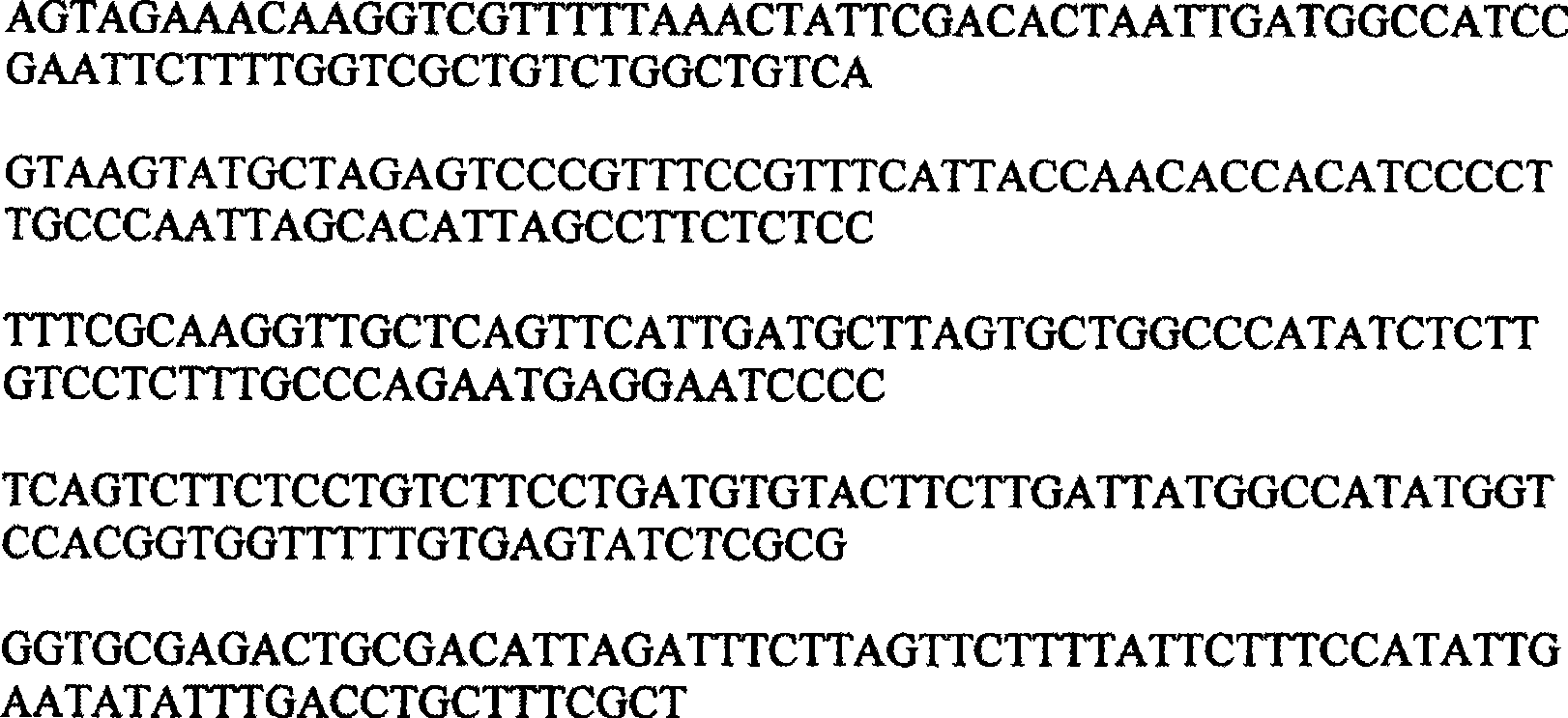Defective interfering virus
A technology of viruses and virions, applied in virology and virus infection in animals, prevention and/or treatment of influenza A, interference with defective viruses, can solve the problem of low yield of DI virus
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0190] Preparation of cloned DI virus
[0191] The methods used in the specific examples are described below and are subject to variation, see also the materials and methods described in Duhaut & Dimmock (2003), supra.
[0192] HEK293T cells were transfected by known methods with the set of plasmids necessary for the preparation of infectious A / PR8 / 34 virus (H1N1).
[0193] The transfected cells are then co-cultured with MDCK cells to amplify infectious virus. Tissue culture fluid containing the infectious virus and any DI virus present was collected, centrifuged at low speed to remove debris, and stored at -70°C. The presence of virus was shown by agglutination of chicken erythrocytes (see below). Virus titers were recorded as hemagglutination units (HAU) per milliliter of virus.
[0194] Tissue culture fluid (500 μl) was then injected into the allantoic cavity of 10-day-old embryonated chicken eggs to boost the concentration of infectious virus and infer the presence of D...
