Methods and means for increasing resistance to cell damage
a cell damage and cell technology, applied in the field of cell damage, can solve the problems that the overall rate of protein turnover can affect the cell's response to cell damage, and achieve the effects of increasing the overall protein turnover in cells, increasing the resistance to cell damage, and increasing the protein turnover in cells
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Plasmids, Antibodies and Cells
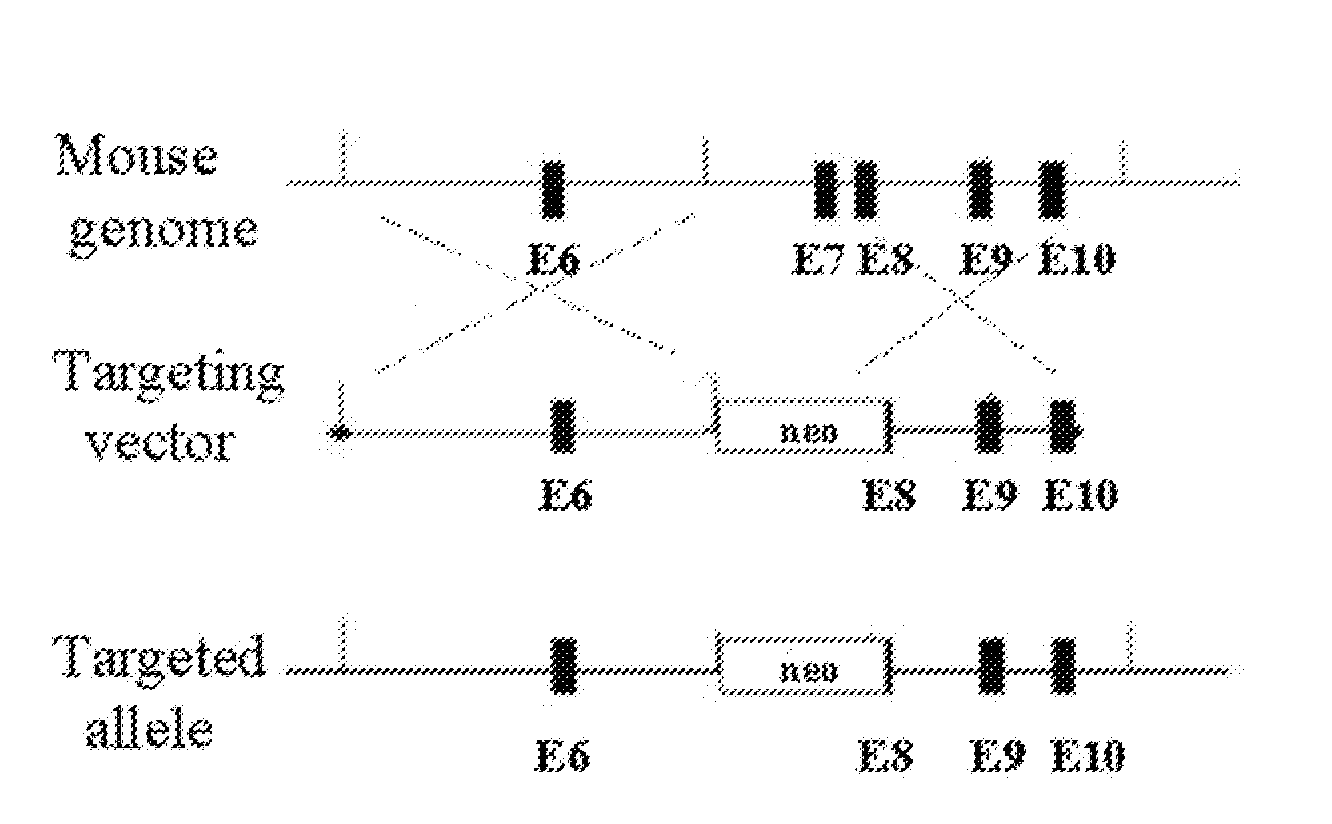
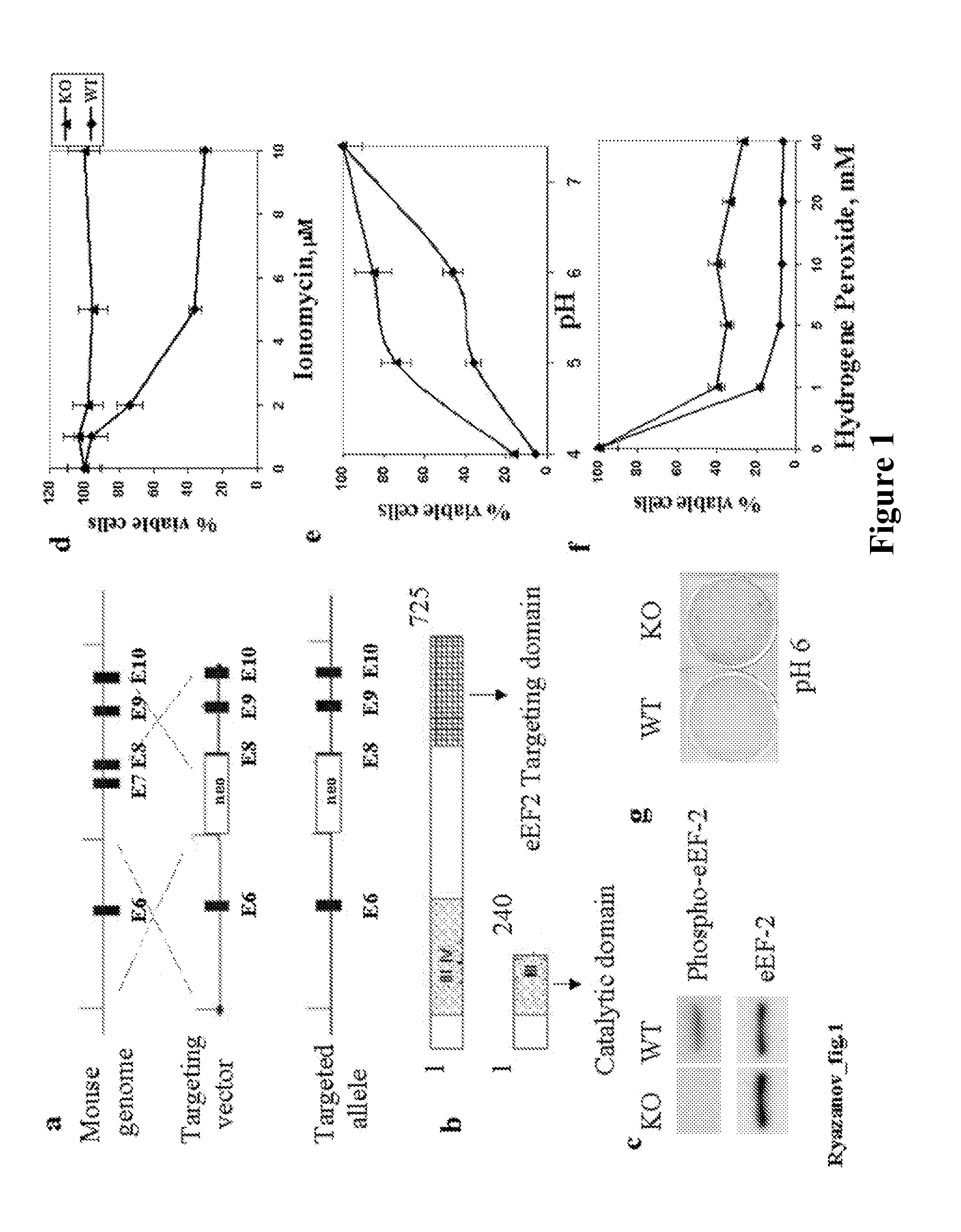
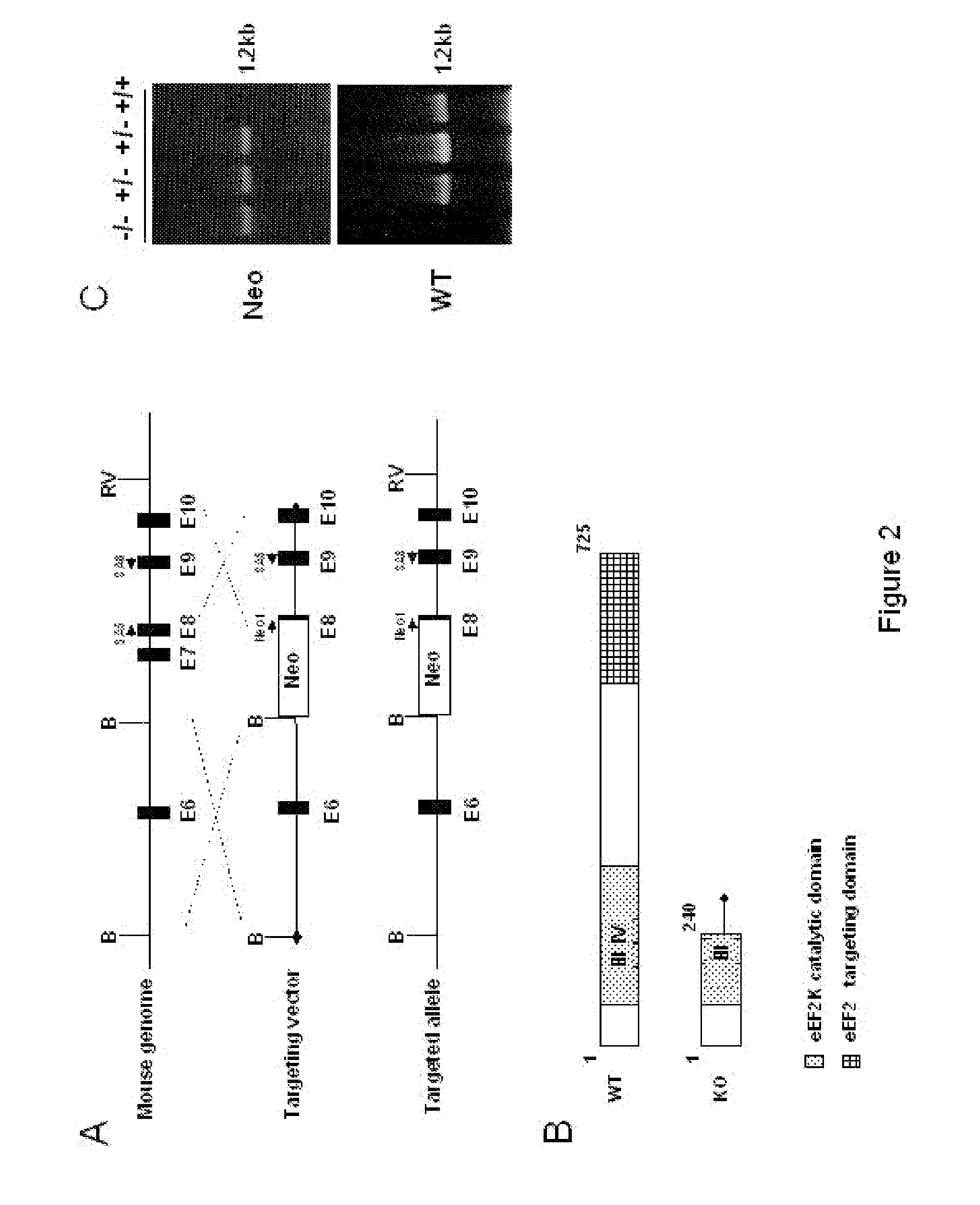
[0128]Retroviral vector used for eEF2 kinase overexpression is constructed by subcloning of eEF2 kinase cDNA from the pSIT retroviral vector7 into LXSN vector (Clontech) using Eco RI / XhoI cloning sites. Stable cell line is prepared through infection of MEFs with pBabe-neo