achieve low cost and high optical purity Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0026] The embodiments of the present invention are described in detail below. This embodiment is implemented on the premise of the technical solution of the present invention, and detailed implementation methods and specific operating procedures are provided, but the protection scope of the present invention is not limited to the following implementation example.
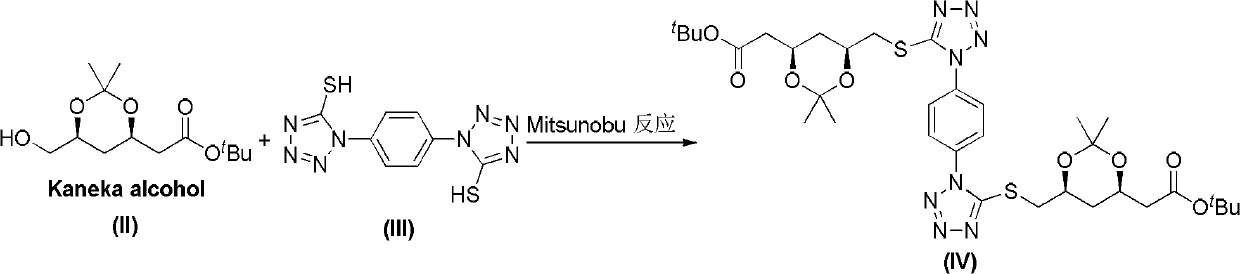
[0027] The preparation of compound IV, reaction formula:
[0028]
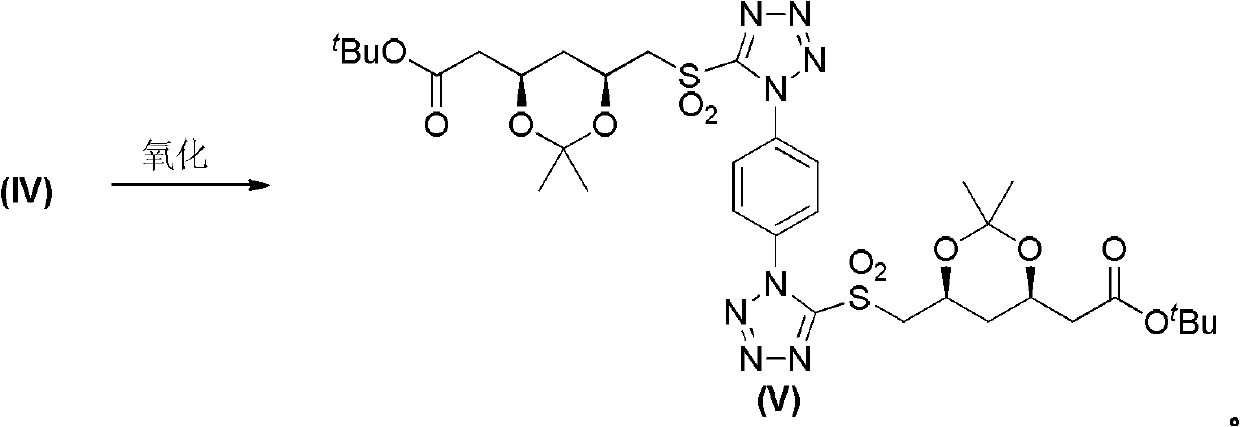
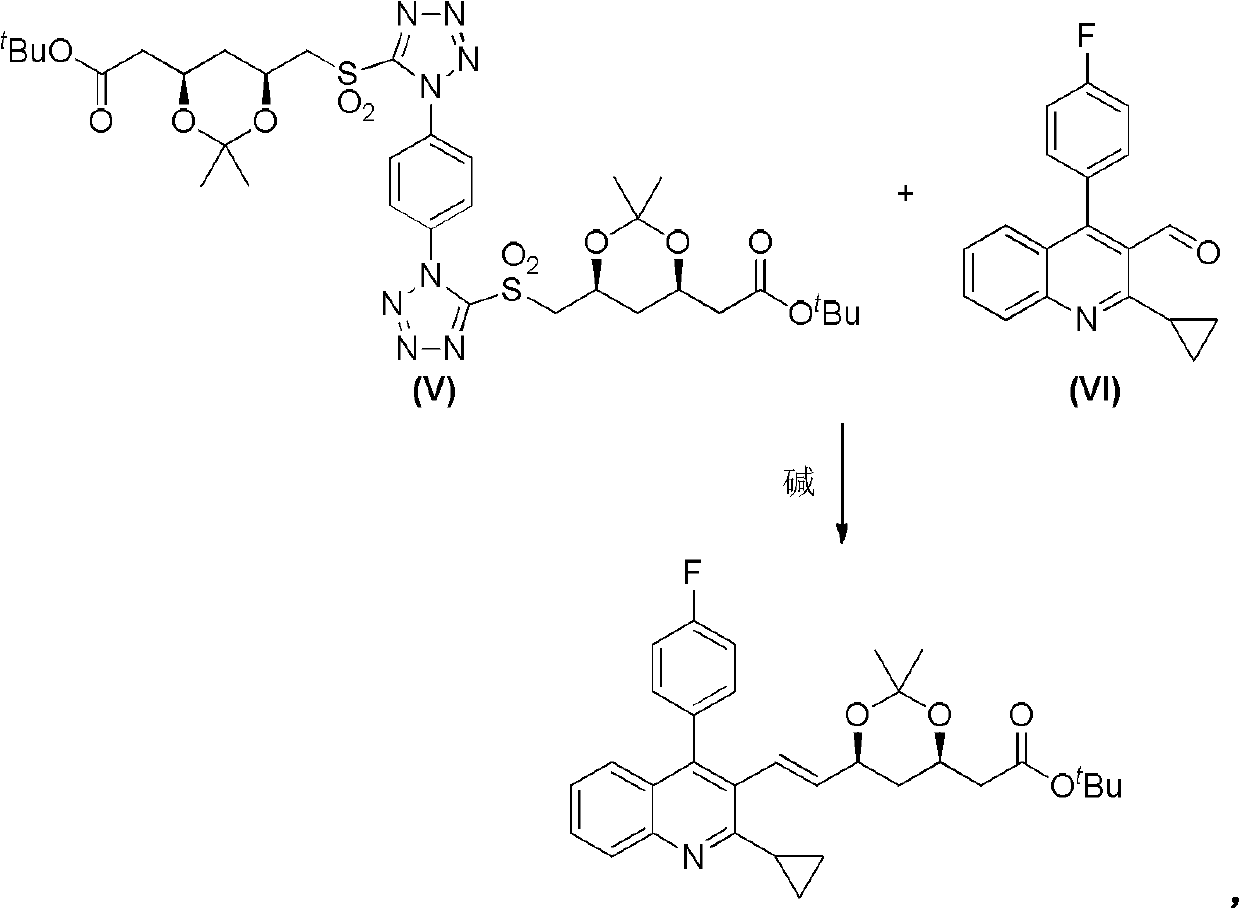
[0029] Steps
[0030] In a 25mL Schlenck reaction tube, under a nitrogen atmosphere, add compound II (1.87g, 7.2mmol), compound III (1.00g, 3.6mmol), triphenylphosphine (2.26g, 8.6mmol) and tetrahydrofuran (10mL), and cool to At 0-5°C, diisopropyl azodicarboxylate (1.74g, 8.6mmol) was added dropwise, and after the addition was completed, the mixture was naturally raised to room temperature for 12h. The tetrahydrofuran was distilled off, 20 mL of ethyl acetate and 15 mL of water were added, stirred, left to stand for liquid separation, and the ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More