Patents
Literature
1037 results about "Neutralizing antibody" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
A neutralizing antibody (NAb) is an antibody that defends a cell from an antigen or infectious body by neutralizing any effect it has biologically. An example of a neutralizing antibody is diphtheria antitoxin, which can neutralize the biological effects of diphtheria toxin.
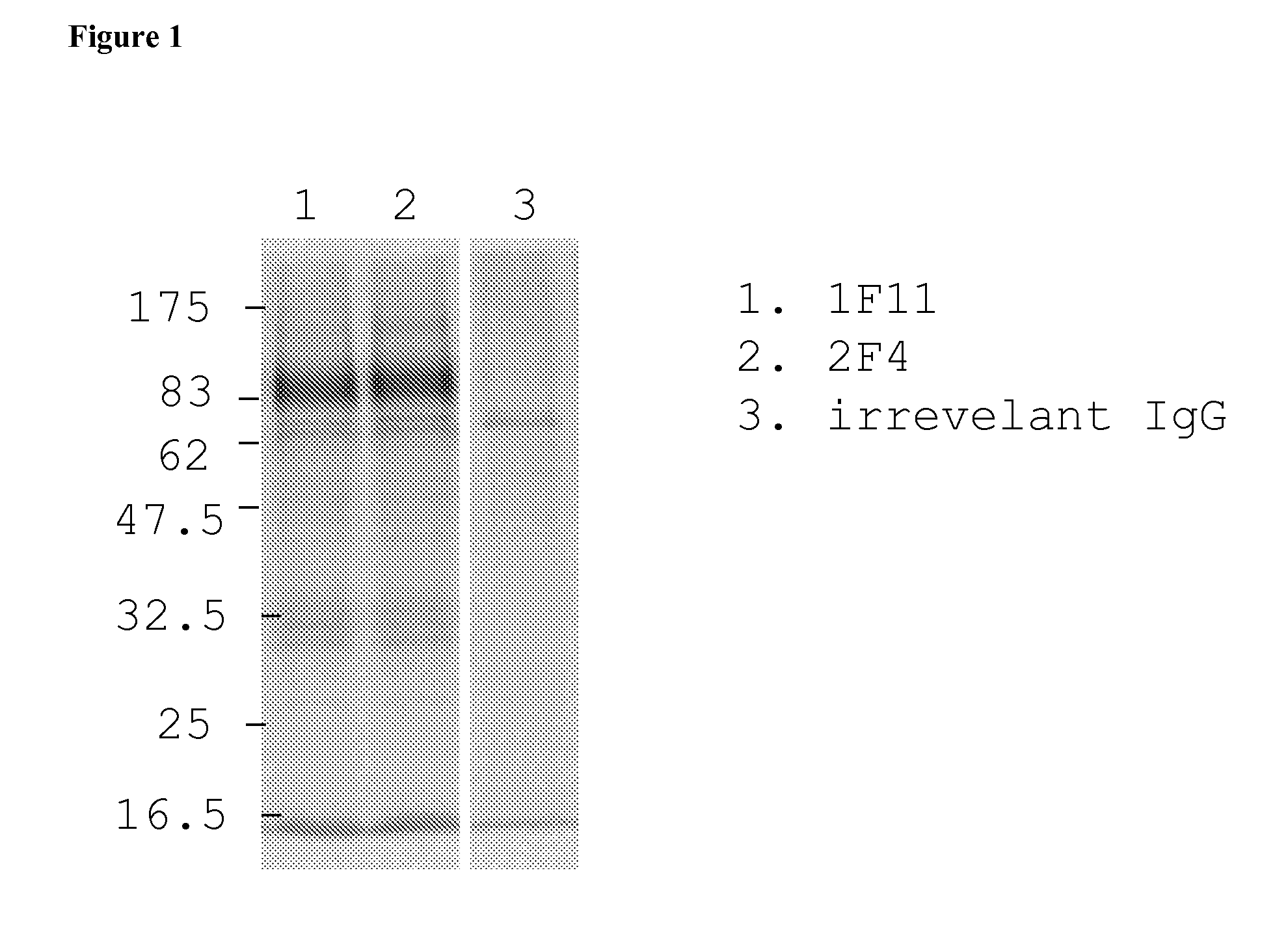
Mutant adeno-associated virus virions and methods of use thereof
ActiveUS20090202490A1Reduce the binding forceAltered infectivityOrganic active ingredientsBiocideCell type specificNeutralizing antibody
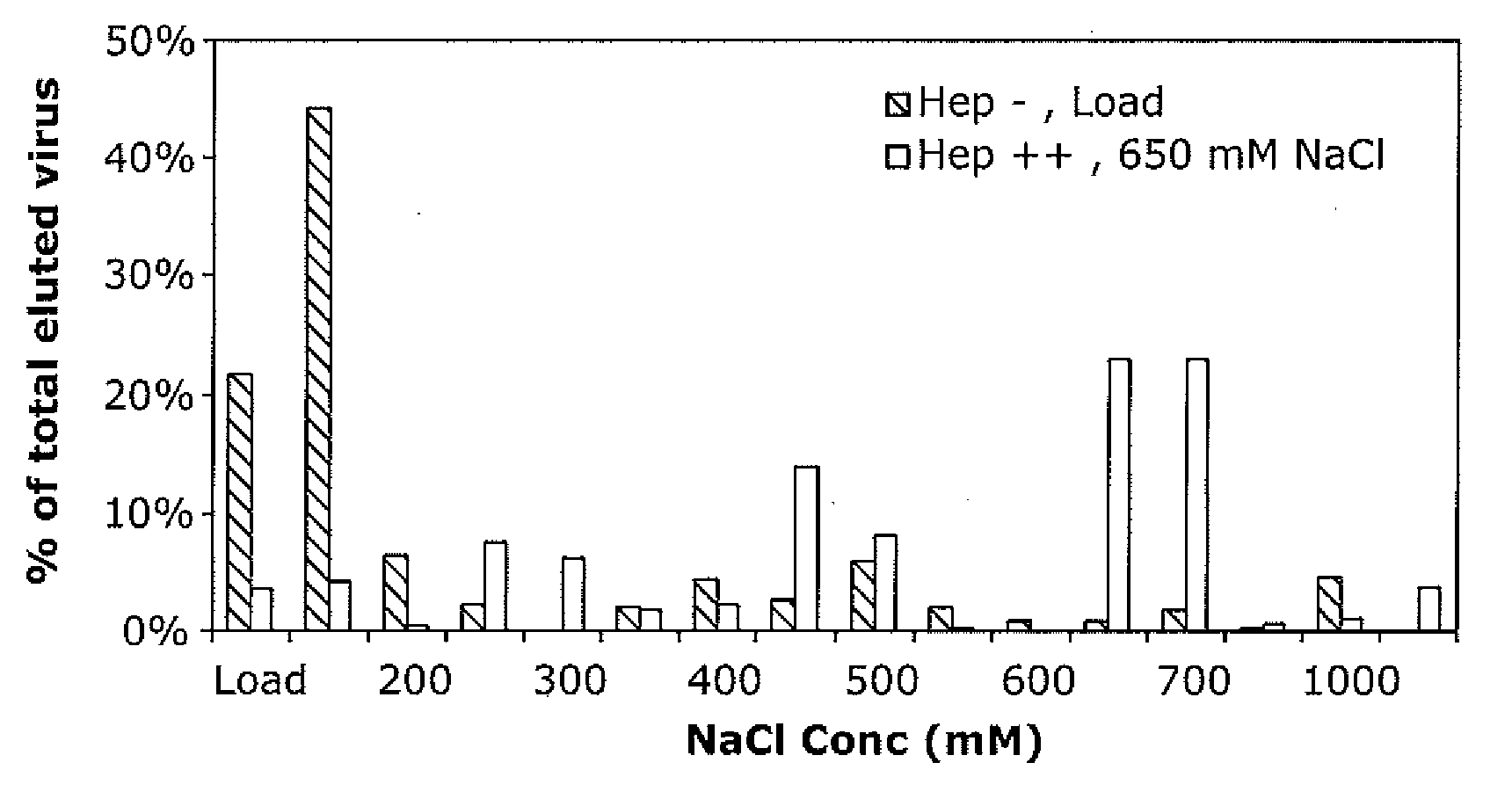
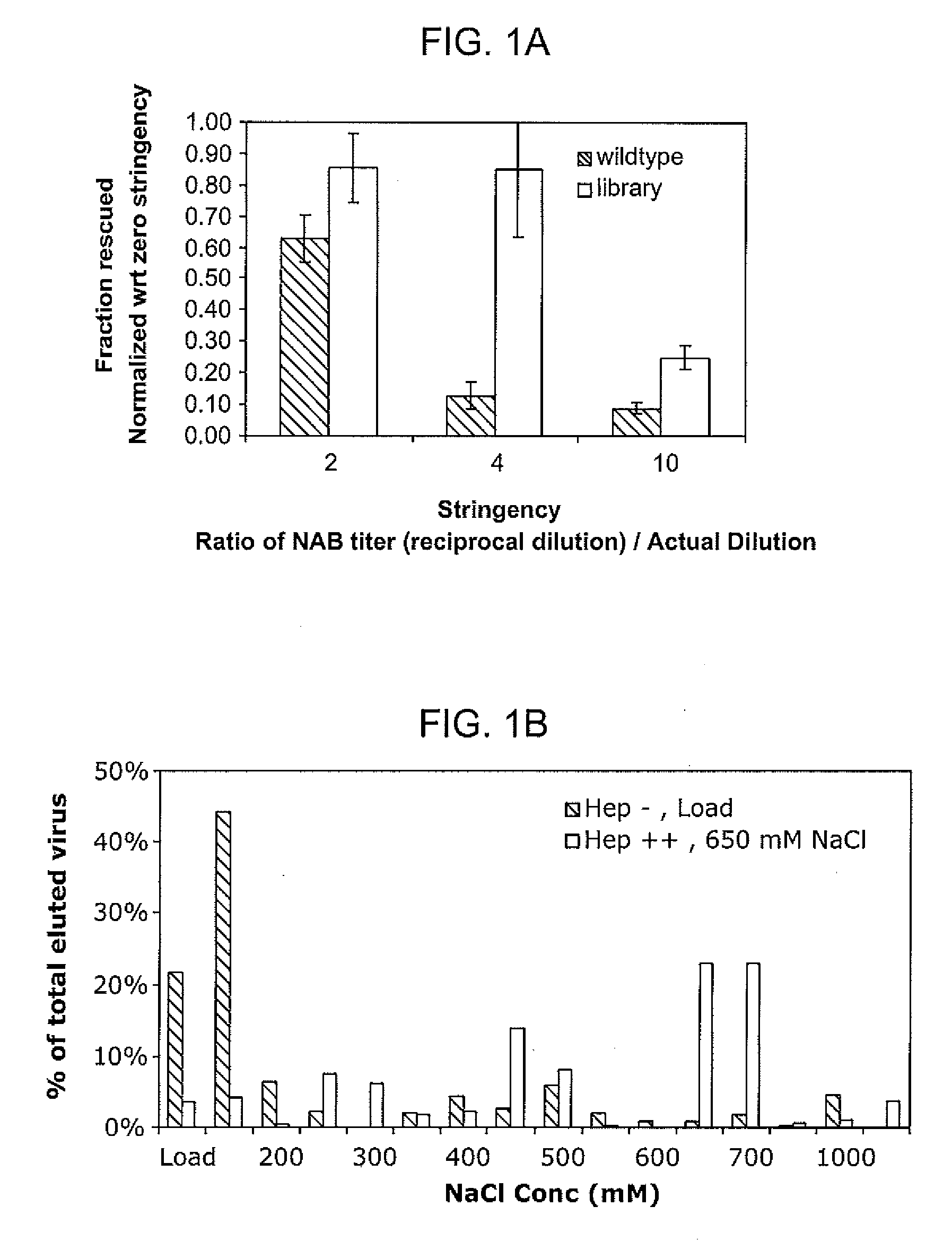
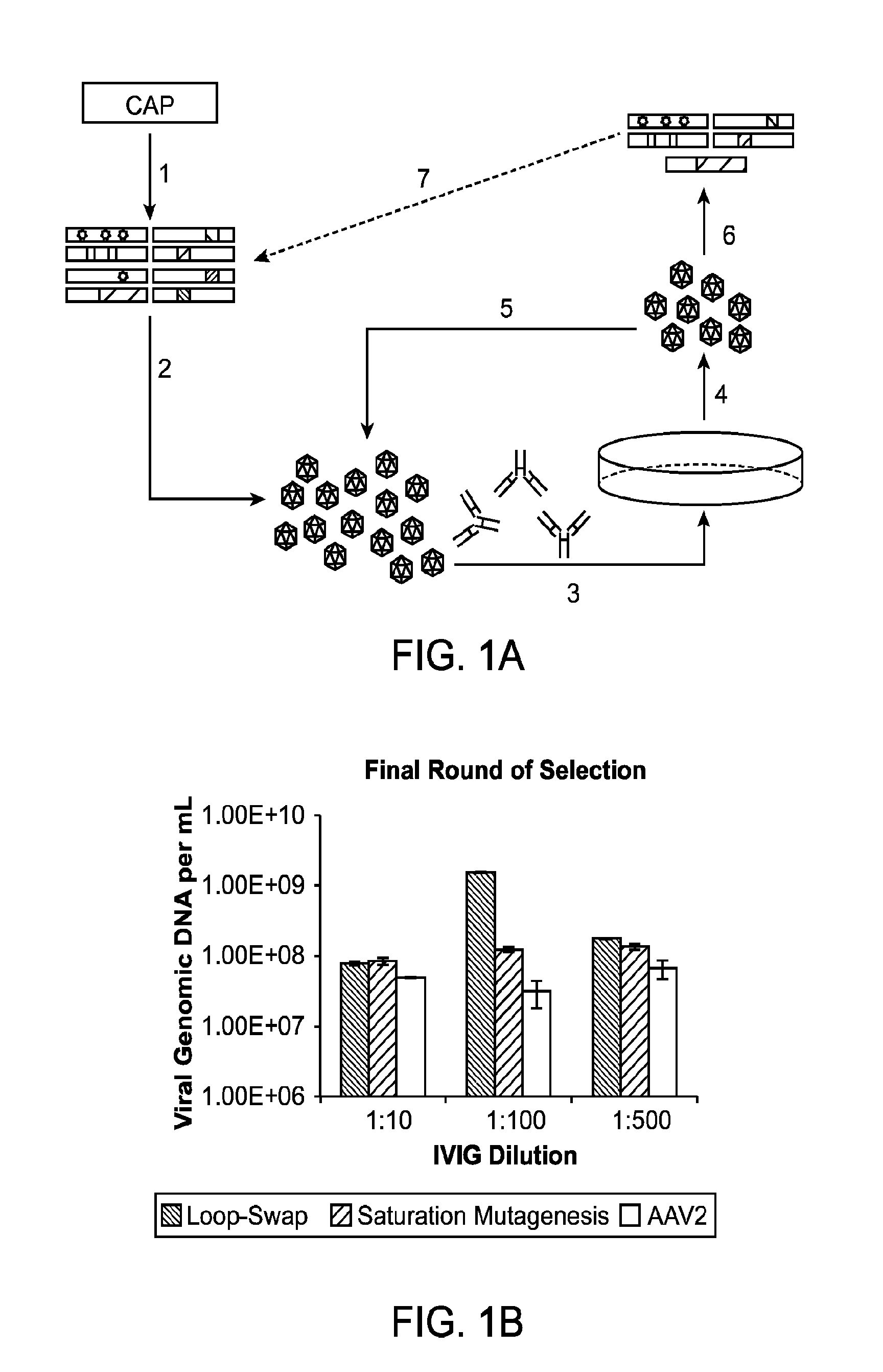
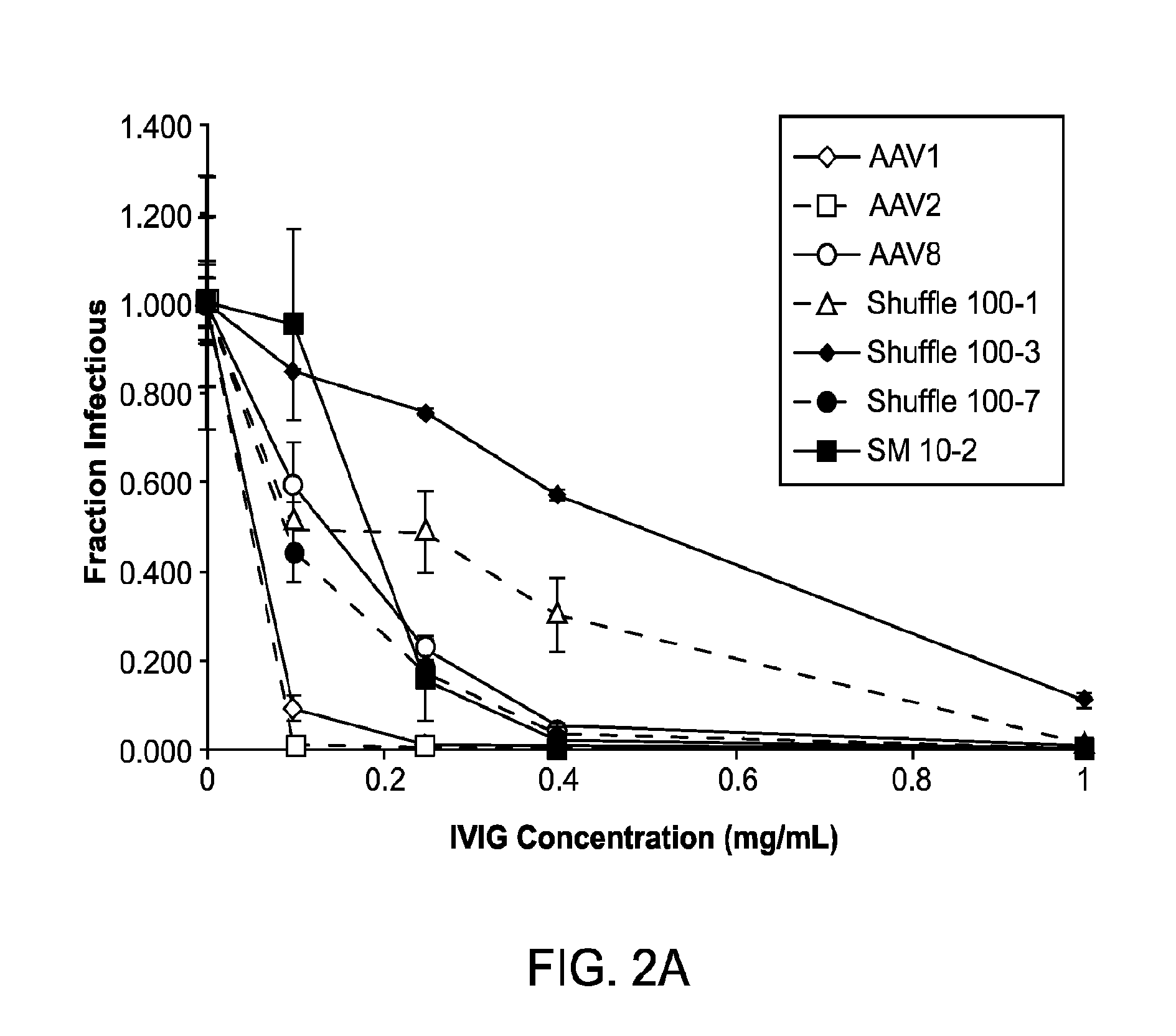
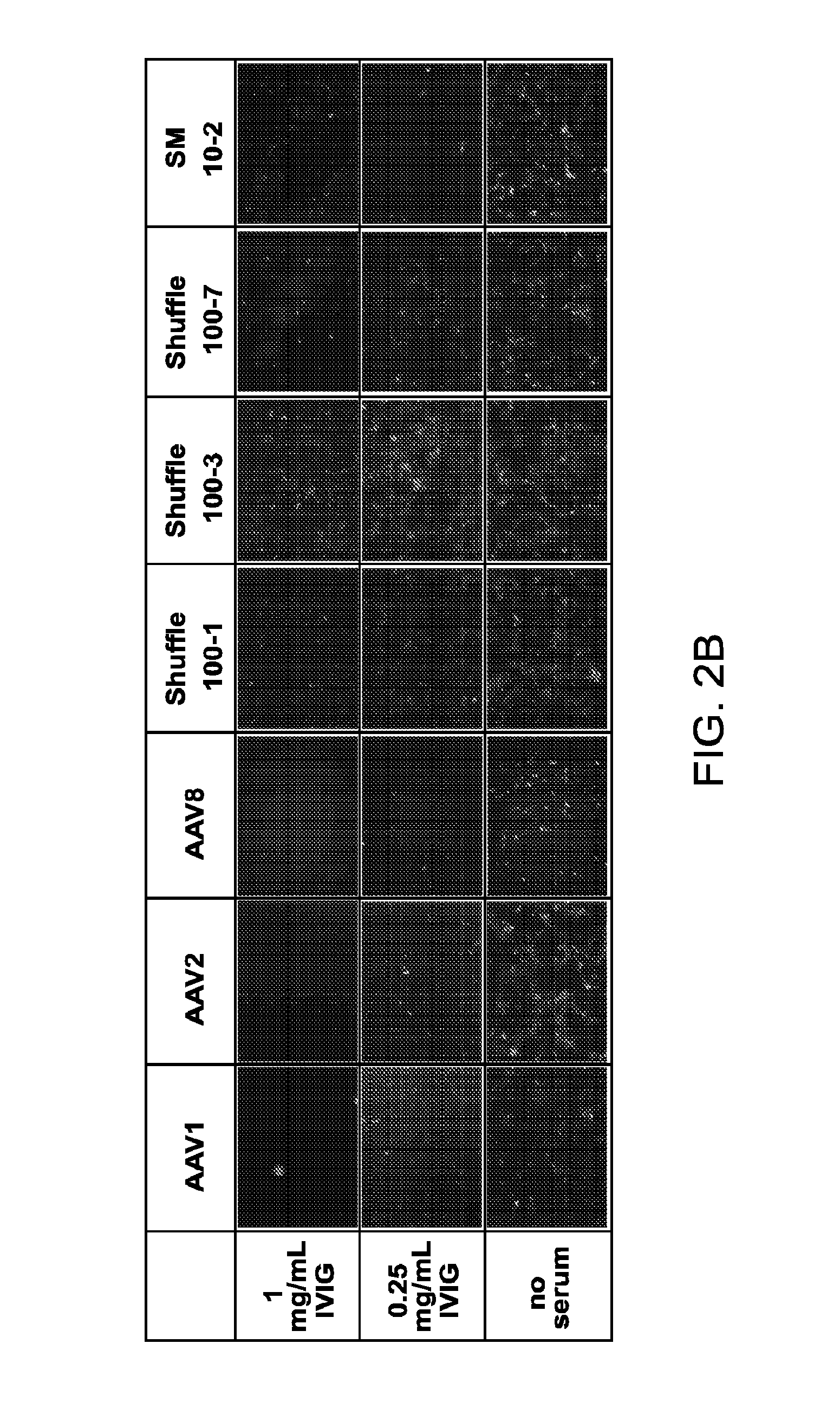
The present invention provides mutant adeno-associated virus (AAV) that exhibit altered capsid properties, e.g., reduced binding to neutralizing antibodies in serum and / or altered heparin binding and / or altered infectivity of particular cell types. The present invention further provides libraries of mutant AAV comprising one or more mutations in a capsid gene. The present invention further provides methods of generating the mutant AAV and mutant AAV libraries, and compositions comprising the mutant AAV. The present invention further provides recombinant AAV (rAAV) virions that comprise a mutant capsid protein. The present invention further provides nucleic acids comprising nucleotide sequences that encode mutant capsid proteins, and host cells comprising the nucleic acids. The present invention further provides methods of delivering a gene product to an individual, the methods generally involving administering an effective amount of a subject rAAV virion to an individual in need thereof.
Owner:RGT UNIV OF CALIFORNIA +1
Mutant adeno-associated virus virions and methods of use thereof