Embodiment 1
[0255] Example 1 - Production of cloned DI virus 220 / PR8
[0256] Previous work known in the art teaches that production of DI virus is optimal when inoculating embryonated chicken eggs with a large inoculum (eg, up to 2 ml per egg). The explanation given is that cells inhabited by non-infectious DI virus also need to receive infectious (helper) virus so that the former can replicate. Furthermore, the presence of DI virus normally increased when passage was repeated two or three times. Eventually, the amount of DI virus produced exceeds the amount of infectious virus present, and overall virus production declines due to the lack of helper virus function.
[0257] Unexpectedly, the inventors found that inoculation of embryonated chicken eggs with routine, i.e., standard amounts of cloned influenza A virus, failed to yield the expected amount of DI virus material; the gap was so large that it affected further laboratory studies, Including in vivo animal studies; not to menti...
Embodiment 2
[0280] Example 2 - Production of protective virus 244 / PR8
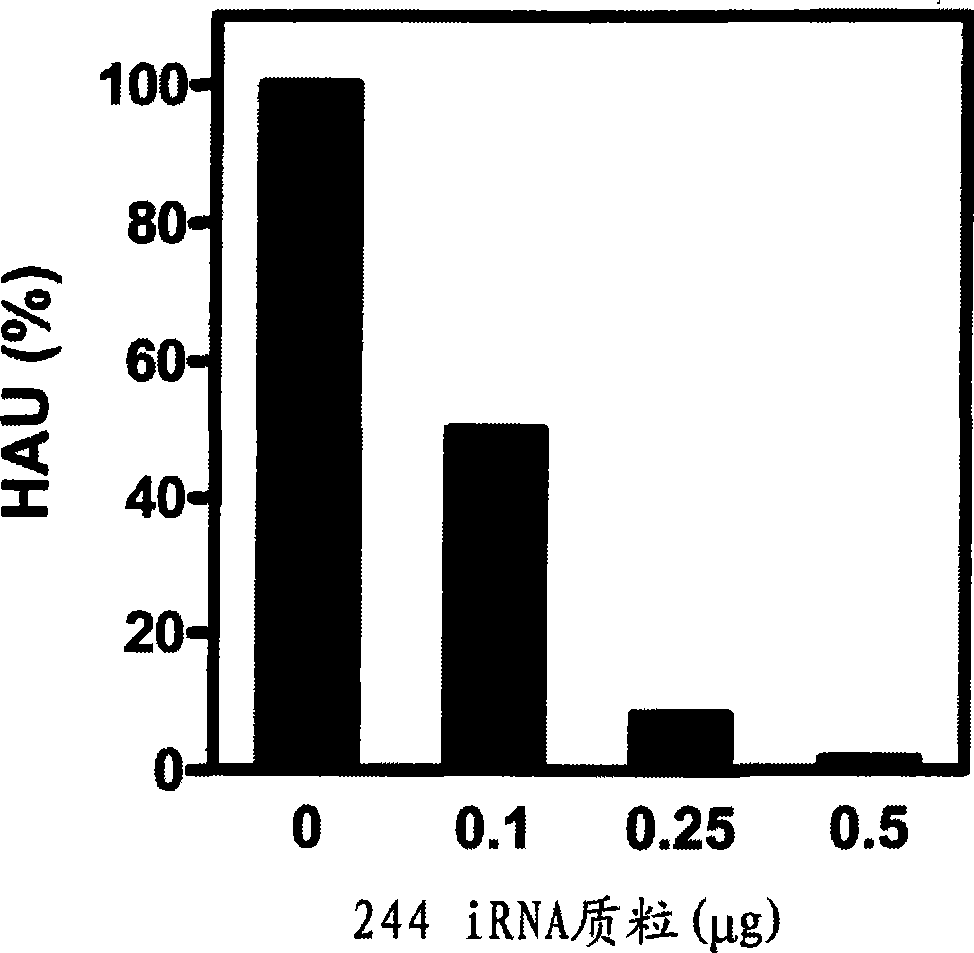
[0281] The primary protective RNA used (segment 1; RNA244) was spontaneously generated during transfection / co-cultivation of a plasmid encoding infectious A / PR / 8 / 34 (Subbarao, K. et al. 2003 Virology 305 : 192-200). The DNA mixture for transfection of 293T cells contained 0.5 μg of each of the 8 A / PR8 gene segments (under the control of the Poll promoter), 0.5 μg of various PB1 and PB2 expression plasmids, 0.1 μg of PA expression plasmids, and 1 μg of NP expression plasmids and Fugene (Roche).
[0282]To optimize the transfection of the 244 iRNA plasmid, the 244 RNA was cloned into the Poll expression plasmid pPOLI-SapIT (Subbarao (2003) supra) in order to express the vRNA sense transcript. Different amounts of 244 plasmid (0-0.5 μg) were transfected into 293T cells as described above. After 24 hours, 293T cells were trypsinized, mixed with MDCK cells and replated. Culture supernatants were harvested after 7 day...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More