Method for improving direct yellow 11 dye-uptake
A direct, p-nitrotoluene technology, applied in the direction of organic dyes, stilbene-azo dyes, etc., can solve the problem of low dyeing rate
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0016] A kind of preparation method of environment-friendly direct yellow 11 with high dye uptake rate, its preparation comprises the following process:
[0017] 4) Weigh diethylene glycol ethyl ether in a three-necked flask, weigh p-nitrotoluene o-sulfonic acid and add it, after stirring evenly, weigh the alcohol amine, slowly drop into the mixed solution, and stir until p-nitrotoluene o-sulfonic acid Sulfonic acid dissolves completely.
[0018] 5) Raise the temperature of the mixture in step 1). After reaching the temperature, slowly add NaOH solution dropwise to the reaction solution. After keeping it warm for a period of time, add ethylene glycol monomethyl ether solution and stir evenly.
[0019] 6) Lower the temperature of the above 2) solution, after room temperature, use H 2 SO 4 The pH value of the solution was adjusted, filtered with suction, and dried to obtain a solid of Direct Yellow 11.
[0020] 1) Diethylene glycol ether, the molecular structure is CH 3 CH ...
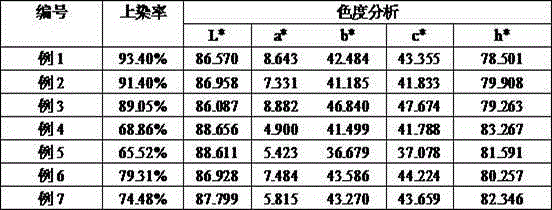
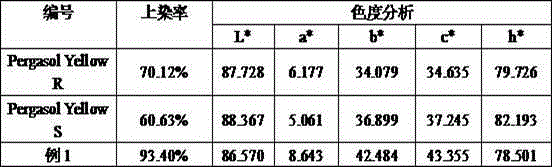
Embodiment 1
[0022] Add 10ml of diethylene glycol ethyl ether to the three-neck flask, weigh 10g of p-nitrotoluene o-sulfonic acid with a content of 80-90%, add it to the water, stir evenly, raise the temperature to 60°C, add about 54ml of 10% NaOH solution , after stirring for 30min, add 10g of triethanolamine, after heat preservation for 8h, add Second Diol monomethyl ether 4ml, stir evenly, use H 2 SO 4 The solution was adjusted to pH 8, filtered with suction and dried.
Embodiment 2
[0024] Add 10ml of diethylene glycol ethyl ether to the three-neck flask, weigh 10g of p-nitrotoluene o-sulfonic acid with a content of 80-90%, add it to the water, stir evenly, raise the temperature to 60°C, add about 54ml of 10% NaOH solution , after stirring for 30min, add 3.4g of diethanolamine, heat preservation reaction for about 8h and finish heating, use H 2 SO 4 The solution was adjusted to pH 8, filtered with suction and dried.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More