ActiveUS20050053922A1Reduce the binding forceAltered infectivityAntibacterial agentsVirusesReassortant VirusesNeutralizing antibody
The present invention provides mutant adeno-associated virus (AAV) that exhibit altered capsid properties, e.g., reduced binding to neutralizing antibodies in serum and / or altered heparin binding and / or altered infectivity of particular cell types. The present invention further provides libraries of mutant AAV comprising one or more mutations in a capsid gene. The present invention further provides methods of generating the mutant AAV and mutant AAV libraries, and compositions comprising the mutant AAV. The present invention further provides recombinant AAV (rAAV) virions that comprise a mutant capsid protein. The present invention further provides nucleic acids comprising nucleotide sequences that encode mutant capsid proteins, and host cells comprising the nucleic acids. The present invention further provides methods of delivering a gene product to an individual, the methods generally involving administering an effective amount of a subject rAAV virion to an individual in need thereof.
Owner:INTEGRATIVE GENE THERAPEUTICS +1
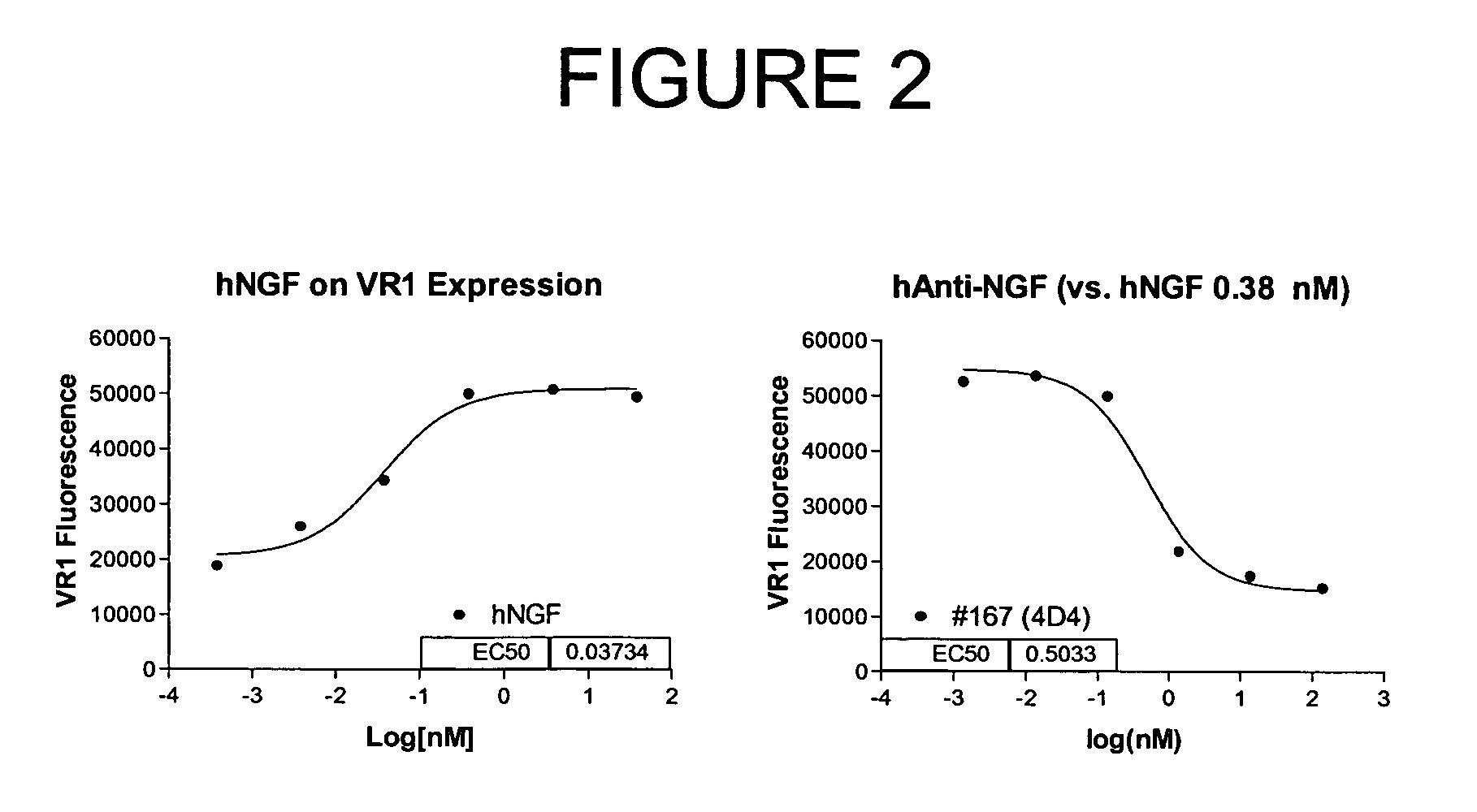
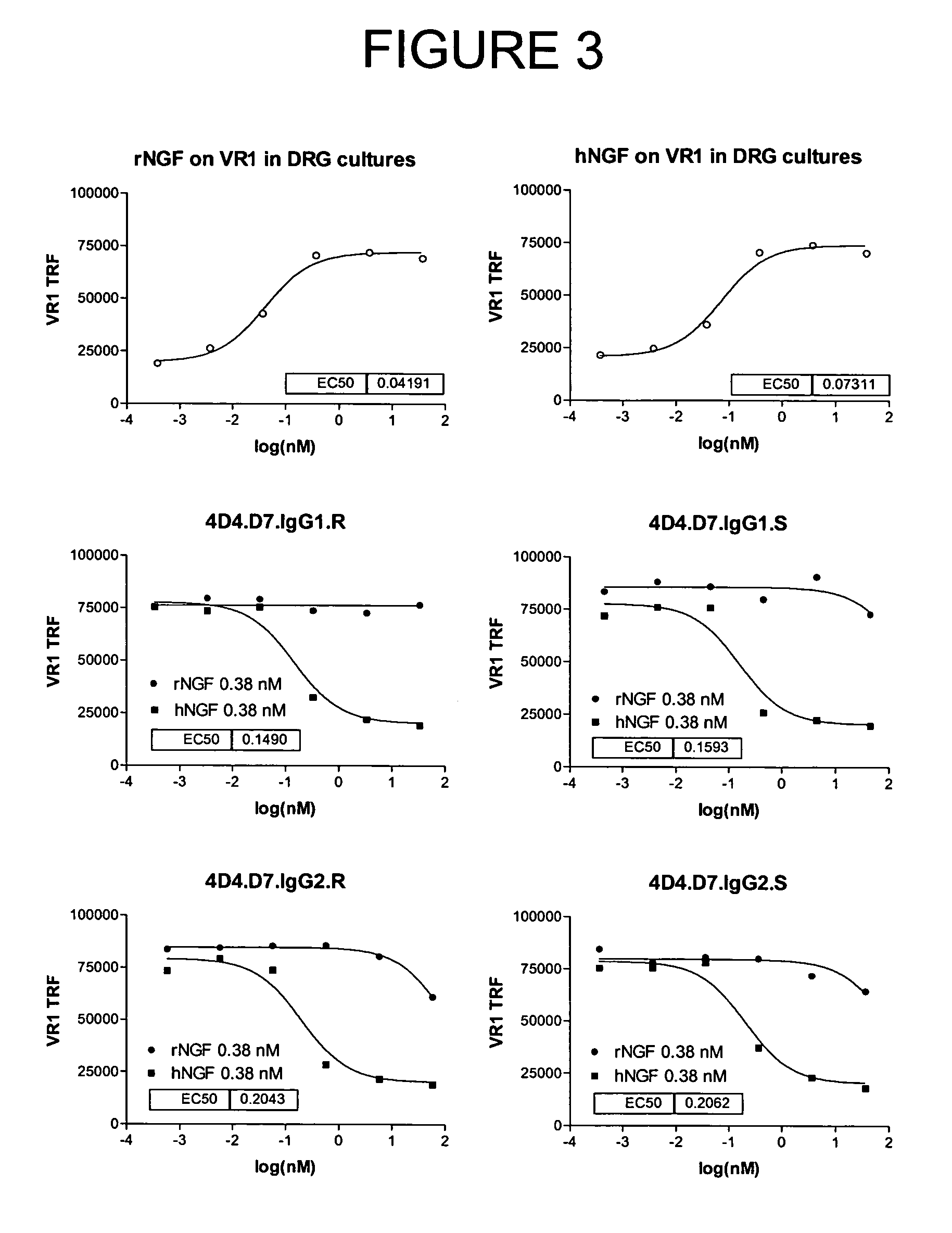
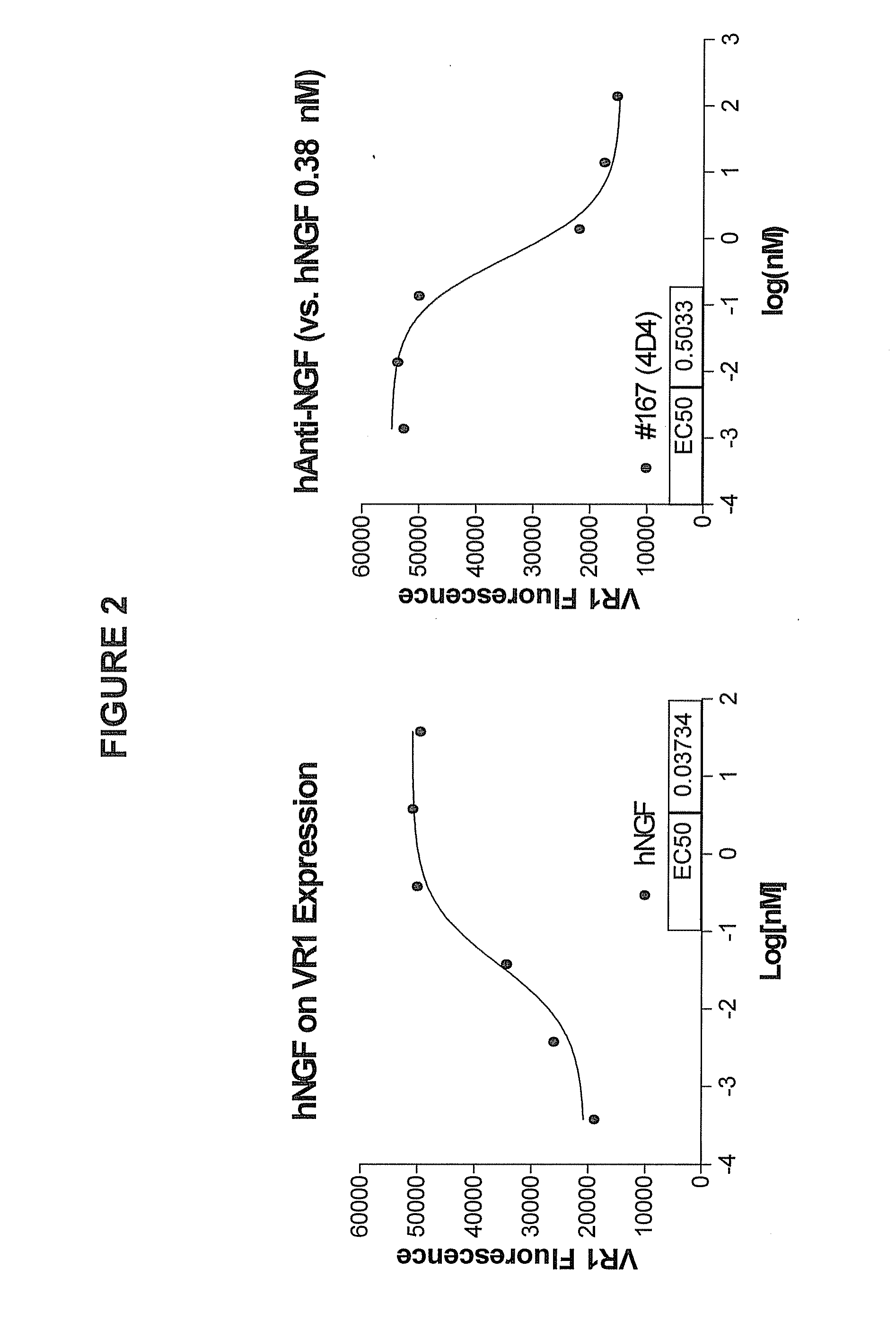
Human anti-NGF neutralizing antibodies as selective NGF pathway inhibitors
InactiveUS7601818B2Increase productionHigh sensitivitySenses disorderAntipyreticChronic painNeutralizing antibody
This invention provides antibodies that interact with or bind to human nerve growth factor (NGF) and neutralize the function of NGF thereby. The invention also provides pharmaceutical compositions of said antibodies and methods for neutralizing NGF function, and particularly for treating NGF-related disorders (e.g., chronic pain) by administering a pharmaceutically effective amount of anti-NGF antibodies. Methods of detecting the amount of NGF in a sample using anti-NGF antibodies are also provided.
