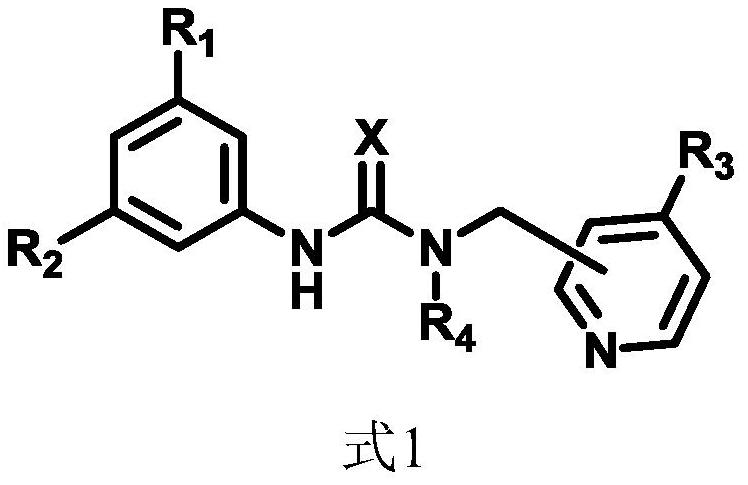
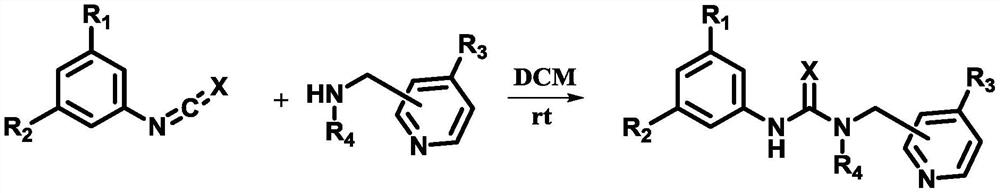
DMAP-thiourea catalyst and preparation method thereof, and high-molecular-weight biodegradable polyester and preparation method thereof
A technology for biodegrading polyester and thiourea catalysts, applied in chemical instruments and methods, physical/chemical process catalysts, organic compounds/hydrides/coordination complex catalysts, etc., can solve the problems of low molecular weight of polyesters, and achieve The effect of high molecular weight, optimized polymerization temperature, and narrow distribution
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
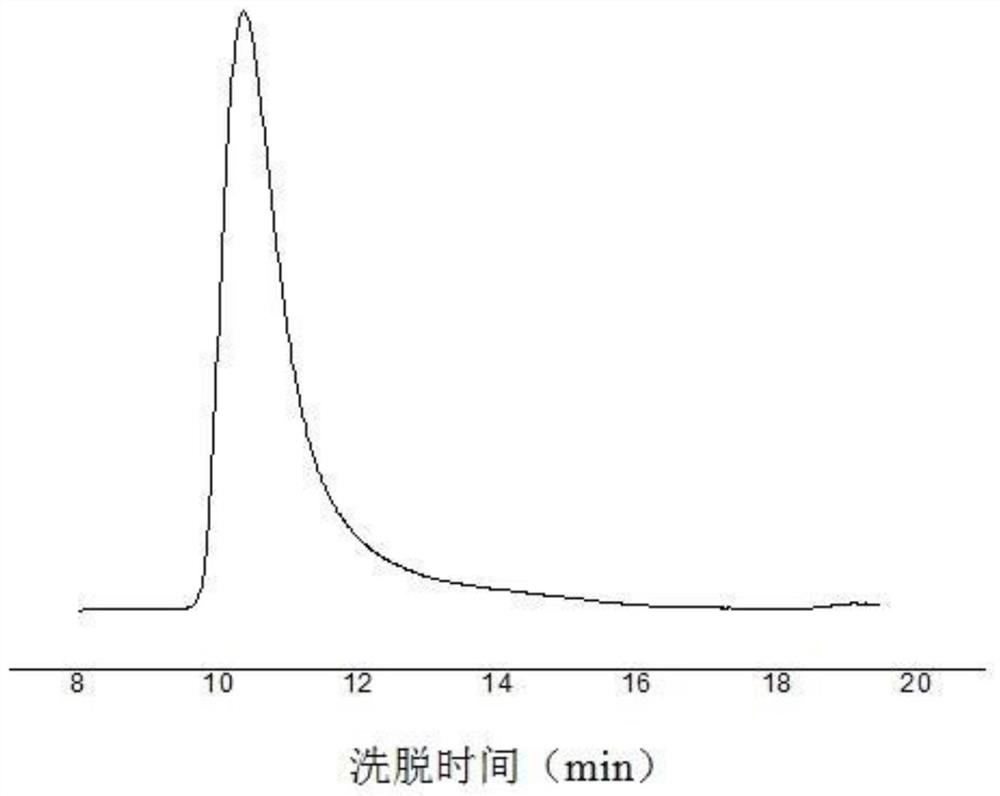
Image
Examples
preparation example Construction
[0079] The preparation method of acid anhydride in O-carboxyl ring:
[0080]
[0081] Dissolve α-hydroxy acid (10mmol) in 30mL of anhydrous tetrahydrofuran, add 200mg of activated carbon, 6.7mmol of triphosgene, stir at room temperature for 20 hours, filter with suction, and recrystallize from diethyl ether three times to obtain O-carboxyl ring anhydride. Spectra and carbon spectrum characterization, as follows.
