Thermoplastic resin composition and molding product thereof
A technology of resin composition and plasticity, which is applied in the field of thermoplastic resin composition and molded products, and can solve the problems of poor compatibility and reduced effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
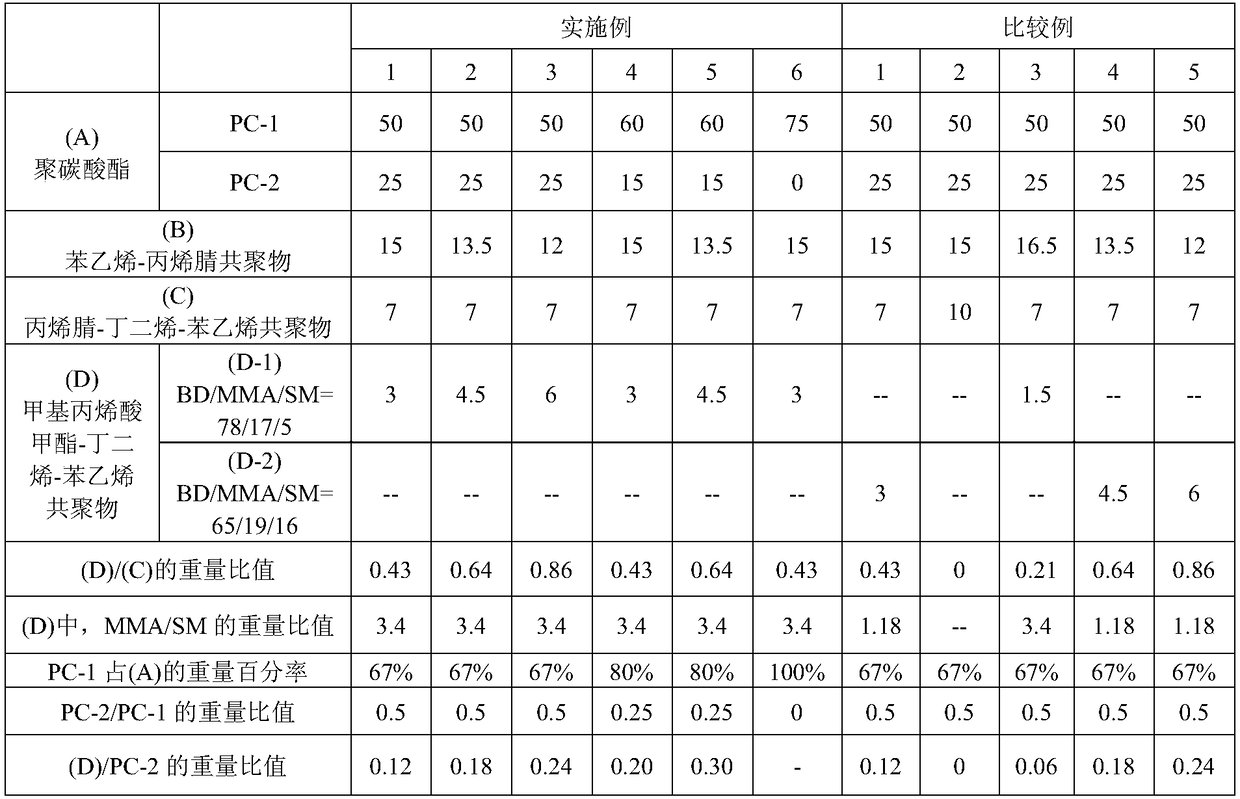
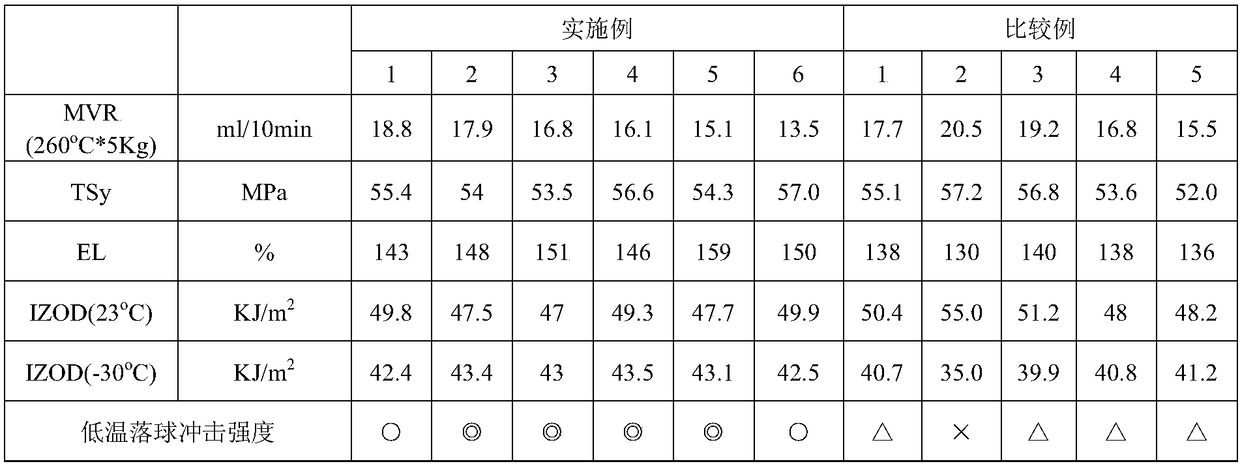
Examples
preparation example Construction
[0024] The polymerization method of styrene-acrylonitrile copolymer can adopt block polymerization method, solution polymerization method, suspension polymerization method, emulsification polymerization method, etc., among which block polymerization method or solution polymerization method is preferred. In addition, the method for preparing the styrene-acrylonitrile copolymer preferably includes performing polymerization using a reactor capable of continuous block or solution polymerization. The reactor includes: a columnar flow reactor, a completely mixed reactor (CSTR), or a tube reactor with static mixing elements, among which the completely mixed reactor is preferred. The number of reactors used may be one, or two or more reactors may be used in combination.
[0025] In one embodiment, the preparation method of the styrene-acrylonitrile copolymer is carried out by solution polymerization. The solvent used in the solution polymerization reaction is, for example, toluene, e...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More