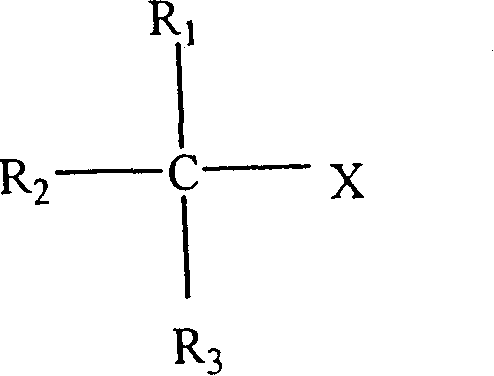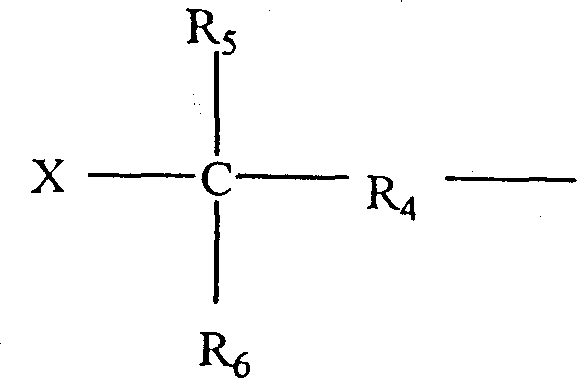Elastomeric compositions
A composition and elastomer technology, applied in tire parts, vehicle parts, special tires, etc., can solve problems such as failure to achieve or achieve high enough laboratory wear resistance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment
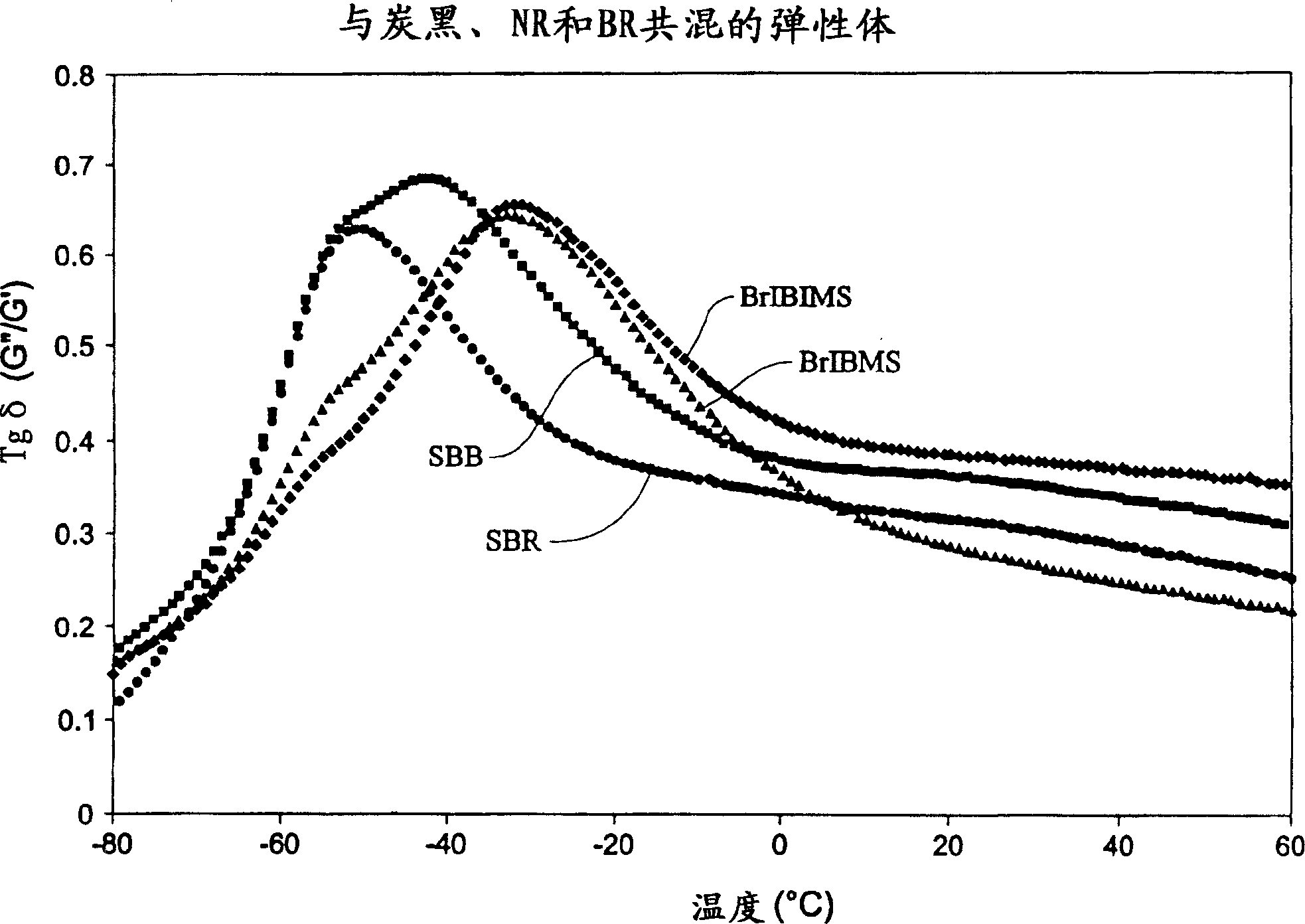
[0094] Although not meant to be limited by the following examples and tables, the present invention may be better understood by reference to the following examples and tables. The following symbols are used throughout the description to describe the rubber components of the present invention: IBIMS {terpolymer; poly(isobutylene-co-p-methylstyrene-co-isoprene)}; BrIBIMS {brominated terpolymer Copolymer; brominated poly(isobutylene-co-p-methylstyrene-co-isoprene)}; IBMS{poly(isobutylene-co-p-methylstyrene)}; BrIBMS{brominated poly( isobutylene-co-p-methylstyrene-co-p-bromomethylstyrene)}; SBB{brominated star-branched butyl rubber}; BR{polybutadiene}; NR{natural rubber}; and SBR {styrene-butadiene rubber}.
[0095] The synthesis of the terpolymers used in the present invention was carried out in two sets of 6-sample batch-operated apparatus, each demonstrating the use of two different initiators. Tert-butyl chloride (t-BuCl) was used as the initiator for tests A-F, and its data...
PUM
| Property | Measurement | Unit |
|---|---|---|
| wear resistance | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com