Owner:ER SQUIBB & SONS INC
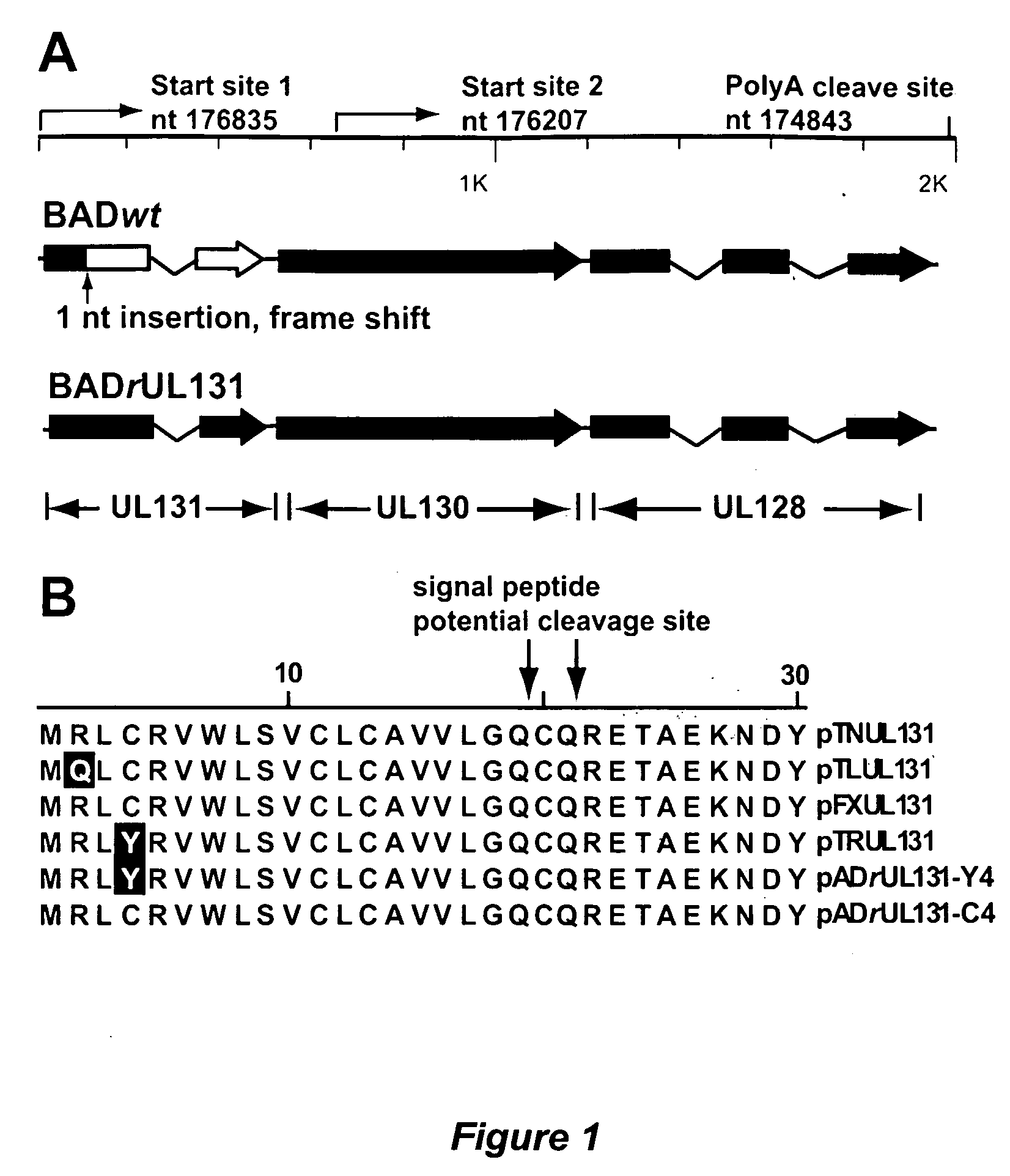
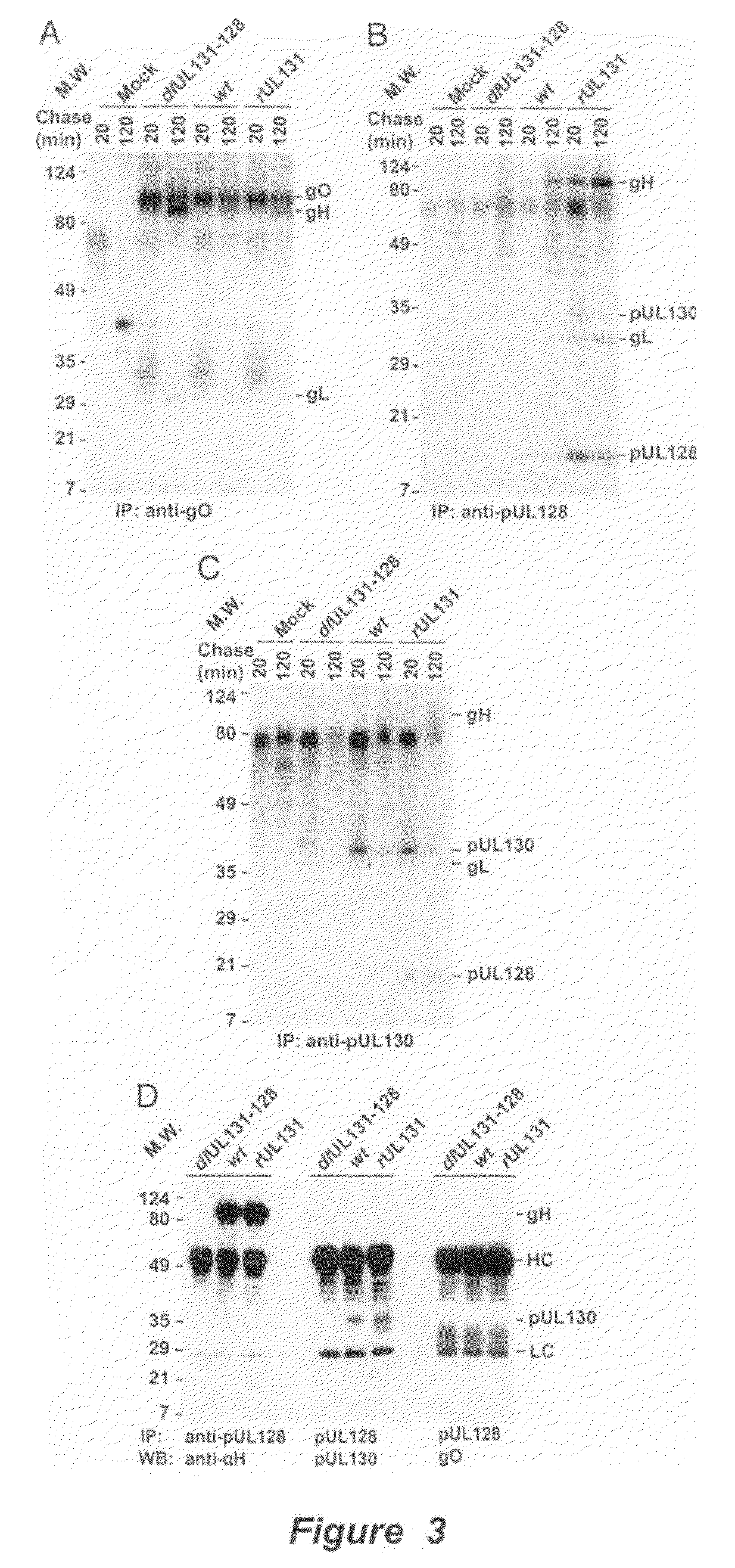
Cytomegalovirus surface protein complex for use in vaccines and as a drug target
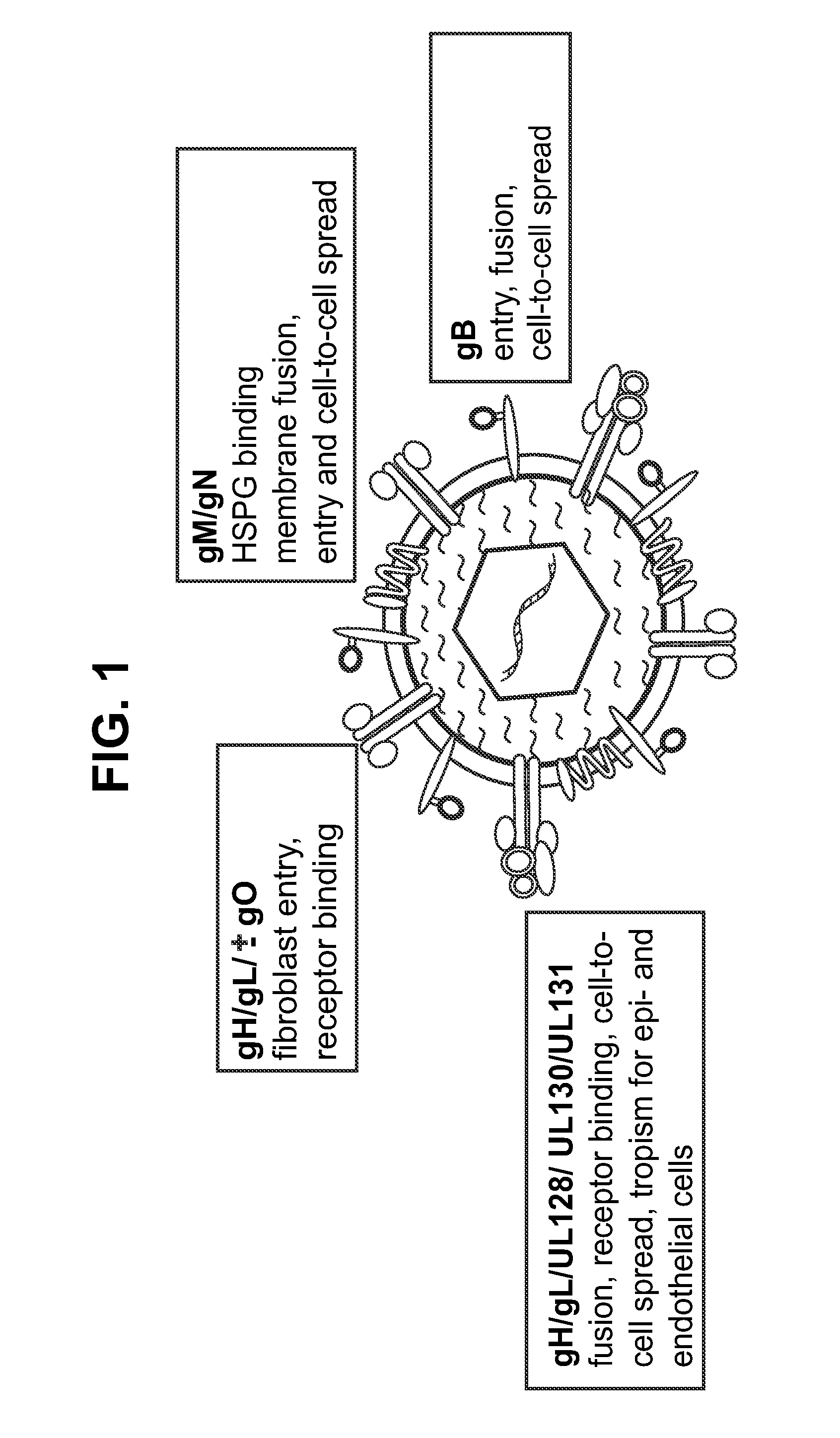
Immunogenic compositions and prophylactic or therapeutic vaccines for use in protecting and treating against human cytomegalovirus (CMV) are disclosed. Subunit vaccines comprising a human CMV protein complex comprising pUL128 or pUL130, and nucleic acid vaccines comprising at least one nucleic acid encoding a CMV protein complex comprising pUL128 or pUL130 are described. Also disclosed are therapeutic antibodies reactive against a CMV protein complex comprising pUL128 or pUL130, as well as methods for screening compounds that inhibit CMV infection of epithelial and endothelial cells, methods for immunizing a subject against CMV infection, methods for determining the capability of neutralizing antibodies to inhibit human CMV infection of cell types other than fibroblasts, and methods of diminishing an CMV infection.
