Preparation method of (2R, 3R, 11bR)-dihydrotetrabenazine and relevant compounds
A technology of dihydrotetrabenazine and tetrabenazine, applied in organic chemistry methods, chemical instruments and methods, organic chemistry, etc., can solve the cumbersome column chromatography purification process, unsuitable for industrial production, difficult industrial production, etc. problem, achieve the effect of improving stereoselectivity and chemical yield, and avoiding the purification process of column chromatography
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
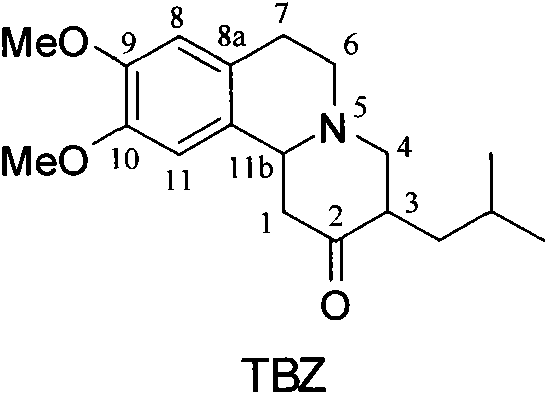
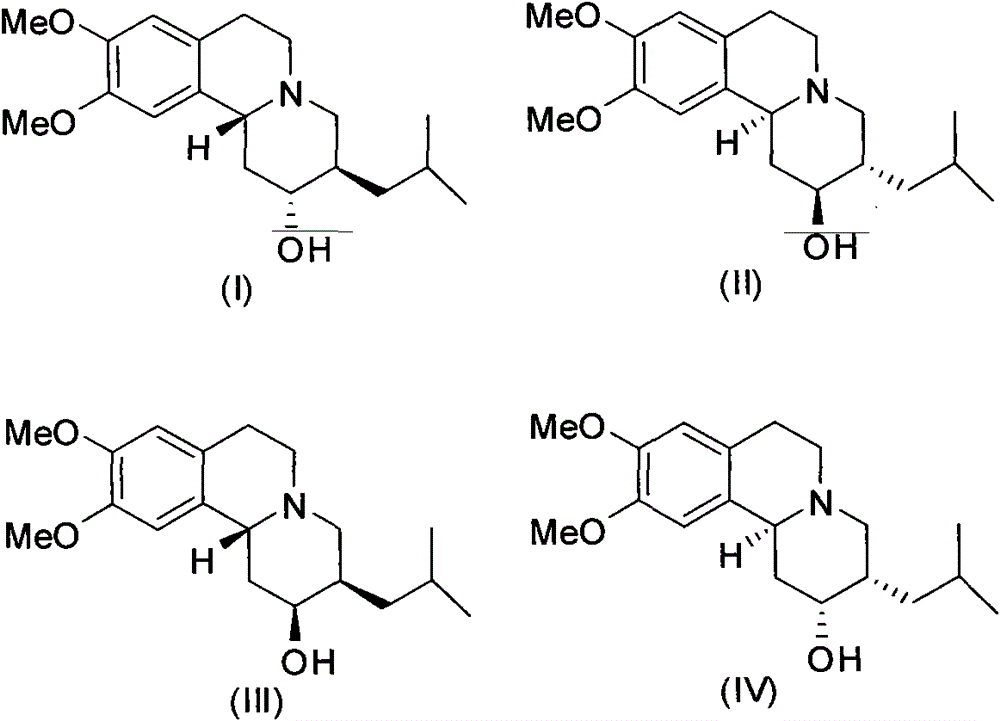
[0022] (2R, 3R, 11bR)-dihydrotetrabenazine ((2R, 3R, 11bR)-DHTBZ)
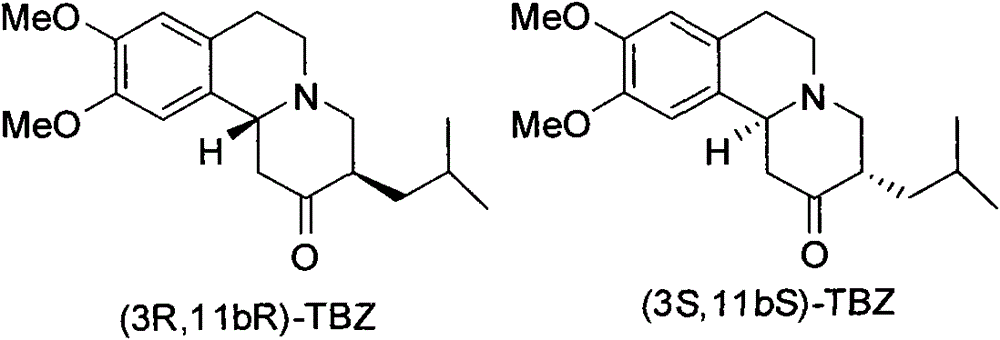
[0023] (3R, 11bR)-TBZ (1.0g, 3.2mmol) was dissolved in 11ml of tetrahydrofuran, and after the solution was cooled to -20°C, 2M borane-dimethylsulfide-tetrahydrofuran (3.2ml, 6.4mmol) was added dropwise into the reaction solution. After the reaction solution was stirred and reacted at -20°C for 2 h, 11 ml of ammonia water was added, and then the mixed solution was heated to 35°C and stirred overnight. Saturated brine was added, and extracted with ether. The ether extract was washed with saturated NaCl solution, anhydrous Na 2 SO 4 It was dried, filtered, and the solvent was distilled off under reduced pressure to obtain 1.07 g of a white solid. The crude product was recrystallized twice through acetone-water to obtain the pure product (2R, 3R, 11bR)-dihydrotetrabenazine (0.64g white solid, yield 64%), [α] D 21 =+58.93° (c 0.6, MeOH). 1 H NMR (300MHz, CDCl 3 ): δ6.68(s, 1H), 6.58(s, 1H), 3.84(s, 6H), 3.3...