retroviral vector containing SV-40 large T antigen (a kind gift from Dr. J. Yuan). GSE 56 cell lines are established using retroviral vector LXSP, containing GSE5612. LXSP vector is prepared from LXSN vector by the substitution of neomycin marker with puromycin. Antibodies against p21 (F5) and p53 (Ab-1) are from Calbiochem Inc.; antibodies against eEF2 and phospho-eEF2 are from Cell Signaling Inc. eEF2 kinase− / − and eEF2 kinase+ / + primary mouse embryonic fibroblasts used in this study are isolated from 10-12 day embryos following standard protocols. Unless indicated, all cell lines are maintained in DMEM with 10% fetal bovine serum.
example 2
Transfection and Retroviral Infection
[0129]Packaging cells (Phoenix line) are plated in 60-mm plates and transfected with 5 μg of retroviral vector DNA using the standard calcium phosphate procedure. Medium is changed after 8 hours. Virus-containing medium supplemented with 8 μg of Polybrene (Sigma) is collected at 24 and 48 hours post-transfection and used for infection. Infected cells are selected for the resistance to an appropriate selection agent.
example 3
Western Analysis
[0130]For protein expression analysis, cells are washed twice with ice-cold PBS, resuspended in lysis buffer (20 mM Na-phosphate [pH 7.5], 25 mM NaF, 1 mM orthovanadate, 5 mM EDTA), dissolved in Laemmli SDS sample buffer and boiled for 10 minutes. Samples are separated on 5-20% gradient SDS-PAGE and proteins are transferred onto a PVDF membrane. Membranes are incubated with antibodies and developed using ECL Plus reagents (Amersham Biosciences).
PUM
| Property | Measurement | Unit |
|---|---|---|
| Electrical resistance | aaaaa | aaaaa |
| Acidity | aaaaa | aaaaa |
| Catalytic activity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More