Owner:THE TRUSTEES FOR PRINCETON UNIV
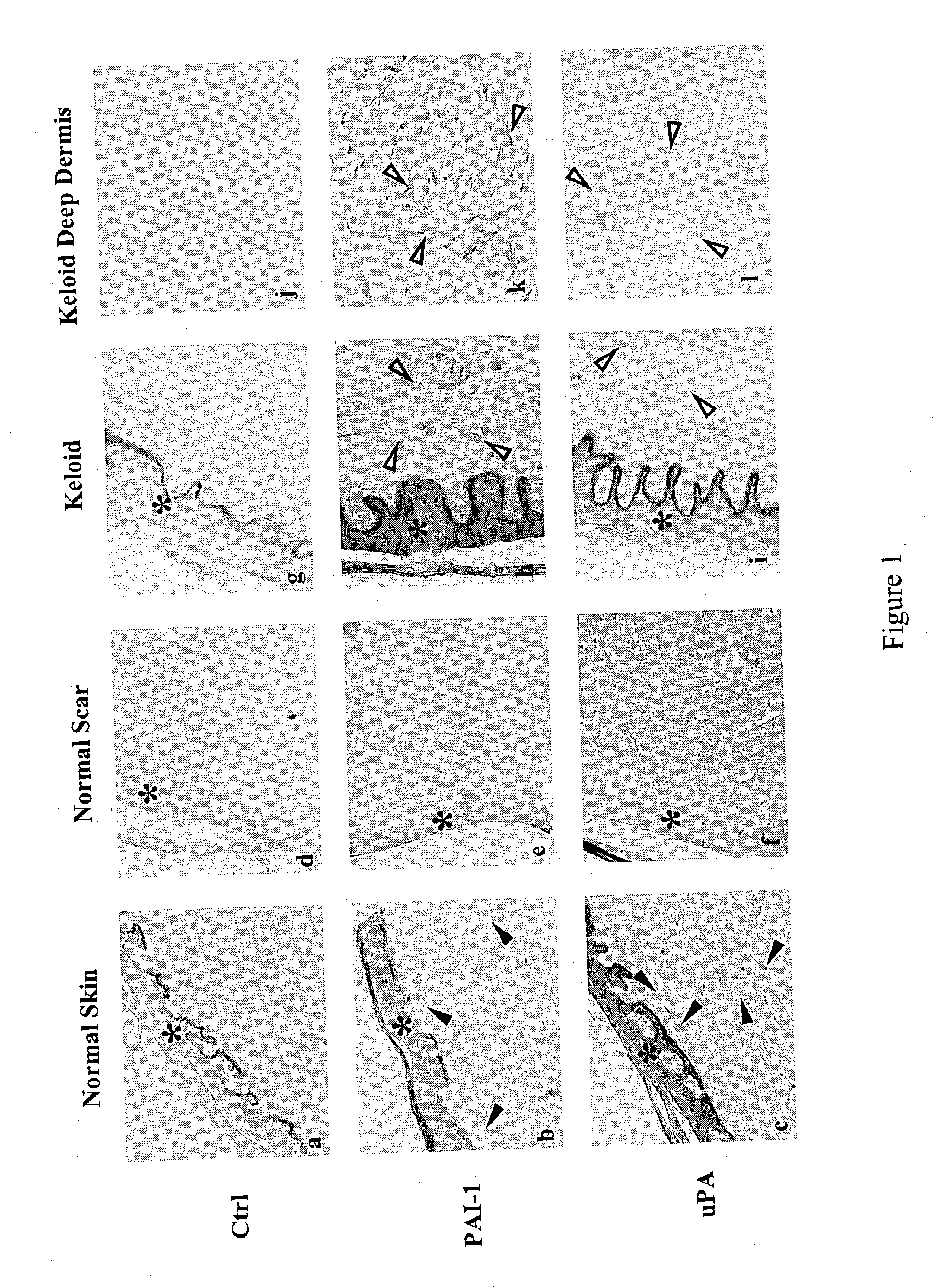
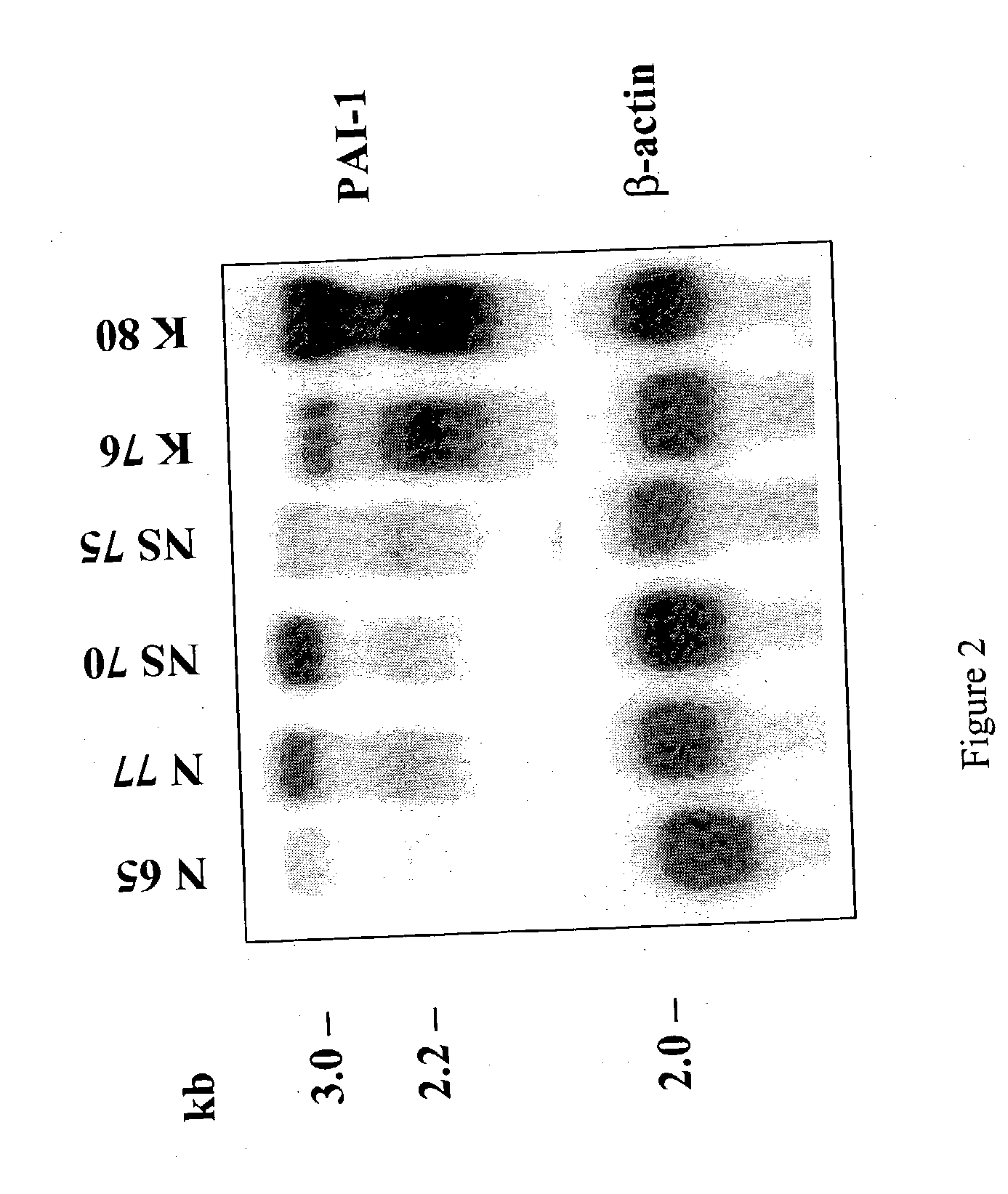
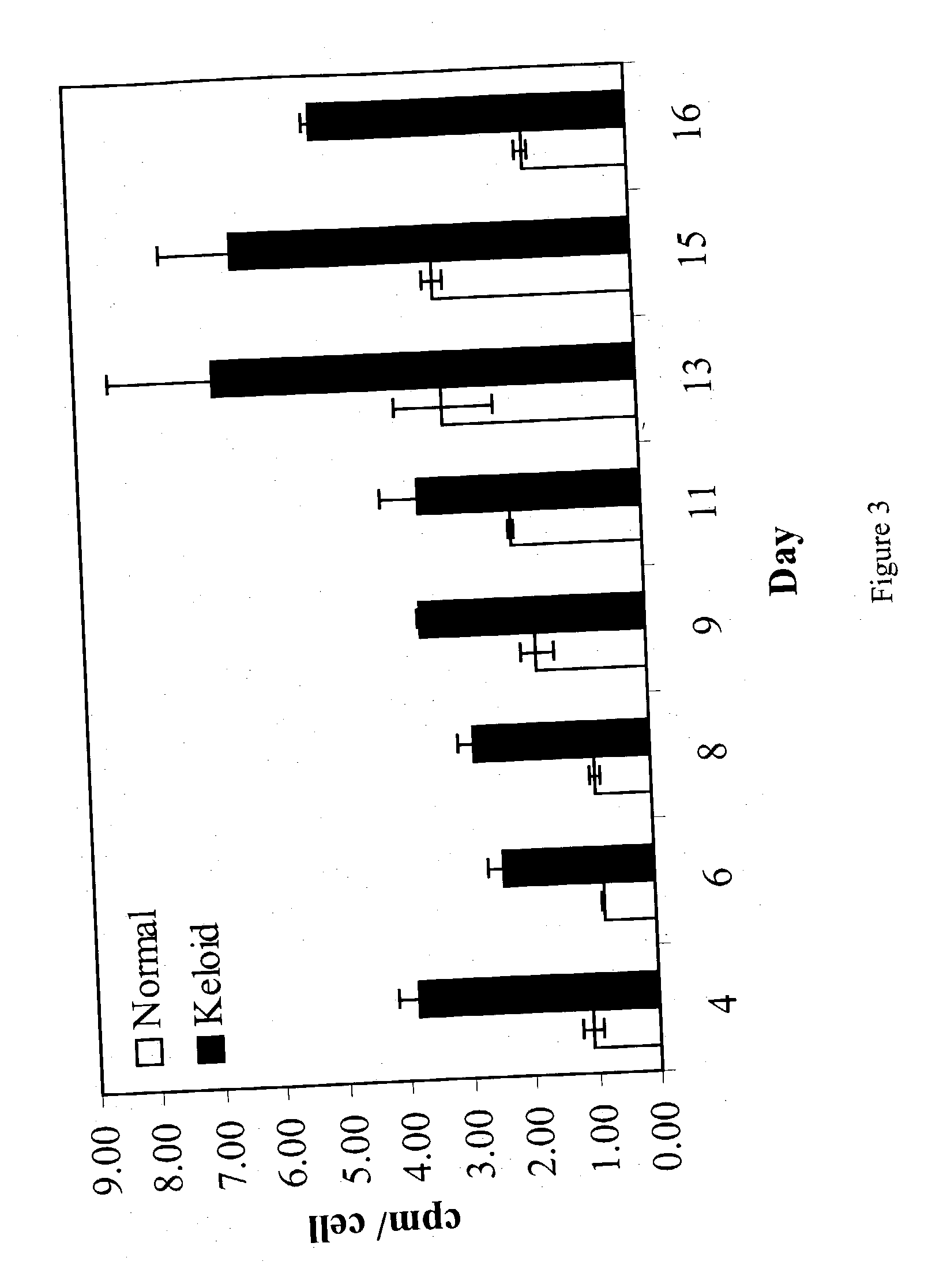
Treatment and prevention of abnormal scar formation in keloids and other cutaneous or internal wounds or lesions
The present invention relates to findings that reducing the activity of Plasminogen Activator Inhibitor-1 (PAI-1) suppresses an excessive deposition of collagen which is known as a cause for the formation of abnormal scars. These abnormal scars include but are not limited to keloids, adhesions, hypertrophic scars, skin disfiguring conditions, fibrosis, fibrocystic conditions, contractures, and scleroderma, all of which are associated with or caused by an excessive deposit of collagen in a wound healing process. Accordingly, aspects of the present invention are directed to the reduction of PAI-1 activity to decrease an excessive accumulation of collagen, prevent the formation of an abnormal scar, and / or treat abnormal scars that result from an excessive accumulation of collagen. The PAI-1 activity can be reduced by PAI-1 inhibitors which include but are not limited to PAI-1 neutralizing antibodies, diketopiperazine based compounds, tetramic acid based compounds, hydroxyquinolinone based compounds, Enalapril, Eprosartan, Troglitazone, Vitamin C, Vitamin E, Mifepristone (RU486), and Spironolactone to name a few. Another aspect of the present invention is directed to methods of measuring PAI-1 activity in a wound healing process and determining the propensity of the formation of an abnormal scar.
Owner:CHILDRENS HOSPITAL OF LOS ANGELES +1
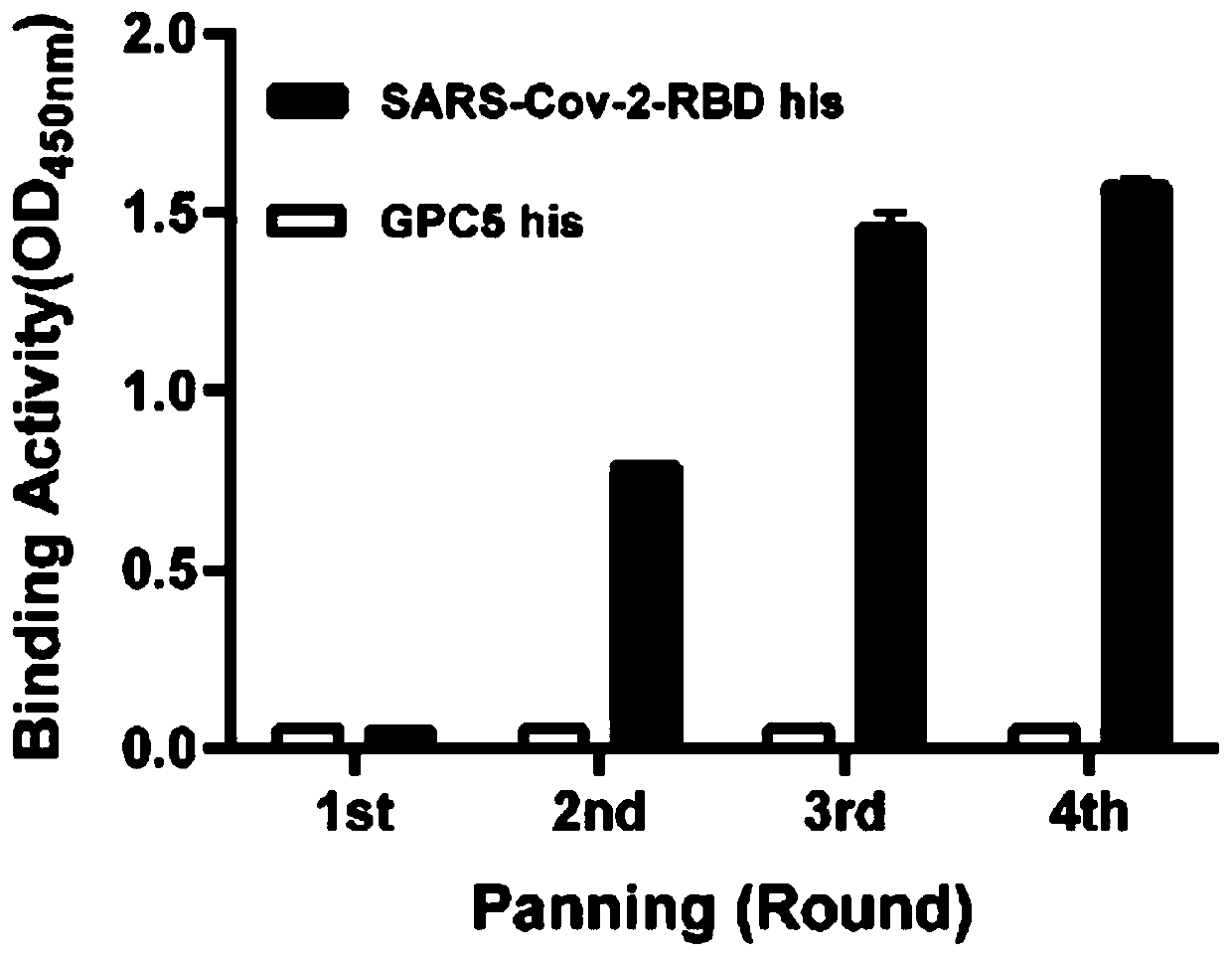
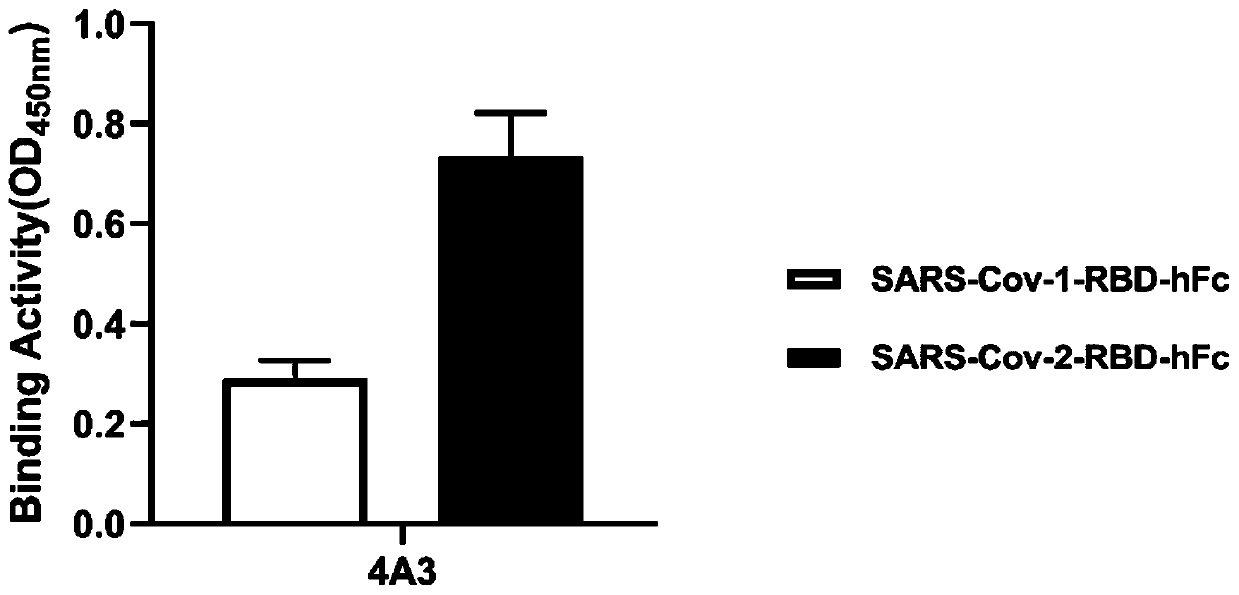
Neutralizing antibody for resisting novel coronavirus SARS-Cov-2 and application thereof
ActiveCN111592595ABlock bindingGood effectImmunoglobulins against virusesAntiviralsNeutralizing antibodyPhage Display Techniques
The invention relates to a neutralizing antibody for resisting a novel coronavirus SARS-Cov-2 and an application of the neutralizing antibody. The antibody at least comprises one of a heavy chain CDR1, a heavy chain CDR2, a heavy chain CDR3, a light chain CDR1, a light chain CDR2 and a light chain CDR3. The antibody can be used for preparing a diagnostic reagent or a diagnostic kit, a drug or a pharmaceutical composition for detecting, preventing and treating a COVID-19. According to the neutralizing antibody, differential antibody screening is carried out through a phage display technology ina manner of targeting SARS-Cov-2-RBD and SARS-Cov-1-RBD; the neutralizing antibody for resisting the novel coronavirus SARS-Cov-2 is obtained; binding of the SARS-Cov-2-RBD and ACE2 positive cells can be blocked; and the neutralizing antibody has a remarkable virus neutralizing effect on an SARS-Cov-2 pseudo virus and provides an effective alternative antibody drug for prevention and treatment ofthe COVID-19.
