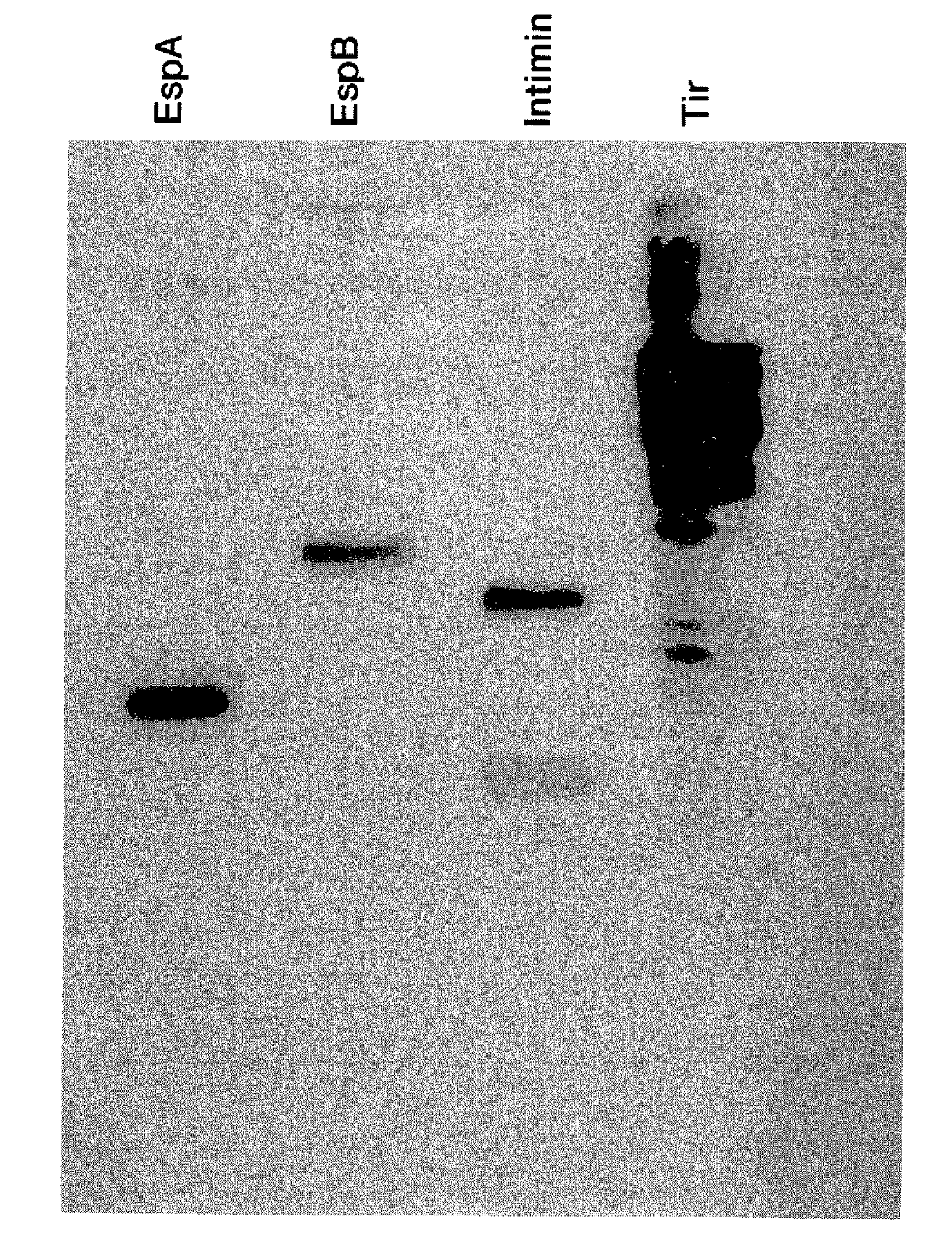Enterohemorrhagic escherichia coli vaccine
a technology of escherichia coli and vaccine, which is applied in the direction of antibacterial agents, immunological disorders, antibody medical ingredients, etc., can solve the problems of difficult and expensive production and purification of recombinant proteins in sufficient quantities for use as antigens, and prevent ehec infection. , the effect of reducing the colonization of ehec in the mammal is easy and relatively inexpensive to prepar
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of Cell Culture Supernatant (CCS)
[0103]Wild type EHEC O157:H7 were grown under conditions to maximize the synthesis of CCS proteins (Li et al., Infect. Immun. (2000) 68:5090). Briefly, an overnight standing culture of EHEC O157:H7 was grown in Luria-Bertani (LB) medium overnight at 37° C. (±5% CO2). The culture was diluted 1:10 in M-9 minimal medium supplemented with 0.1% Casamino Acids, 0.4% glucose, 8 mM MgSO4 and 44 mM NaHCO3. Cultures were grown standing at 37° C. in 5% CO2 to an optical density at 600 nm of 0.7 to 0.8 (6-8 h). Bacteria were removed by centrifugation at 8000 rpm for 20 min at 4° C. The supernatant was concentrated 100 fold by ultrafiltration and total protein was determined by the bicinchoninic acid protein assay method.
[0104]FIG. 1 shows molecular weight markers (lane 1) and a typical CCS protein profile obtained by electrophoresis of CCS in a SDS-10% polyacrylamide gel (SDS-PAGE) followed by Coomassie blue staining (lane 2). The positions of EspA (...
example 2
Preparation of Recombinant Proteins
[0105]The genes coding for EspA, EspB, Intimin and Tir were isolated (Li et al., Infect. Immun. (2000) 68:5090). A clinical isolate of EHEC O157:H7 was used as the source of DNA. EspA, EspB, Tir, and the region of eae encoding the 280 carboxyl-terminal amino acids of Intimin were amplified from chromosomal DNA using PCR to introduce unique restriction sites, followed by cloning into appropriate plasmids. The resulting plasmids were cleaved and ligated to create histidine-tagged fusions. Plasmids were electrocuted into an expression strain of E. coli and the E. coli were propagated (Ngeleka et al., Infect. Immun. (1996) 64:3118). Gene expression was driven using the Tac promoter following IPTG (isopropyl-β-D-thiogalactopyranoside) induction. Bacteria were pelleted, resuspended in Tris-buffered saline and lysed by sonication. The lysate was centrifuged to remove insoluble material and the histidine-tagged proteins were purified by passage through a s...
example 3
Vaccine Formulation and Delivery
[0107]Vaccines were formulated by mixing CCS or rEspA+rTir in 2 ml of a carrier containing from 30 to 40% of an adjuvant. Vaccines were delivered subcutaneously. Animals were immunized on day 1 and again at a 3-4 week intervals (boost). Serum samples were obtained prior to the first immunization, at the time of each boost and at the end of the experiment.
[0108]The serological response to immunization was determined using an enzyme-linked immunosorbent assay (ELISA). One hundred p1 of rEspA (0.16 μg / well), rTir (0.1 μg / well), rEspB (0.24 μg / well) and rIntimin (0.187 μg / well) were used to coat the wells in microtiter plates and the plates were incubated overnight at 4° C. The wells were washed 3×, blocked with 0.5% nonfat dried milk in phosphate-buffered saline. Serial dilutions of sera were added to each well and incubated for 2 h at 37° C. The wells were washed and blocked and 100 μl of peroxidaseconjugated rabbit anti-bovine immunoglobulin G antibodi...
PUM
| Property | Measurement | Unit |
|---|---|---|
| optical density | aaaaa | aaaaa |
| v/v | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More