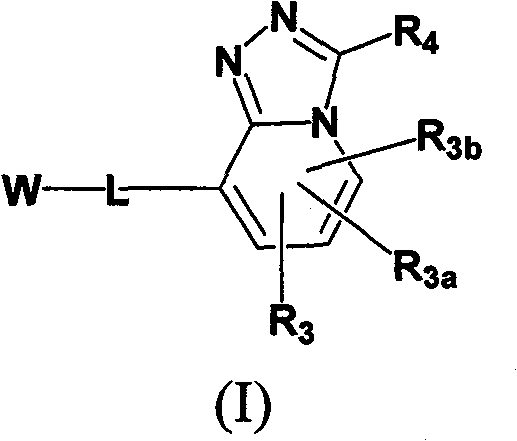
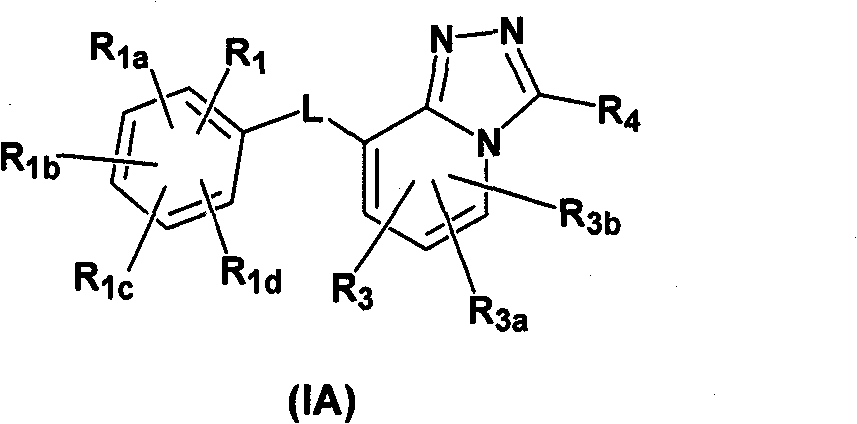
Imidazo- and triazolopyridines as inhibitors of 11-beta hydroxysteroid dehyftogenase type I
一种苯基、基团的技术,应用在作为I型11-β-羟基类固醇脱氢酶抑制剂的咪唑并吡啶和三唑并吡啶领域,能够解决血浆葡萄糖水平低等问题
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0339] 3-cycloheptyl-8-((2,6-dichlorophenoxy)methyl)-[1,2,4]triazolo[4,3-a]pyridine
[0340]
[0341] Compound 1A.1-(3-((tert-butyldimethylsilyloxy)methyl)pyridin-2-yl)hydrazine
[0342]
[0343] Add imidazole (2.4 g, 35.3 mmol) and tert-butyldimethylsilyl chloride (4.3 g, 28.5 mmol) to (2-chloropyridin-3-yl)methanol (3.4 g, 23.7 mmol) at RT 50 mL of dichloromethane solution. The mixture was stirred at RT for 60 min, then diluted with 100 mL of hexane. The white solid was filtered off, and the filtrate was concentrated under reduced pressure. Additional solids were removed by trituration with 5% ethyl acetate in hexanes to give a light yellow oil. The light yellow oil was dissolved in 40 mL of dioxane, then hydrazine (7.5 mL, 238.7 mmol) was added. The resulting mixture was heated to reflux for 36h. After this time, the mixture was cooled to RT and the solvent was removed in vacuo to give a residue. The residue was diluted with ethyl acetate, washed with water, was...
Embodiment 2
[0356] 3-cycloheptyl-8-((2,6-dichlorophenylthio)methyl)-[1,2,4]triazolo[4,3-a]pyridine
[0357]
[0358] A solution of compound 1D (140 mg, 0.57 mmol) in 15 mL of dichloromethane was washed with SOCl at RT 2 (0.166 mL, 2.28 mmol) was treated. The reaction mixture was stirred at RT for 2 h. After this time, the solvent was evaporated under reduced pressure to obtain a white powder. The white powder was suspended in 20 mL of dichloromethane, treated with DIEA (0.478 mL, 2.85 mmol) then 2,6-dichlorothiophenol (0.206 g, 1.15 mmol) at RT, then stirred at RT for 2 h. The resulting mixture was concentrated and purified by silica gel chromatography (20-50% ethyl acetate in hexanes) to afford Example 2 (212 mg, 91%) as a colorless oil. HPLCR t (Method A): 3.44 min. LC / MS(m / z)=406(M+H) + . 1 H NMR: δ7.69(d, J=7Hz, 1H), 7.25(d, J=8Hz, 2H), 7.08(t, J=8Hz, 1H), 6.65(d, J=7Hz, 1H), 6.53 (t, J=7Hz, 1H), 4.40(s, 2H), 3.12-3.24(m, 1H), 1.92-2.10(m, 4H), 1.72-1.89(m, 2H), 1.48-1.72(...
Embodiment 3
[0360] 3-cycloheptyl-8-((2,6-dichlorophenylsulfonyl)methyl)-[1,2,4]triazolo[4,3-a]pyridine
[0361]
[0362] A solution of Example 2 (110 mg, 0.271 mmol) in 20 mL of dichloromethane was treated with mCPBA (390 mg, 1.35 mmol) for 4 h at RT. After this time, the reaction mixture was analyzed by LCMS, indicating the presence of sulfoxide. Additional mCPBA (156 mg, 0.542 mmol) was added. After the addition was complete, the reaction mixture was stirred for an additional 2 h. After this time, the reaction mixture was diluted with dichloromethane, washed with 1N NaOH, brine and water, washed with MgSO 4 Drying and concentration gave crude product. The crude product was purified by silica gel chromatography (50% ethyl acetate in hexanes) to afford Example 3 (57.5 mg, 48%) as a light brown thick oil. HPLCR t (Method A): 2.99 min. LC / MS(m / z)=438(M+H) + . 1 H NMR: δ7.78 (dd, J=1, 7Hz, 1H), 7.39 (dd, J=1, 7Hz, 1H), 7.19-7.29 (m, 3H), 6.78 (t, J=7Hz, 1H) , 5.08 (s, 2H), 3.07-3...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More