Owner:NANJING MEDICAL UNIV
Antigen delivery platforms
InactiveUS20140030292A1Improve stabilityAvoid reduce stimulationSsRNA viruses positive-senseFusion with post-translational modification motifAntigen deliveryHerpes simplex virus DNA
This disclosure provides platforms for delivery of herpes virus proteins to cells, particularly proteins that form complexes in vivo. In some embodiments these proteins and the complexes they form elicit potent neutralizing antibodies. Thus, presentation of herpes virus proteins using the disclosed platforms permits the generation of broad and potent immune responses useful for vaccine development.
Owner:GLAXOSMITHKLINE BIOLOGICALS SA
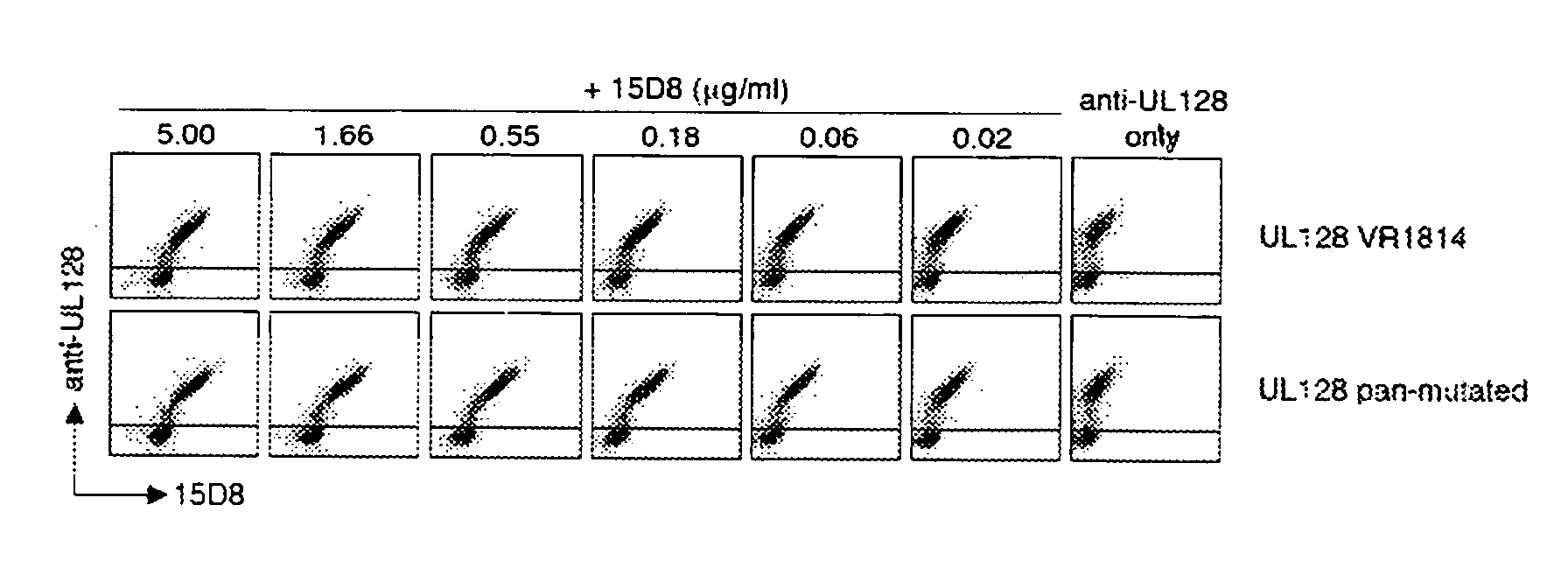
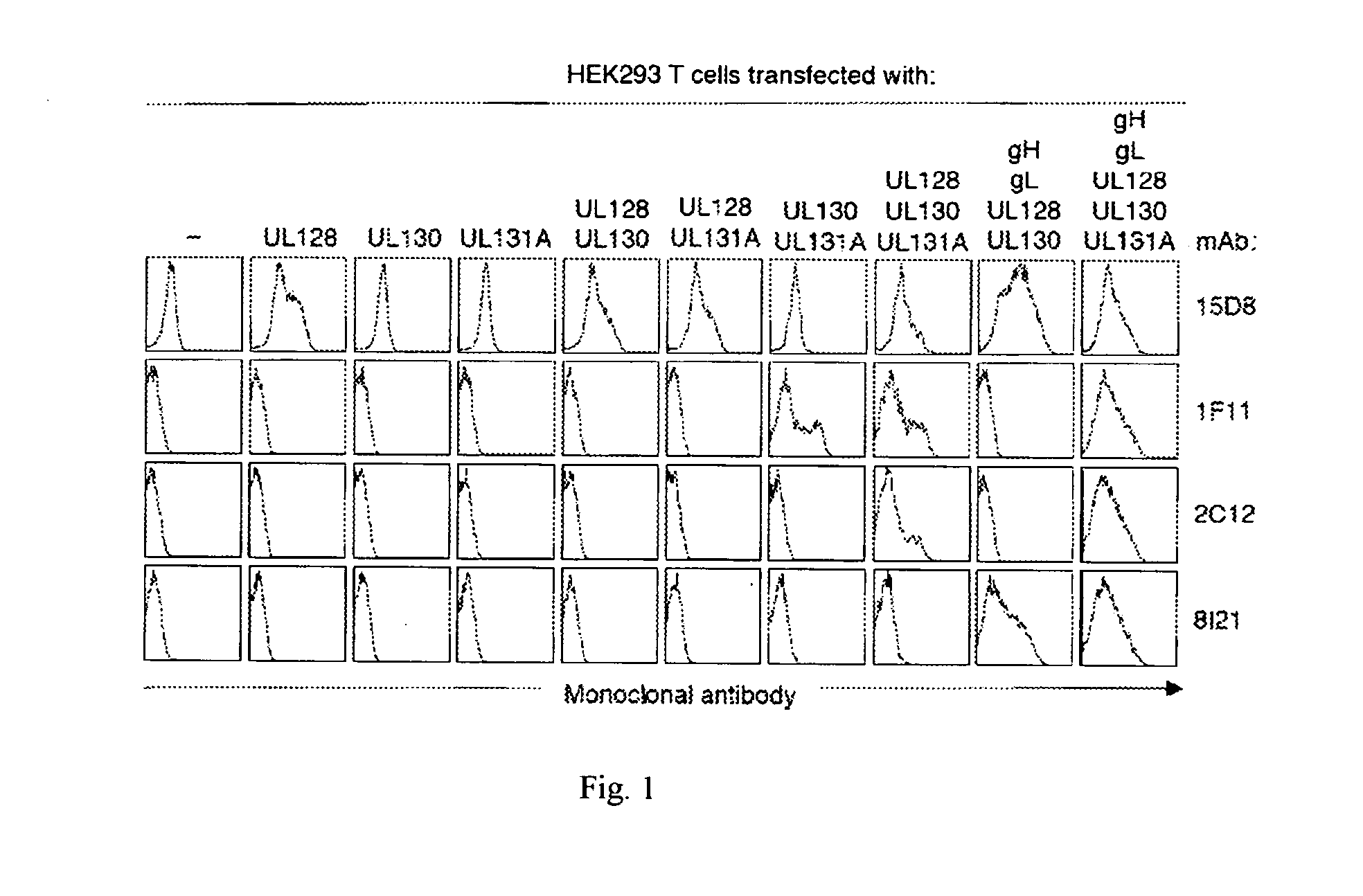
Human cytomegalovirus neutralising antibodies and use thereof
The invention relates to neutralizing antibodies, and antibody fragments thereof, having high potency in neutralizing hCMV, wherein said antibodies and antibody fragments are specific for one, or a combination of two or more, hCMV gene UL products. The invention also relates to immortalized B cells that produce, and to epitopes that bind to, such antibodies and antibody fragments. In addition, the invention relates to the use of the antibodies, antibody fragments, and epitopes in screening methods as well as in the diagnosis, prevention, and therapy of disease.
Owner:INSTITUTE FOR RESEARCH IN BIOMEDECINE
High potency recombinant antibodies and method for producing them
InactiveUS7700735B2Improve performanceIncreasing rate constantPeptide/protein ingredientsImmunoglobulins against virusesAntigenDisease
High potency antibodies, including immunologically active fragments thereof, having high kinetic association rate constants and optional high affinities are disclosed, along with methods for producing such antibodies. The high potency antibodies disclosed herein are of either the neutralizing or non-neutralizing type and have specificity for antigens displayed by microorganisms, especially viruses, as well as antigenic sites present on cancer cells and on various types of toxins, and the products of toxins. Processes for producing high potency neutralizing antibodies and increasing the potency of already existing neutralizing antibodies are also described. Methods of using said antibodies in the prevention and / or treatment of diseases, especially diseases induced or caused by viruses, are disclosed.
Owner:MEDIMMUNE LLC
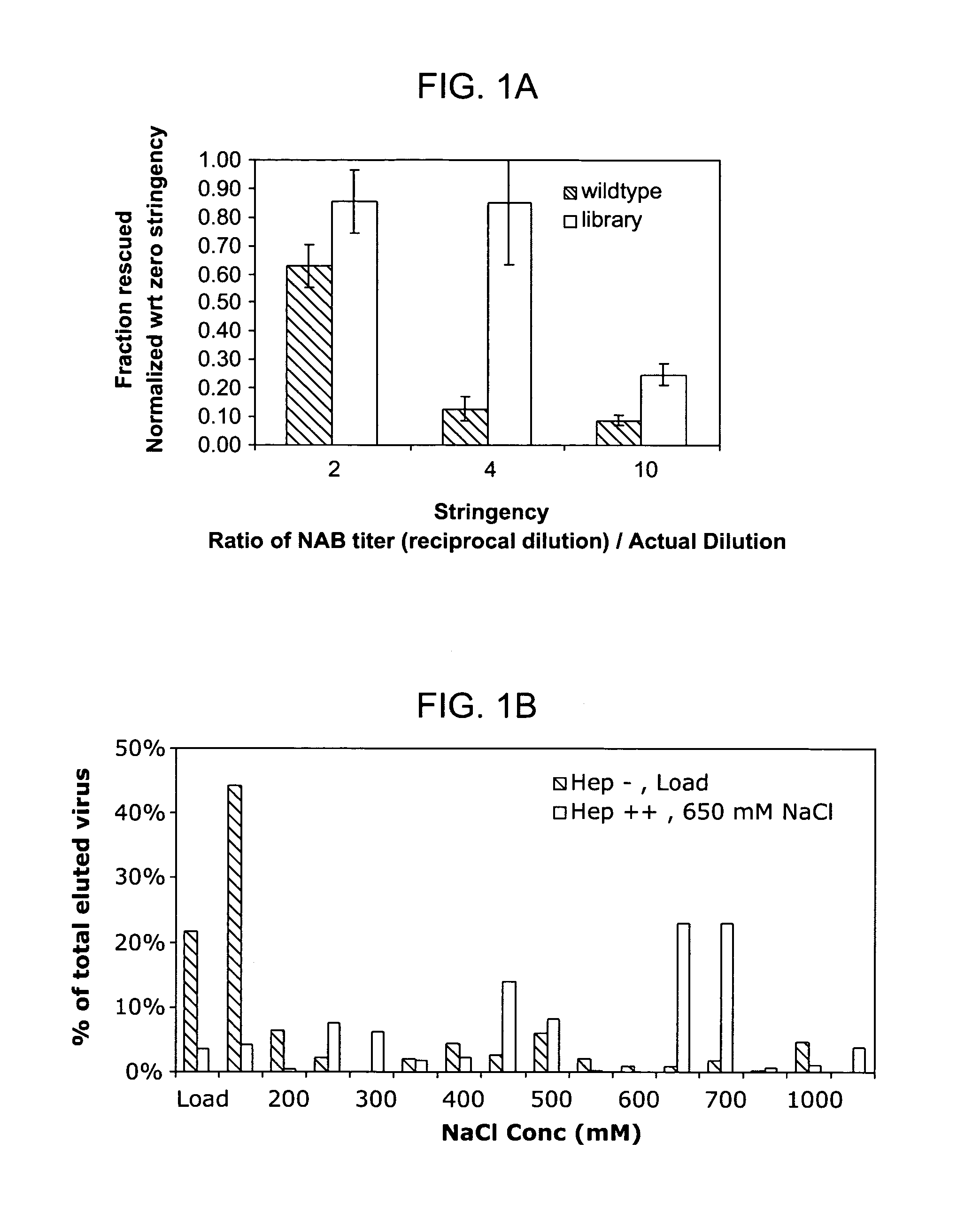
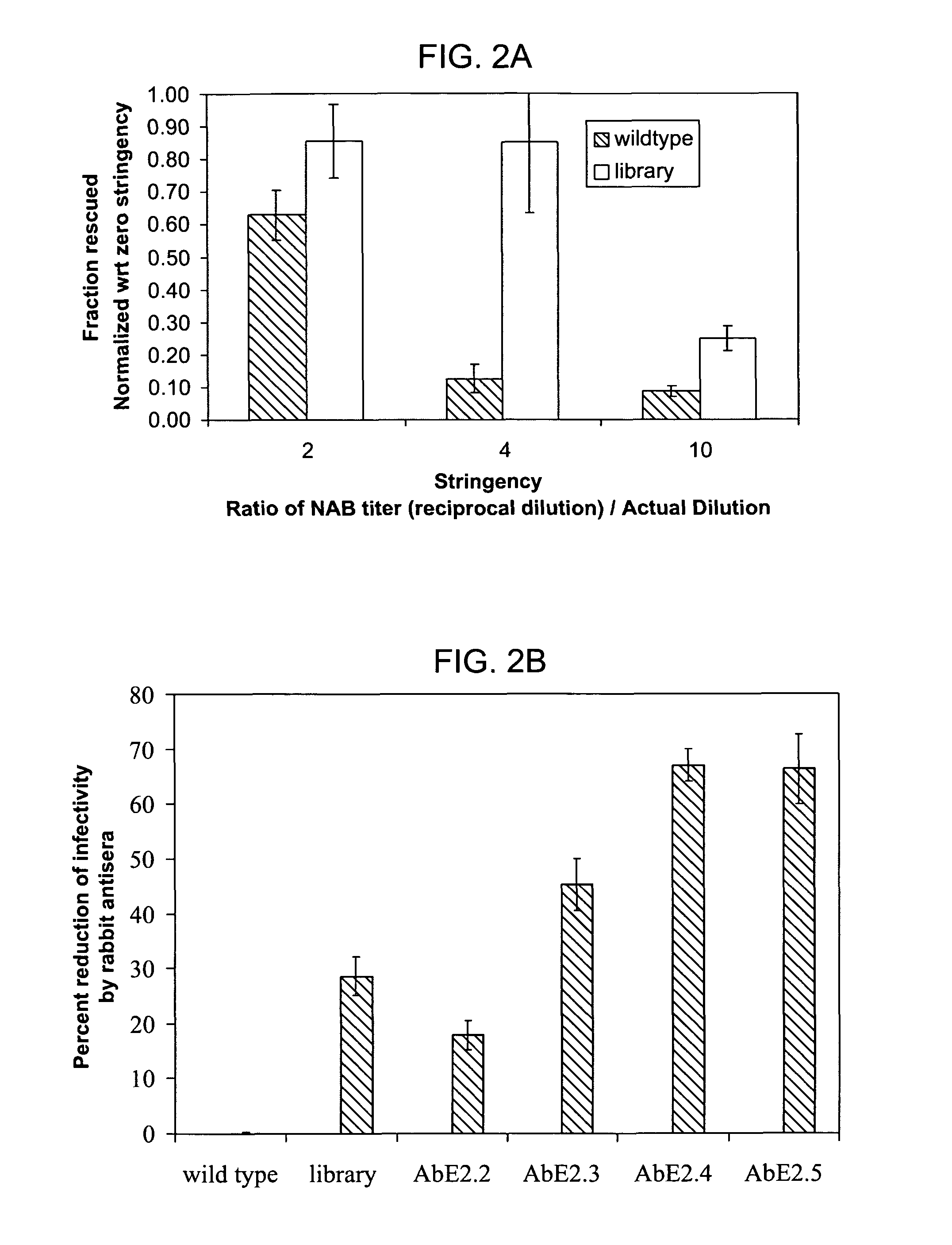
Directed evolution and in vivo panning of virus vectors
The present invention provides methods of achieving directed evolution of viruses by in vivo screening or “panning” to identify viruses comprising scrambled AAV capsids having characteristics of interest, e.g., tropism profile and / or neutralization profile (e.g., ability to evade neutralizing antibodies). The invention also provides scrambled AAV capsids and virus particles comprising the same.
