Method for stabilization of proteins using non-natural amino acids
a technology of amino acids and proteins, applied in the field of improved stabilization of polypeptides, can solve the problems of loss of coiled coil stability, modest and unpredictable gains in stability, limited protein engineering scope, etc., and achieve rapid and predictable approach, increased stability, and high stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
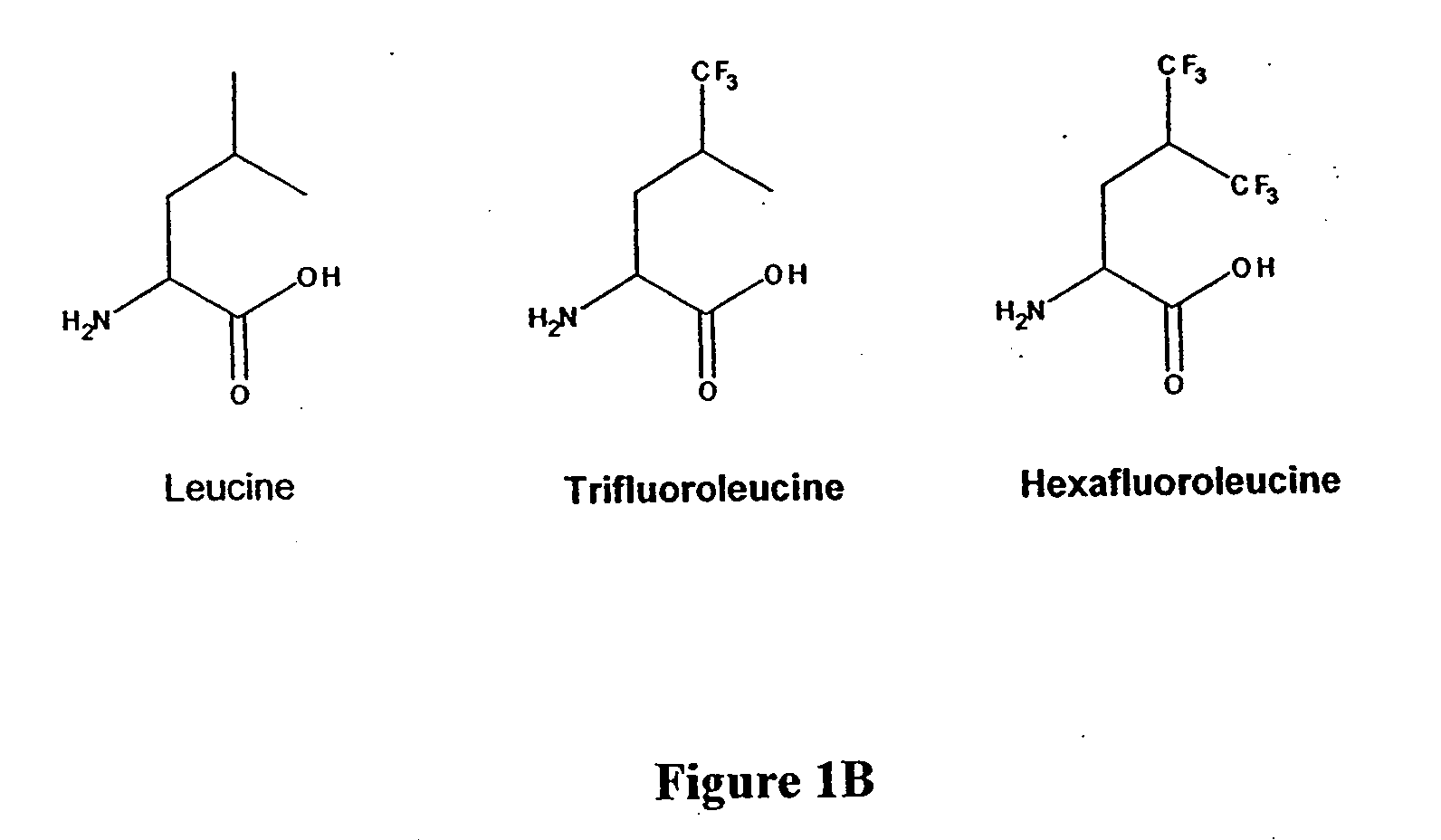
[0055]The following Example provides a description of how the non-natural amino acid analogs trifluoroleucine (Tfl) and hexafluoroleucine (Hfl) were prepared.
[0056]Trifluoroleucine (Tfl, FIG. 1B) was synthesized in an overall yield of 22% in seven steps starting from β-trifluoromethylcrotonic acid (Oakwood Chemical, Columbia, S.C.), according to the procedure of Rennert et al. with slight modifications (Rennert, O. M.; Anker, H. S. Biochemistry 1963, 2, 471). DL-trifluoroleucine prepared by this method as the N-acetylated racemic mixture was resolved to L-trifluoroleucine by treatment with porcine kidney acylase (Sigma) to >99% enantiomeric excess (e.e). (Chenault et al. J. Am. Chem. Soc. 111, 6354-6364, 1989). The yield for the resolution was 67%. The determination of e.e. was accomplished by 1H NMR spectroscopy following derivatization with Mosher's acid, R-(+)-methoxytrifluoromethylphenylacetic acid.
[0057]Hexafluoroleucine (Hfl, FIG. 1B) was prepared by modification of the proced...
example 2
[0058]The following Example provides a description of the chemical synthesis of “wild type” (Leu-GCN4-p1) and trifluoleucine incorporated (Tfl-GCN4-p1) forms of the leucine zipper peptide Leu-GCN4-p1.
[0059]The amino acid sequence of GCN4-p1 is shown in FIG. 1A. Both the “wild type” (Leu-GCN4-p1) and fluorinated (Tfl-GCN4-p1) forms of the leucine zipper peptide GCN4-p1 were synthesized at the Biopolymer Synthesis Center at the California Institute of Technology (Pasadena, Calif. 91125). Automated, stepwise solid-phase synthesis was performed on an ABI 433A synthesizer employing Fmoc chemistry. To prepare the fluorinated peptide (Tfl-GCN4-p1), N-Fmoc-5,5,5-trifluoro-L-leucine prepared as described in Example 1 was used as an equimolar mixture of the 2S,4S- and the 2S,4R-isomers, and incorporated into the peptide with extended coupling cycles. After chain assembly was complete, the peptide was deprotected and removed from the resin support with trifluoroacetic acid in the presence of 1...
example 3
[0060]The following Example provides a description of the procedure used to express in E. coli cells, the “wild type” proteins and the corresponding “artificial” proteins of the invention, in which the leucine residues are replaced with a non-natural fluorinated amino acid.
[0061]Analog Incorporation Assay. The expression vector pQE-A1, which contains the coding sequences for the protein A1 (FIG. 4A) was obtained from US Army Natick RD&E Center (Natick, Mass.). The E. coli leucine auxotroph SG13009 was obtained from Qiagen (Chatsworth, Calif.) and transformed with plasmids pREP4 and pQE-A1, to yield the expression host LAE-A 1.
[0062]M9AA medium (30 ml) supplemented with 1 mM MgSO4, 1 mM CaCl2, 20 wt % glucose, 1 mg / L thiamin and the antibiotics ampicillin (200 mg / L) and kanamycin (25 mg / L) were inoculated with 1 ml of an overnight 2xYT culture of the expression strain. After the culture had grown to an OD600 of 1.0 at 37° C., the cells were collected by centrifugation at 5,000 g for ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| enantiomeric excess | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More