Embodiment 2
[0025] (2S, 3S, 11bS)-Dihydrotetrabenazine ((2S, 3S, 11bS)-DHTBZ)
[0026] According to the experimental procedure described in Example 1, (3S, 11bS)-TBZ was reduced with borane-dimethyl sulfide to obtain (2S, 3S, 11bS)-dihydrotetrabenazine (white solid, yield 63%) , [α] D 22 = -56.1° (c 0.23, MeOH). 1H NMR (300MHz, CDCl 3 ): δ6.67(s, 1H), 6.58(s, 1H), 3.87(s, 6H), 3.43-3.34(m, 1H), 3.14-2.96(m, 4H), 2.65-2.54(m, 2H ), 2.53(ddd, 1H, J=11.1, 11.1, 3.6Hz), 2.01-1.94(m, 1H), 1.77-1.64(m, 2H), 1.62-1.36(m, 2H), 1.10-0.99(m , 1H), 0.95-0.70 (m, 6H); 13C NMR (75MHz, CDCl 3 )δ147.50, 147.23, 129.36, 126.42, 111.49, 107.96, 74.63, 60.88, 60.08, 55.94, 55.84, 51.89, 41.65, 40.58, 39.70, 29.18, 25.35, 24.12, 21.76 / z; M+H]+, HRMS-EIS (m / z): 320.2242 [M+H]+ (Calcd for C19H30NO3 320.2226).
Embodiment 3
[0028] (±)-α-Dihydrotetrabenazine ((±)-α-DHTBZ)
[0029] According to the experimental procedure described in Example 1, use borane-dimethyl sulfide to reduce tetrabenazine (TBZ) to obtain (±)-α-dihydrotetrabenazine (white solid), 1 H NMR (300MHz, CDCl 3 )δ6.68(s, 1H), 6.58(s, 1H), 3.84(s, 6H), 3.39(m, 1H), 3.16-2.97(m, 4H), 2.67-2.41(m, 3H), 1.99 (t, J=11.3Hz, 1H), 1.75-1.44(m, 5H), 1.10-1.01(m, 1H), 0.93(t, J=6.5Hz, 6H); ESI-MS m / z 320.3[M +H] + , 342.3[M+Na] + .
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More