Owner:THE UNIV OF NORTH CAROLINA AT CHAPEL HILL
Adeno-associated virus variants and methods of use thereof
ActiveUS20160017295A1Improve the immunityEfficient deliveryAntibacterial agentsVirusesHeterologousGene product
The present disclosure provides infectious recombinant adeno-associated virus (rAAV) virions that comprise a variant capsid protein and a heterologous nucleic acid. The present disclosure further provides the variant adeno-associated virus (AAV) capsid proteins (and / or a nucleic acid encoding the variant AAV capsid proteins), which confer to an infectious rAAV virion an increased resistance to human AAV neutralizing antibodies. The present disclosure further provides host cells comprising an infectious rAAV virion and / or a nucleic acid encoding a subject variant AAV capsid protein. The present disclosure further provides methods of delivering a heterologous nucleic acid to a target cell where the target cell is contacted with a subject infectious rAAV virion. The present disclosure further provides methods of delivering a gene product to an individual, the methods generally involving administering an effective amount of a subject rAAV virion to an individual in need thereof.
Owner:RGT UNIV OF CALIFORNIA
Human cytomegalovirus neutralising antibodies and use thereof
The invention relates to neutralizing antibodies and antibody fragments having high potency in neutralizing hCMV, wherein said antibodies and antibody fragments are specific for a combination of hCMV proteins UL130 and UL131A, or for a combination of hCMV proteins UL128, UL130 and UL131A. The invention relates also to immortalized B cells that produce, and to epitopes that bind to, such antibodies and antibody fragments. In addition, the invention relates to the use of the antibodies, antibody fragments, and epitopes in screening methods as well as in the diagnosis and therapy of disease.
Owner:HUMABS LLC
Mutant adeno-associated virus virions and methods of use thereof
ActiveUS9441244B2Altered capsid propertiesReduce the binding forceAntibacterial agentsVirusesNucleotideCell type specific
The present invention provides mutant adeno-associated virus (AAV) that exhibit altered capsid properties, e.g., reduced binding to neutralizing antibodies in serum and / or altered heparin binding and / or altered infectivity of particular cell types. The present invention further provides libraries of mutant AAV comprising one or more mutations in a capsid gene. The present invention further provides methods of generating the mutant AAV and mutant AAV libraries, and compositions comprising the mutant AAV. The present invention further provides recombinant AAV (rAAV) virions that comprise a mutant capsid protein. The present invention further provides nucleic acids comprising nucleotide sequences that encode mutant capsid proteins, and host cells comprising the nucleic acids. The present invention further provides methods of delivering a gene product to an individual, the methods generally involving administering an effective amount of a subject rAAV virion to an individual in need thereof.
Owner:INTEGRATIVE GENE THERAPEUTICS +1
Antibodies That Bind Myostatin, Compositions And Methods
There are disclosed selective myostatin antagonists (including antibodies), nucleic acids encoding them, and methods of making and using them. Neutralizing antibodies recognizing the conformational epitope near position 21 to 31 and position 50 to 60.
Owner:AMGEN INC
Directed Evolution and In Vivo Panning of Virus Vectors
The present invention provides methods of achieving directed evolution of viruses by in vivo screening or “panning” to identify viruses comprising scrambled AAV capsids having characteristics of interest, e.g., tropism profile and / or neutralization profile (e.g., ability to evade neutralizing antibodies). The invention also provides scrambled AAV capsids and virus particles comprising the same.
Owner:THE UNIV OF NORTH CAROLINA AT CHAPEL HILL
Human Anti-ngf neutralizing antibodies as selective ngf pathway inhibitors
InactiveUS20090155274A1Increase productionHigh sensitivitySenses disorderAntipyreticChronic painNeutralizing antibody
This invention provides antibodies that interact with or bind to human nerve growth factor (NGF) and neutralize the function of NGF thereby. The invention also provides pharmaceutical compositions of said antibodies and methods for neutralizing NGF function, and particularly for treating NGF-related disorders (e.g., chronic pain) by administering a pharmaceutically effective amount of anti-NGF antibodies. Methods of detecting the amount of NGF in a sample using anti-NGF antibodies are also provided.
Owner:ER SQUIBB & SONS INC
Human anti-IFN-γ neutralizing antibodies as selective IFN-γ pathway inhibitors
ActiveUS7335743B2Prevents and antagonizes inhibitionNot inhibit or modulate the biological activityNervous disorderAntipyreticNeutralizing antibodyDisease cause
This invention provides antibodies that interact with or bind to human interferon-gamma (IFN-γ) and methods for treating IFN-γ mediated diseases by administering a pharmaceutically effective amount of antibodies to IFN-γ. Methods of detecting the amount of IFN-γ in a sample using antibodies to IFN-γ are also provided.
Owner:ER SQUIBB & SONS INC
Human Anti-ngf neutralizing antibodies as selective ngf pathway inhibitors
InactiveUS20080033157A1Increase productionHigh sensitivitySenses disorderAntipyreticDiseaseChronic pain
This invention provides antibodies that interact with or bind to human nerve growth factor (NGF) and neutralize the function of NGF thereby. The invention also provides pharmaceutical compositions of said antibodies and methods for neutralizing NGF function, and particularly for treating NGF-related disorders (e.g., chronic pain) by administering a pharmaceutically effective amount of anti-NGF antibodies. Methods of detecting the amount of NGF in a sample using anti-NGF antibodies are also provided.
Owner:ER SQUIBB & SONS INC
Human anti-OPGL neutralizing antibodies as selective OPGL pathway inhibitors
Monoclonal antibodies and hybridomas producing them that interact with osteoprotegerin ligand (OPGL) are provided. Methods of treating osteopenic disorders by administering a pharmaceutically effective amount of antibodies to OPGL are also provided. Methods of detecting the amount of OPGL in a sample using antibodies to OPGL are further provided.
Owner:ER SQUIBB & SONS INC
Cytomegalovirus surface protein complex for use in vaccines and as a drug target
Owner:THE TRUSTEES FOR PRINCETON UNIV
Neutralizing Antibodies to Influenza Viruses
The present invention concerns methods and means for identifying, producing, and engineering neutralizing antibodies against influenza A viruses, and to the neutralizing antibodies produced. In particular, the invention concerns neutralizing antibodies against various influenza A virus subtypes, including neutralizing antibodies against two or more of H1, H2, H3, H5, H7 and H9, such as, for example all of H1, H2, H3, and H5 subtypes, and methods and means for making such antibodies. More specifically, the invention concerns antibodies capable of neutralizing more than one, preferably all, isolates of an influenza A virus subtype.
Owner:I2 PHARMA INC
Ultra high affinity neutralizing antibodies
InactiveUS7740851B2Low costReduce efficacyHybrid immunoglobulinsAntibody mimetics/scaffoldsDiseaseComplementarity determining region
Ultra high affinity antibodies with binding affinities in the range of 1010 M−1, and even 1011 M−1 are disclosed. Such antibodies include antibodies having novel high affinity complementarity determining regions (CDRs), especially those with framework and constant regions derived from either humans or mice. Methods of preparing and screening such antibodies, as well as methods of using them to prevent and / or treat disease, especially virus-induced diseases, are also disclosed.
Owner:MEDIMMUNE LLC
INDUCTION OF BROADLY REACTIVE NEUTRALIZING ANTIBODIES BY FOCUSING THE IMMUNE RESPONSE ON V3 EPITOPES OF THE HIV-1 gp120 ENVELOPE
InactiveUS20080279879A1Vigorous Ab responseViral antigen ingredientsAntibody mimetics/scaffoldsHeterologousNeutralizing antibody
Compositions, kits and methods for boosting, or for priming and boosting, high titer broadly neutralizing cross-clade antibody responses focused on single HIV-1 neutralizing epitopes are disclosed. gp120 DNA plasmids comprising HIV env genes are used to prime the antibody response. Primed subjects are immunized with recombinant fusion proteins that comprise a “carrier” protein fusion partner, preferably a truncated form of the MuLV gp70 Env protein, and a desired HIV neutralizing epitopes. Preferred epitopes are epitopes of V3 from one or more HIV clades. Immune sera from such immunized subjects neutralized primary isolates from virus strains heterologous to those from which the immunogens were constructed. Neutralizing activity was primarily due to V3-specific antibodies and cross-clade neutralizing Abs were present. This approach results in more potent and broader neutralizing antibody levels, a result of “immunofocusing” the humoral immune response on neutralizing epitopes such as V3.
Owner:NEW YORK UNIV
Methods and Compositions for Vaccination of Animals with Prrsv Antigens with Improved Immunogenicity
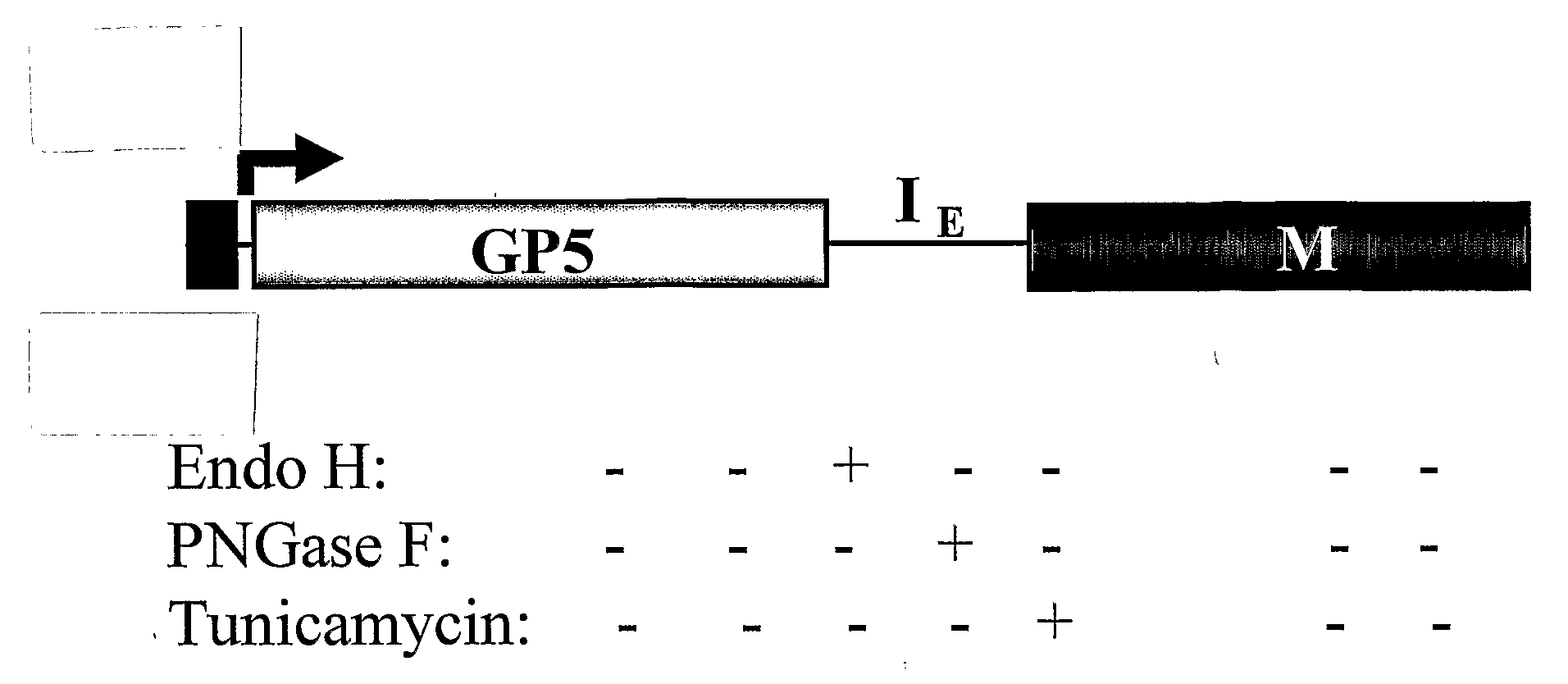
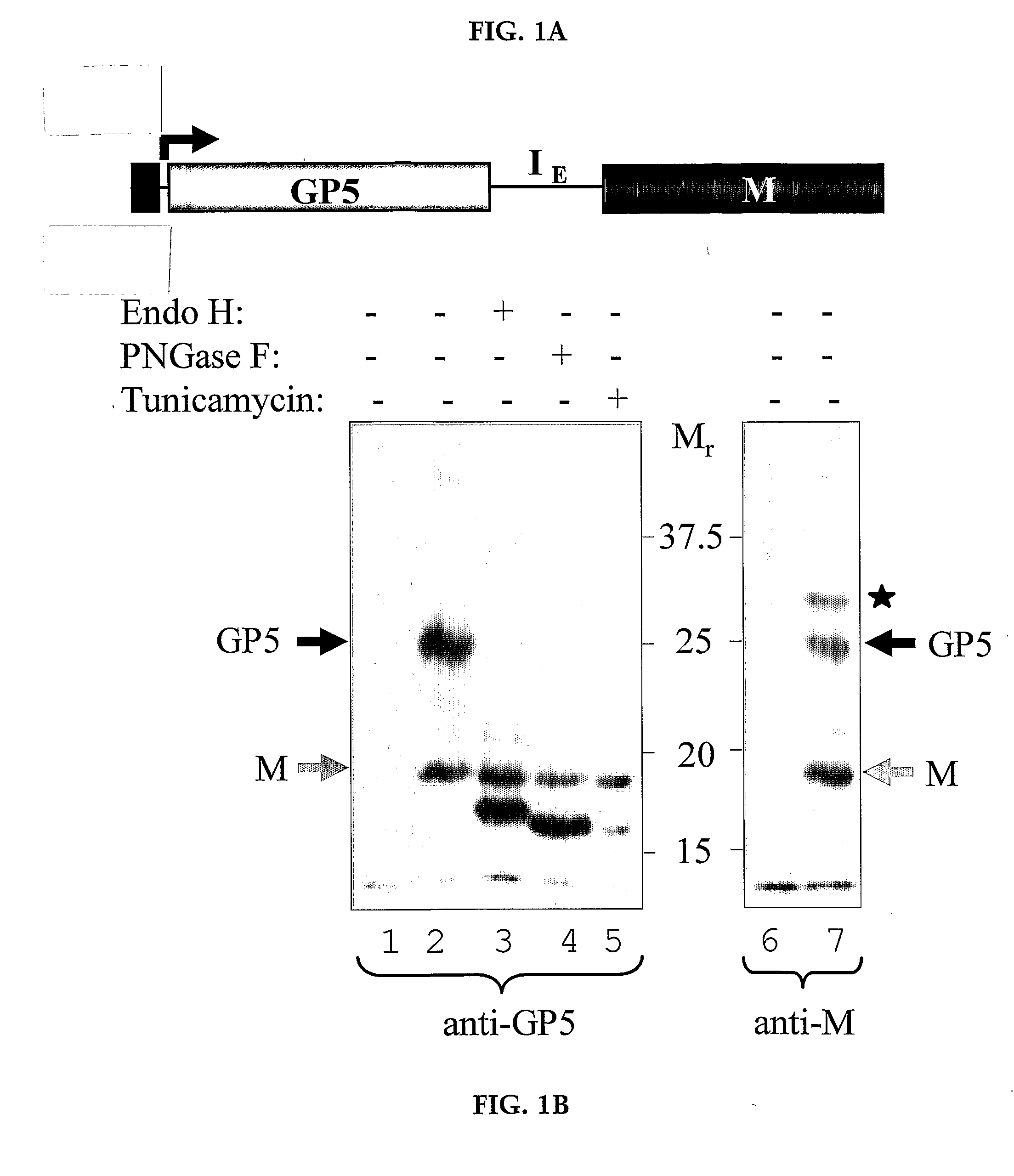
Pigs challenged with hypoglycosylated variants of Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) major surface protein GP5 exhibited increased production of PRRSV-neutralizing antibodies relative to the levels of neurtalizing antibodies produced by pigs immunized with wild type (wt) or glycosylated GP5. This invention provides for methods of obtaining improved immune responses in pigs to PRRSV, compositions useful for obtaining the improved immune responses as well as isolated polynucleotides that encode hypoglycosylated variants of PRRSV major surface protein GP5.