[0082] (1) L-EthylOCA: 1 H NMR (300MHz, CDCl 3 ):δ5.06-5.10(t,1H),1.90-2.14(m,2H),1.07-1.12(t,3H). 13 C NMR (300MHz, CDCl 3 ): δ167.02, 148.50, 79.92, 21.45, 10.21.
[0083] (2) L-tBuOCA: 1 H NMR (300MHz, CDCl 3 ): δ4.71(s,1H),1.12(s,9H). 13 C NMR (300MHz, CDCl 3 ): δ165.76, 148.77, 86.80, 35.33, 25.03.
[0084] (3)L-LacOCA: 1 H NMR (300MHz, CDCl 3 ):δ5.13(q,1H),1.68(d,3H). 13 C NMR (300MHz, CDCl 3 ): δ167.96, 148.39, 76.41, 16.34.
[0085] (4) L-Ser(Bn)OCA: 1 H NMR (300MHz, CDCl 3 ):δ7.26-7.40(m,5H),5.10(m,1H),4.60(m,2H,),3.91(m,2H,). 13 C NMR (C...
preparation Embodiment 1
[0095] Preparation Example 1 Preparation of Catalyst 1
[0096]
[0097] Specific steps are as follows:
[0098] (1) Add 1.43g (10mmol) of 4-chloro-2-pyridinemethanol, 3.15g (12mmol) of triphenylphosphine, 1.77g (12mmol) of phthalimide and 15mL of anhydrous tetrahydrofuran into a round bottom flask . Stir at 0°C for 15 minutes. Azodiisopropyl ester 2.4mL (15mmol) was slowly added dropwise into the solution. After the dropwise addition, slowly return to room temperature and stir for 12h. A white solid was obtained by suction filtration with a yield of 60%. With deuterated chloroform (CDCl 3 ) as a reagent with a 300 MHz nuclear magnetic resonance instrument (hydrogen spectrum, 1 H NMR) to characterize its structure. 1 H NMR (300MHz, CDCl 3 ): δ8.43(d,1H),7.89-7.91(m,2H),7.74-7.77(m,2H),7.29(s,1H),7.20(d,1H),4.99(s,2H).
[0099] (2) Dissolve 1.36g (5mmol) of the product from the previous step in 15mL of ethanol, add 2.4mL of hydrazine hydrate and reflux for 3h. Extr...
preparation Embodiment 2
[0102] Preparation Example 2 Preparation of Catalyst 5
[0103]
[0104] Specific steps are as follows:
[0105] Steps (1) and (2) follow steps (1) and (2) of Catalyst 1 synthesis.
[0106] (3) Put 0.50g (3.5mmol) of 4-chloro-2-pyridinemethylamine, 10mL of water, 2.49g (35mmol) of pyrrolidine and 1.4g (35mmol) of sodium hydroxide in a sealed tank, and stir at 100°C for 24 hours , to obtain 4-pyrrolidinyl-2-pyridinemethanamine with a yield of 60%. deuterated dimethyl sulfoxide (DMSO-d 6 ) as a reagent with a 300 MHz nuclear magnetic resonance instrument (hydrogen spectrum, 1 H NMR) characterized the structure of 4-pyrrolidinyl-2-pyridinemethylamine. 1 H NMR (300 MHz, DMSO-d 6 ):δ8.00(d,1H),6.48(s,1H),6.30(d,1H),3.61(s,2H),3.24(m,4H),1.94(m,4H).
[0107] (4) Add 0.354g (2mmol) of 4-pyrrolidinyl-2-pyridinemethanamine, 0.540g (2mmol) of 3,5-bis(trifluoromethyl)phenylisothiocyanate and 3 mL of dichloromethane. Stir at room temperature for 12 hours. Catalyst 5 was obtain...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap