Preparation method of nitrogen-doped porous carbon material
A nitrogen-doped porous carbon and nitrogen-doped carbon technology, which is applied in the preparation/purification of carbon, can solve problems such as staying in the academic research stage and restricting practical applications, and achieves avoiding complicated steps, convenient operation, and concentrated pore size distribution. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
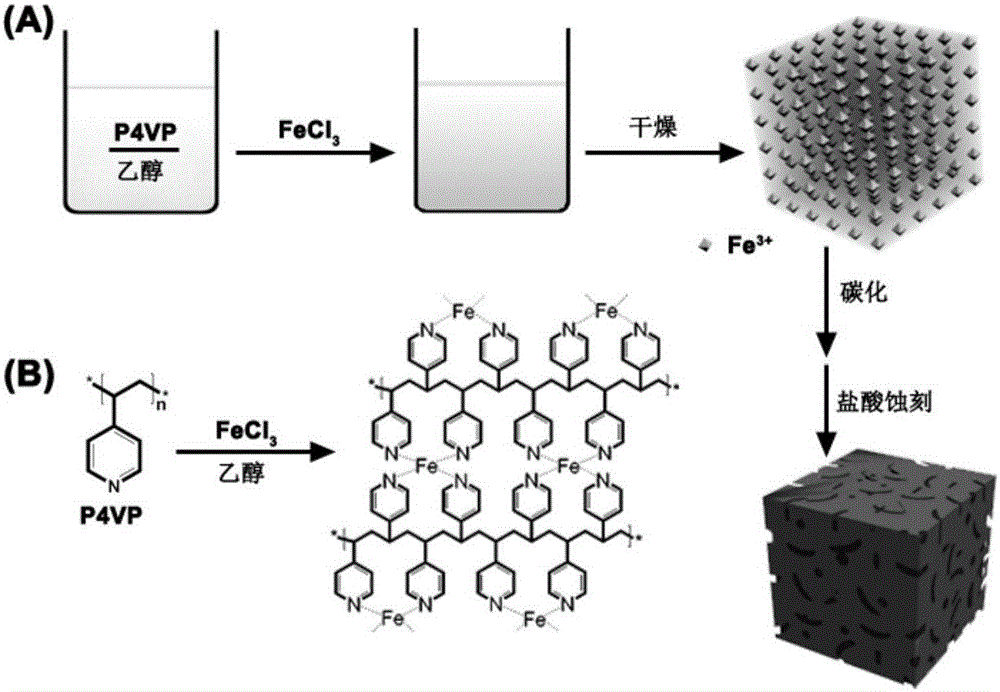
[0038] (1) Preparation of P4VP / dehydrated ethanol solution: at room temperature, take 1.05g of P4VP and add it to 250ml of dehydrated ethanol, and magnetically stir to dissolve P4VP to obtain a P4VP / dehydrated ethanol solution with a concentration of 0.04mol / L, which is ready for use.
[0039] (2) Preparation of coordination metal solution (with Fe 3+ For example): take 2.7g FeCl 3 Dissolve in 250ml absolute ethanol, magnetically stir to make FeCl 3 Dissolve FeCl with a concentration of 0.04mol / L 3 / absolute ethanol solution for use.
[0040] (3) Preparation of carbonized precursor: under the condition of magnetic stirring at room temperature, take 25ml FeCl 3 / dehydrated ethanol solution is added in 100mlP4VP / dehydrated ethanol solution, molar ratio (P4VP: Fe 3+ =4:1; After 24 hours of reaction, a stable complex precipitate was formed, and then subjected to vacuum distillation at 60°C and vacuum drying at 120°C for 12 hours to obtain the carbonized precursor.
[0041] (...
Embodiment 2
[0045] (1) Preparation of P4VP / dehydrated ethanol solution: at room temperature, take 1.05g of P4VP and add it to 250ml of dehydrated ethanol, and magnetically stir to dissolve P4VP to obtain a P4VP / dehydrated ethanol solution with a concentration of 0.04mol / L, which is ready for use.
[0046] (2) Preparation of coordination metal solution (with Fe 3+ For example): take 2.7g FeCl 3 Dissolve in 250ml absolute ethanol, magnetically stir to make FeCl 3 Dissolve FeCl with a concentration of 0.04mol / L 3 / absolute ethanol solution for use.
[0047] (3) Preparation of carbonized precursor: under the condition of magnetic stirring at room temperature, take 100ml FeCl 3 / dehydrated ethanol solution is added in 100mlP4VP / dehydrated ethanol solution, molar ratio (P4VP: Fe 3+ =1:1; After 24 hours of reaction, a stable complex precipitate was formed, and then subjected to vacuum distillation at 60°C and vacuum drying at 120°C for 12 hours to obtain the carbonized precursor.
[0048] ...
Embodiment 3
[0052] (1) Preparation of P4VP / dehydrated ethanol solution: at room temperature, take 1.05g of P4VP and add it to 250ml of dehydrated ethanol, and magnetically stir to dissolve P4VP to obtain a P4VP / dehydrated ethanol solution with a concentration of 0.04mol / L, which is ready for use.
[0053] (2) Preparation of coordination metal solution (with Fe 3+ For example): take 2.7g FeCl 3 Dissolve in 250ml absolute ethanol, magnetically stir to make FeCl 3 Dissolve FeCl with a concentration of 0.04mol / L 3 / absolute ethanol solution for use.
[0054] (3) Preparation of carbonized precursor: under the condition of magnetic stirring at room temperature, take 100ml FeCl 3 / dehydrated ethanol solution is added in 25mlP4VP / dehydrated ethanol solution, molar ratio (P4VP: Fe 3+ =1:4; After 24 hours of reaction, a stable complex precipitate was formed, and then subjected to vacuum distillation at 60°C and vacuum drying at 120°C for 12 hours to obtain the carbonized precursor.
[0055] (...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Specific surface area | aaaaa | aaaaa |
| Specific surface area | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More