Owner:NUTECH VENTURES
Human Anti-NGF Neutralizing Antibodies as Selective NGF Pathway Inhibitors
InactiveUS20100034818A1Neutralize biological activityGood effectSenses disorderNervous disorderChronic painNeutralizing antibody
This invention provides antibodies that interact with or bind to human nerve growth factor (NGF) and neutralize the function of NGF thereby. The invention also provides pharmaceutical compositions of said antibodies and methods for neutralizing NGF function, and particularly for treating NGF-related disorders (e.g., chronic pain) by administering a pharmaceutically effective amount of anti-NGF antibodies. Methods of detecting the amount of NGF in a sample using anti-NGF antibodies are also provided.
Owner:AMGEN INC +1
Neutralizing antibodies to influenza viruses
The present invention concerns methods and means for identifying, producing, and engineering neutralizing molecules against influenza A viruses, and to the neutralizing molecules produced. In particular, the invention concerns neutralizing molecules against various influenza A virus subtypes, including neutralizing antibodies against H5 and / or H3 and / or H1, such as, for example all of H1, H3, and H5 subtypes, and methods and means for making such molecules.
Owner:I2 PHARMA INC
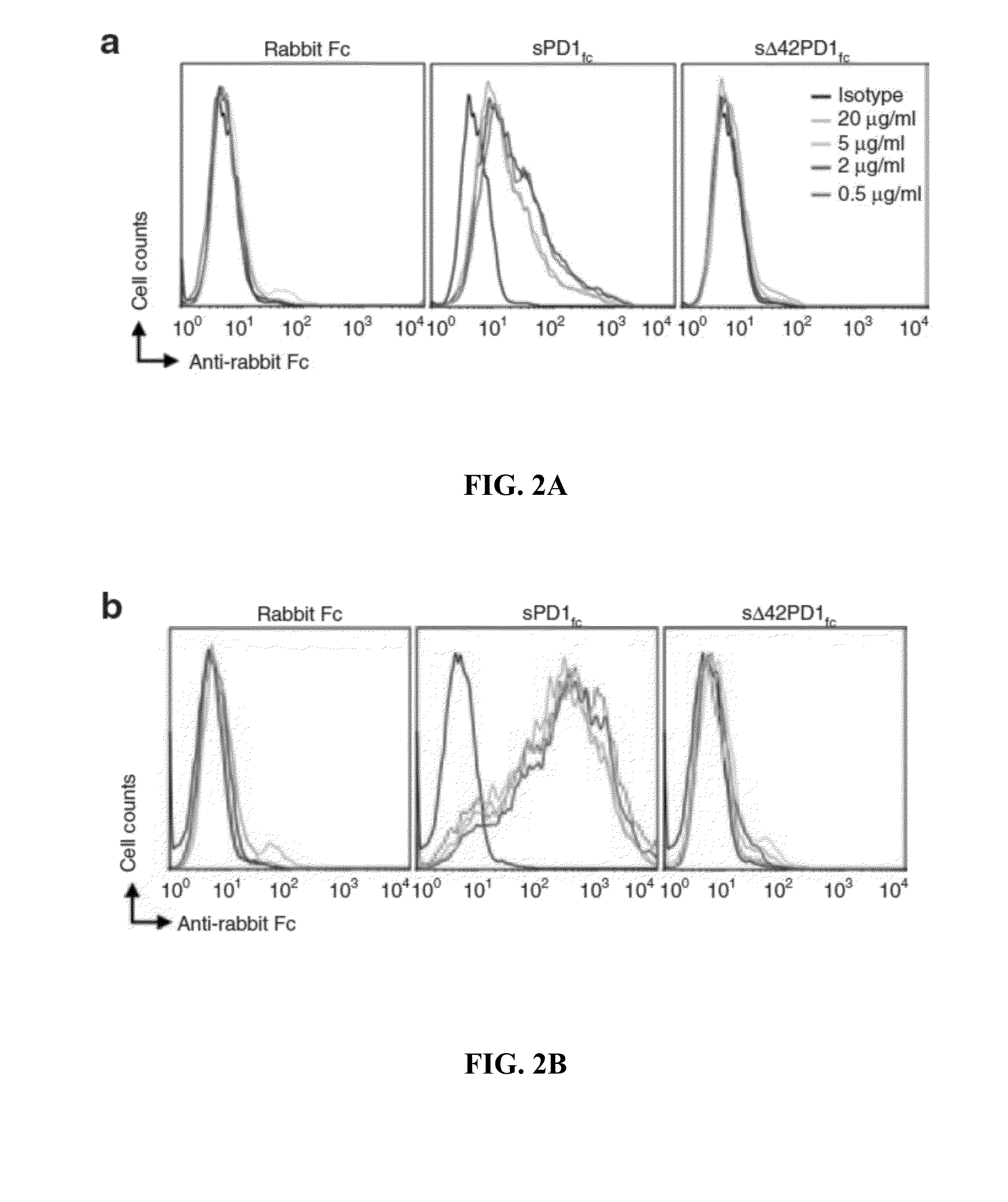
Novel pd1 isoforms, and uses thereof for potentiating immune responses
ActiveUS20140302070A1Prevention treatment ameliorationPeptide/protein ingredientsGenetic material ingredientsAutoimmune conditionAdjuvant
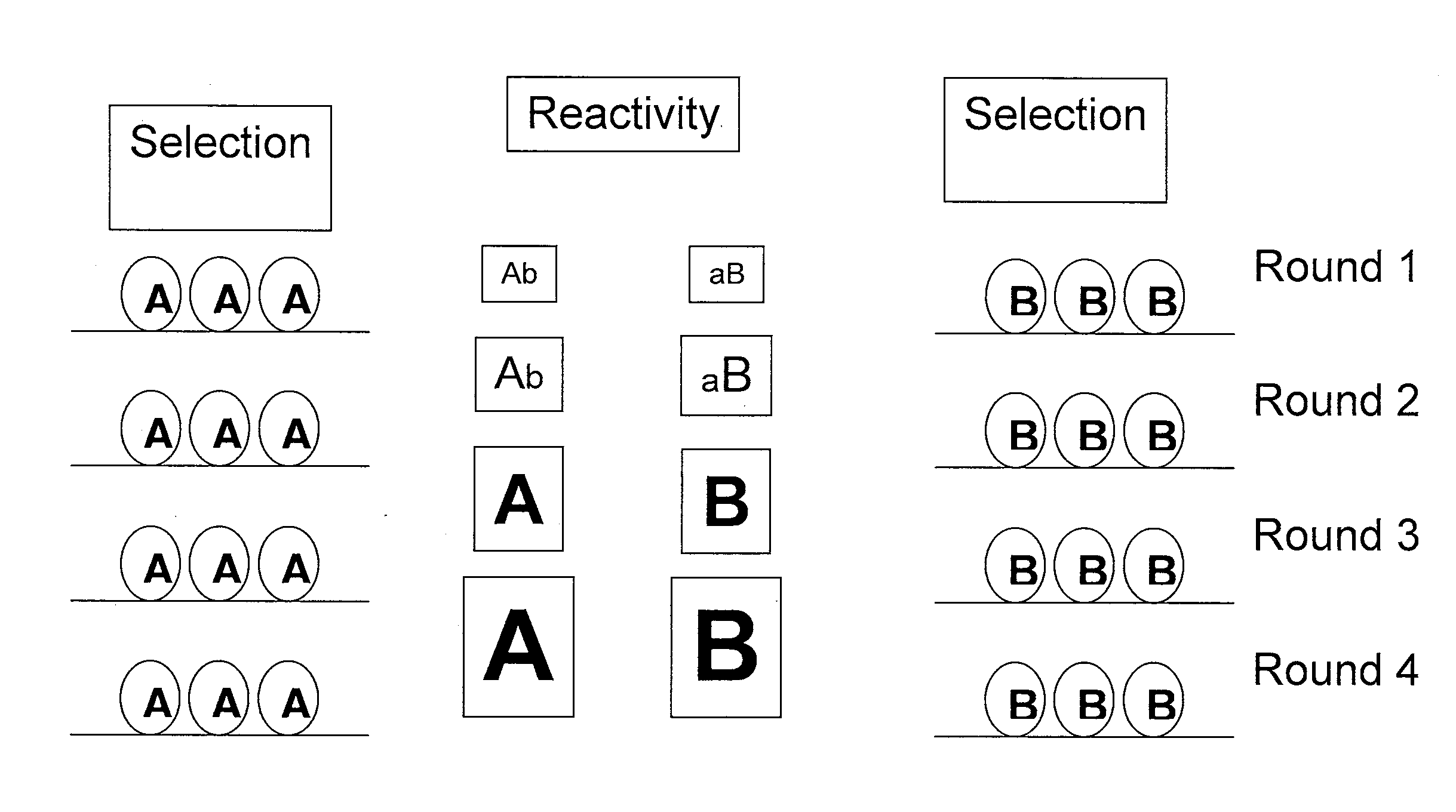
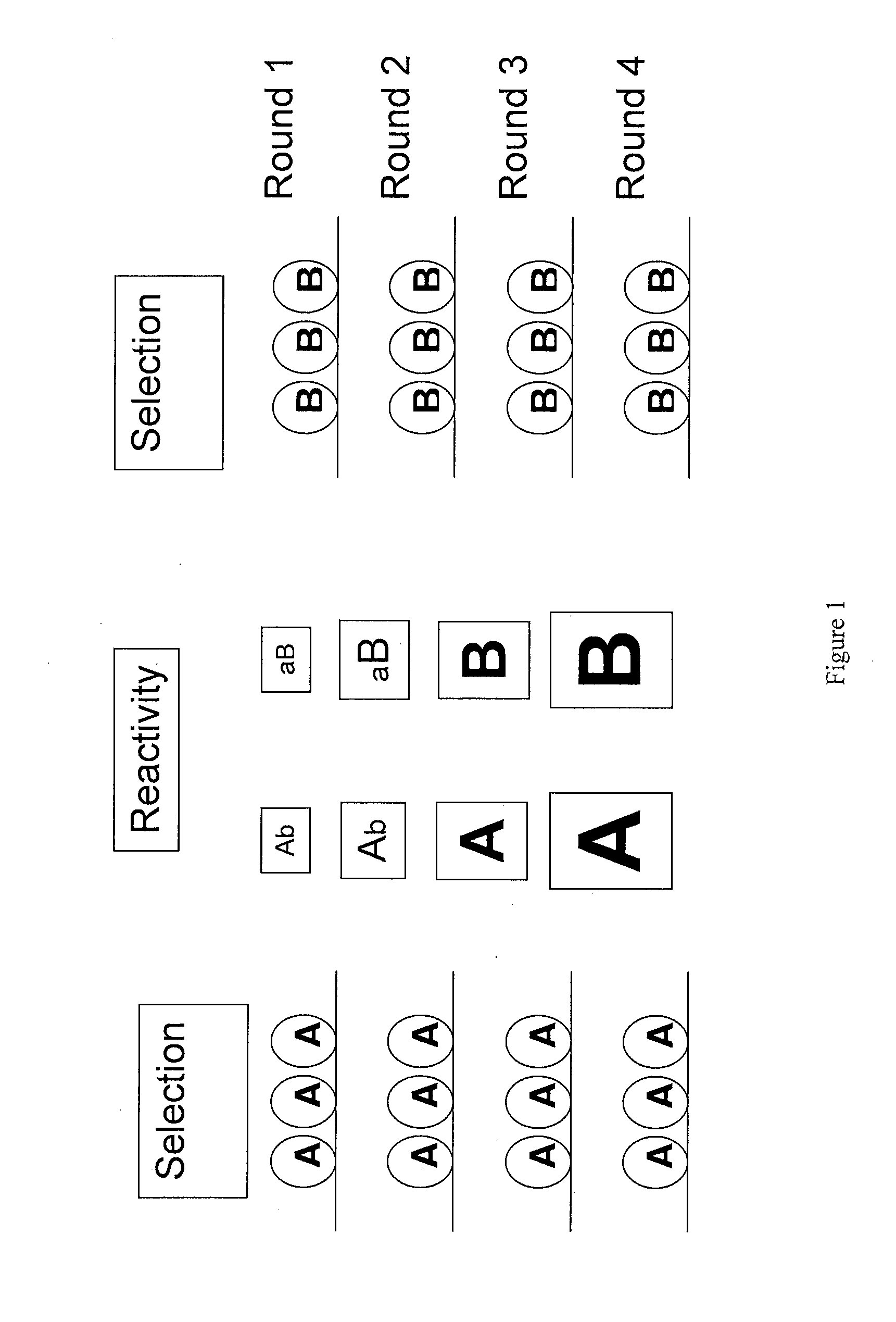
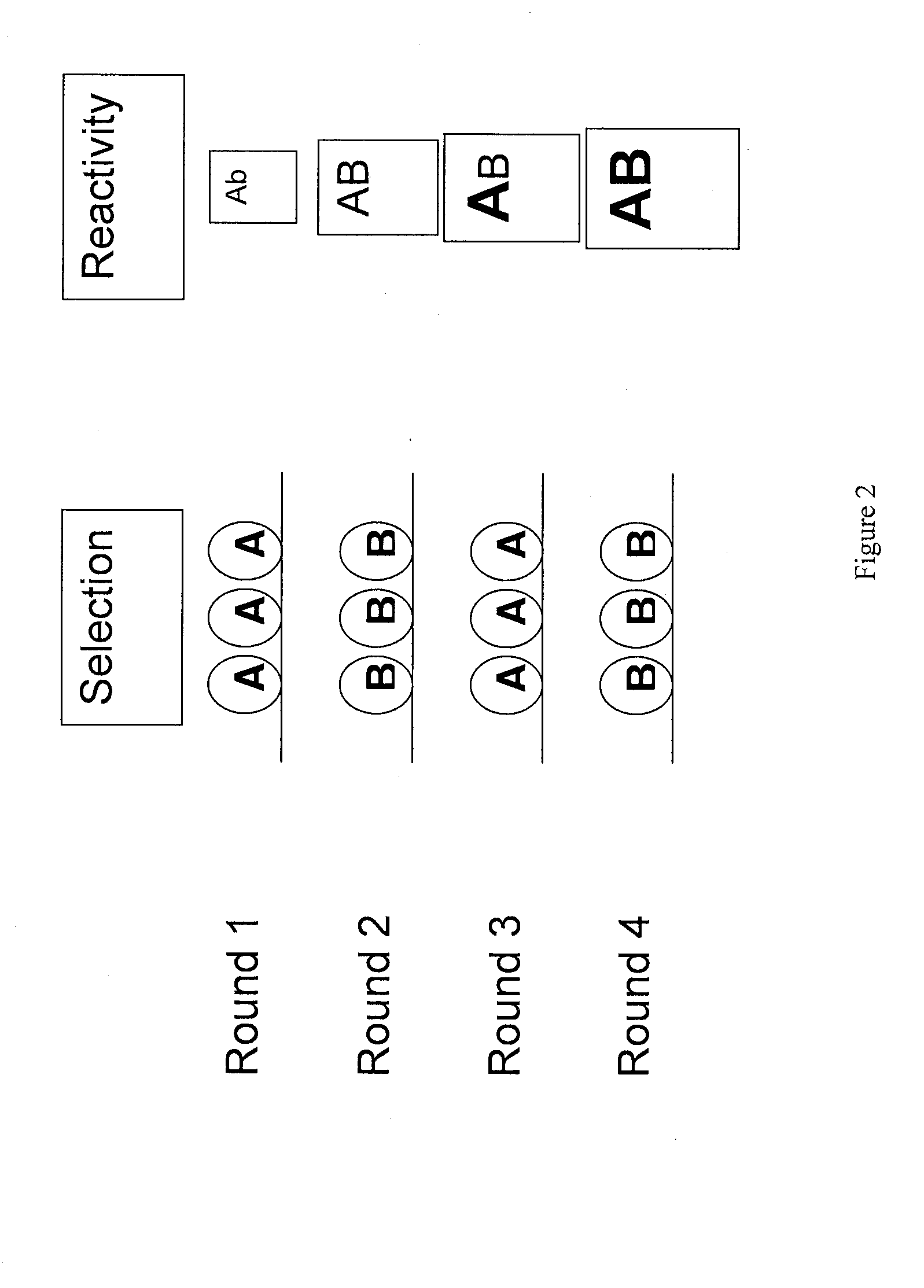
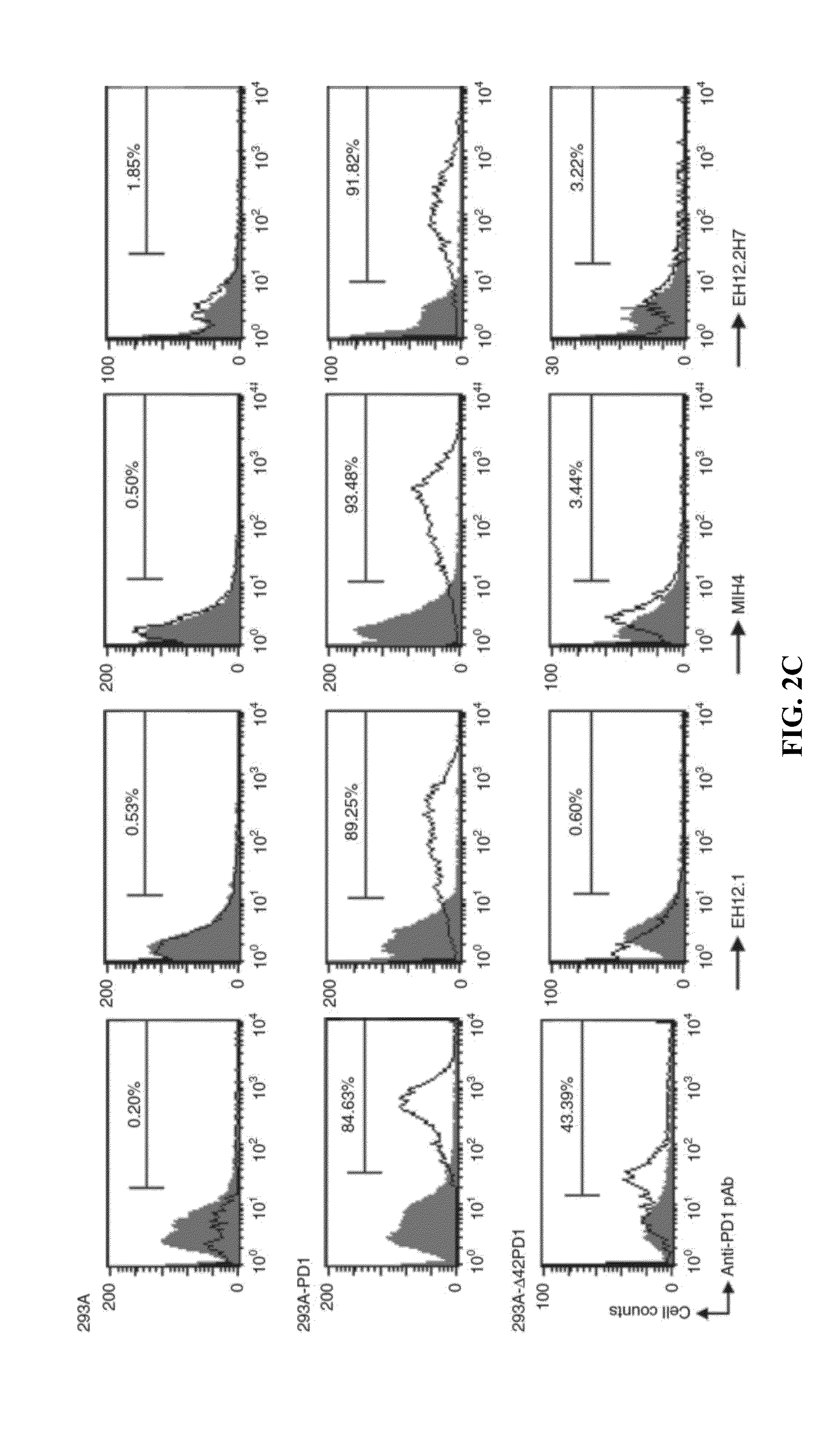
In one embodiment, the present invention provides a new isoform of human PD1 (Δ42PD1) that contains a 42-nucleotide in-frame deletion located at exon 2 domain. Δ42PD1 does not engage PD-L1 / PD-L2, and can induce the production of pro-inflammatory cytokines In one embodiment, Δ42PD1 can be used as an intramolecular adjuvant to develop a fusion DNA vaccine for enhancing antigen-specific CD8+ T cell immunity and for prevention of pathogenic infection and / or cancer. In one embodiment, soluble Δ42PD1 protein could be a therapeutic target for autoimmune diseases. In other embodiments, proteins or peptides or nucleic acids encoding proteins or peptides containing Δ42PD1 could be used as immunogens for developing antibodies binding specifically to Δ42PD1. In yet another embodiment, neutralizing antibodies could block sΔ42PD1 function and accordingly could be used as treatment for autoimmune disorders.
Owner:VERSITECH LTD
Novel HIV-1 envelope glycoprotein
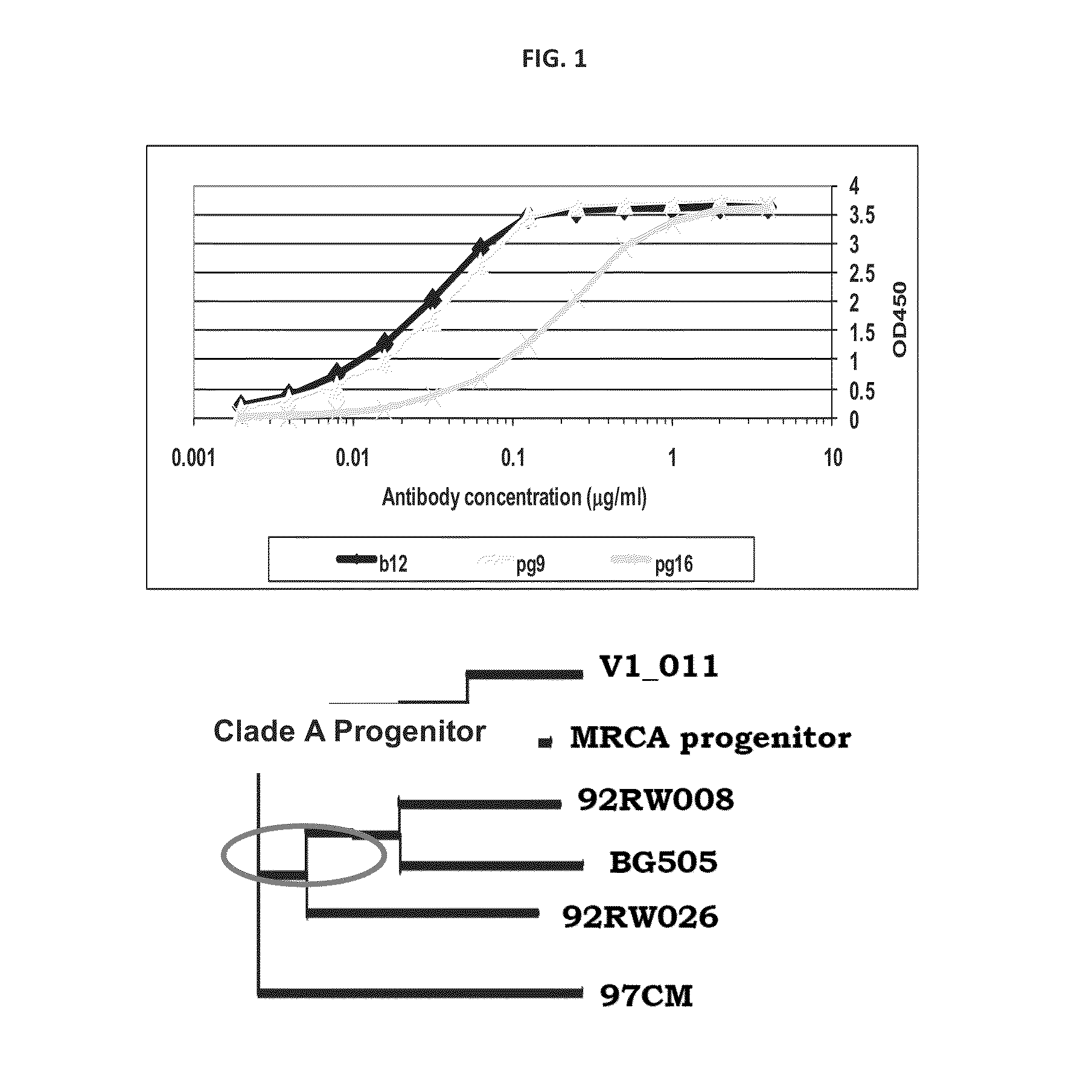
The present application relates to novel HIV-1 envelope glycoproteins, which may be utilized as HIV-1 vaccine immunogens, and antigens for crystallization, electron micrsocopy and other biophysical, biochemical and immunological studies for the identification of broad neutralizing antibodies. The present invention encompasses the preparation and purification of immunogenic compositions, which are formulated into the vaccines of the present invention.
Owner:THE SCRIPPS RES INST +2
Immune globulin formulations for the treatment and prevention of an orthopoxvirus infection
InactiveUS20060110407A1Serum immunoglobulinsMicrobiological testing/measurementGenus OrthopoxvirusIntravenous IG
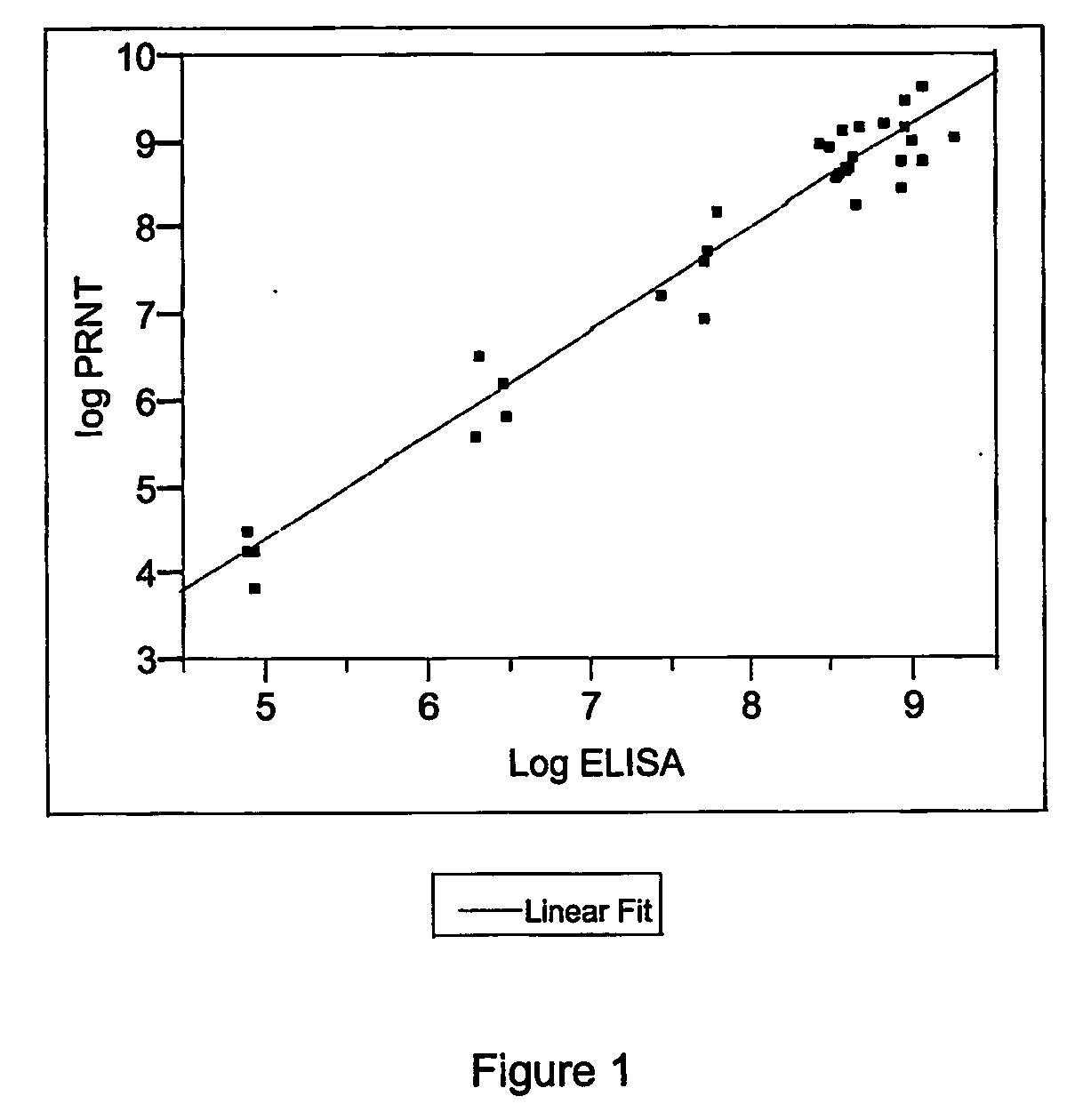
The invention provides an immune globulin having a high titre of antibody to Orthopoxvirus, pharmaceutical compositions comprising the immune globulin and methods for making same. In one embodiment the immune globulin is intravenously injectable. The invention also provides a colorimetric assay to detect neutralizing antibodies to vaccinia virus. The invention has a number of uses including detection of neutralizing antibodies to vaccinia virus and the immunization of persons against the Orthopoxvirus and in the treatment of related conditions.
Owner:CANGENE CORP
Synthetic human papillomavirus genes
InactiveUS7001995B1Easy to insertEasy to removeBiocideGenetic material ingredientsPolynucleotide VaccinesHuman papillomavirus
Synthetic DNA molecules encoding papillomavirus proteins are provided. The codons of the synthetic molecules are codons preferred by the projected host cell. The synthetic molecules may be used as a polynucleotide vaccine which provides effective immunoprophylaxis against papillomavirus infection through stimulation of neutralizing antibody and cell-mediated immunity.
Owner:MERCK SHARP & DOHME CORP