Anti-dll4 antibodies and methods using same.
A technology of antibody and variant, applied in the field of molecular biology
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0432] v. Preparation of antibody-drug conjugates
[0433] In the antibody-drug conjugate (ADC) of the present invention, the antibody (Ab) is coupled to one or more drug moieties (D) via a linker (L), for example, about 1 to about 20 moieties per antibody. drug module. The ADC of general formula I can be prepared through several routes using organic chemical reactions, conditions and reagents known to those skilled in the art, including: (1) the nucleophilic group of the antibody reacts with a divalent linker reagent through a covalent bond to form Ab-L, which subsequently reacts with the drug moiety D; and (2) the nucleophilic group of the drug moiety reacts via a covalent bond with a divalent linker reagent to form D-L, which subsequently reacts with the nucleophilic group of the antibody. Additional methods for preparing ADCs are described herein.
[0434] Ab-(L-D)p I
[0435] A joint may consist of one or more joint components. Exemplary linker building blocks include...
Embodiment 1
[0512] Example 1: Materials and methods
[0513] The following materials and methods were used in the examples.
[0514] HUVEC fibrin gel bead assay. Details of the HUVEC fibrin gel bead assay have been described (Nakatsu, M.N. et al., Microvasc Res 66, 102-12 (2003)). Briefly, Cytodex was coated with 350-400 HUVEC / bead TM 3 beads (Amersham Pharmacia Biotech). Embed approximately 200 HUVEC-coated beads into the fibrin clot in one well of a 12-well tissue culture plate. will be 8 x 10 4 SF cells were plated on top of the clot. Assays were terminated between days 7 and 9 for immunostaining and imaging. In some experiments, HUVEC shoots were visualized by staining with biotin-anti-CD31 (clone WM59, eBioscience) and streptavidin-Cy3. For HUVEC nuclei staining, fibrin gels were fixed overnight in 2% paraformaldehyde (PFA) and stained with 4',6-diamidino-2-phenylindole (DAPI, Sigma). For Ki67 staining, fibrin gels were treated with 10X trypsin-EDTA for 5 min to remove upper...
Embodiment 2
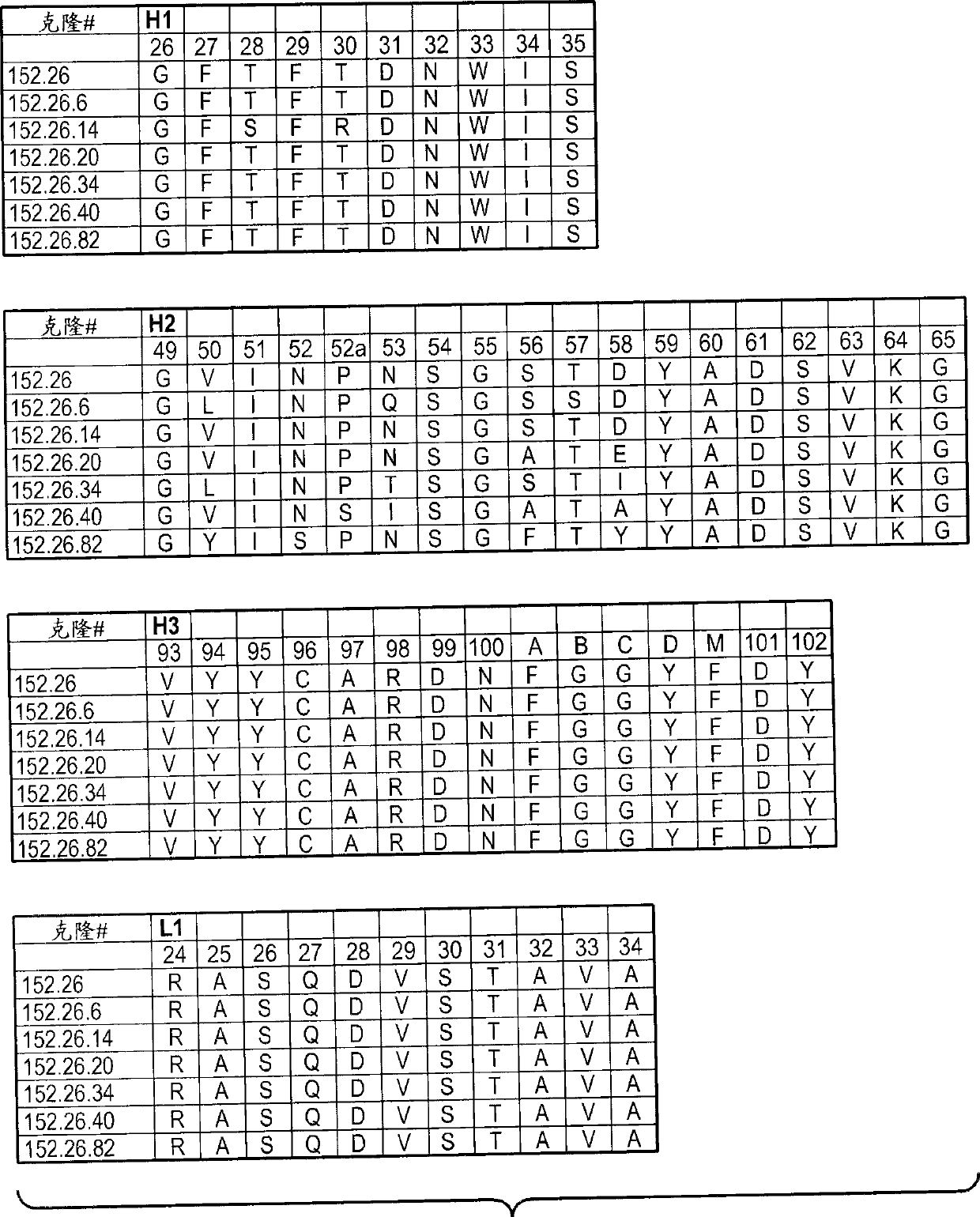
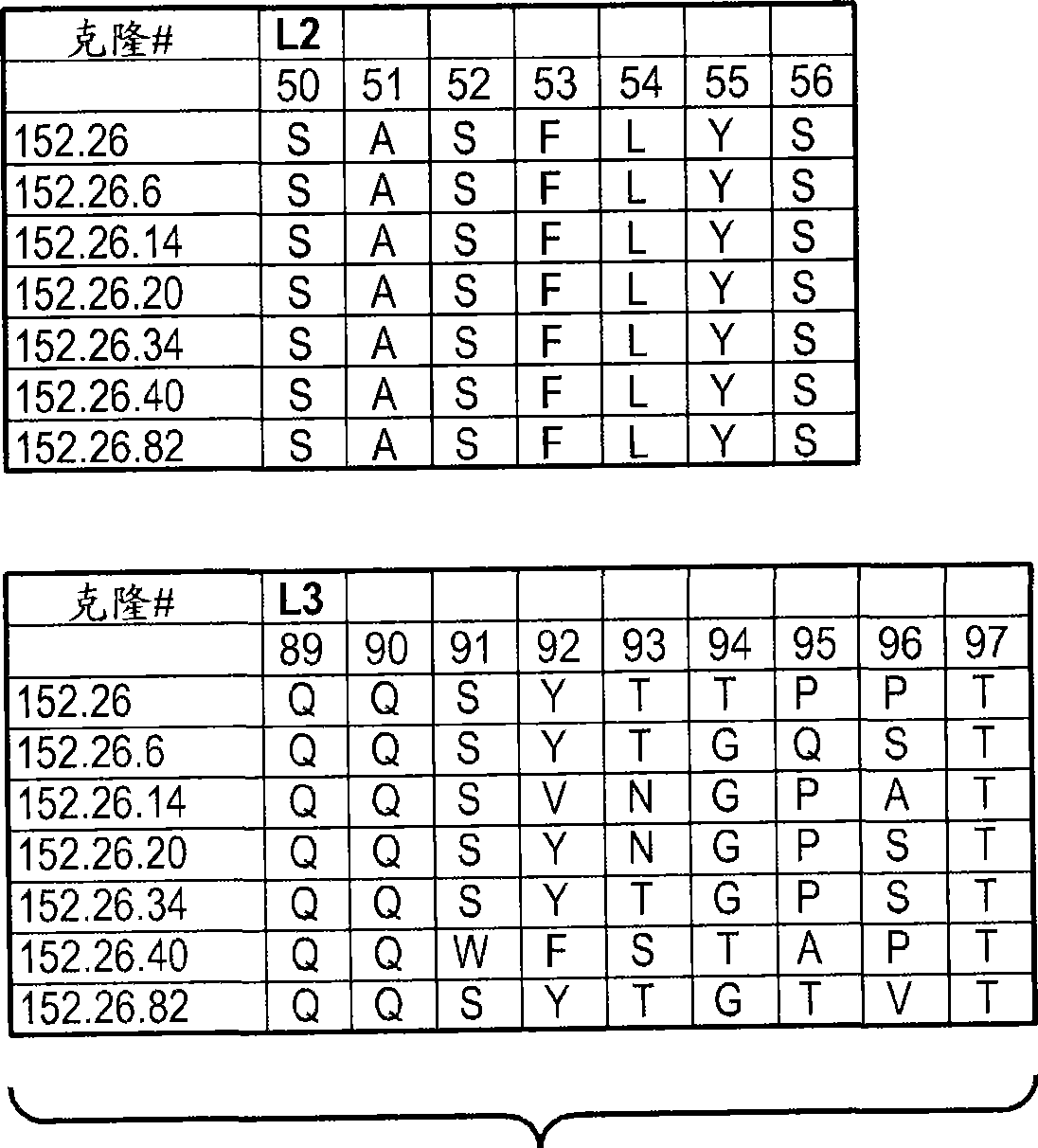
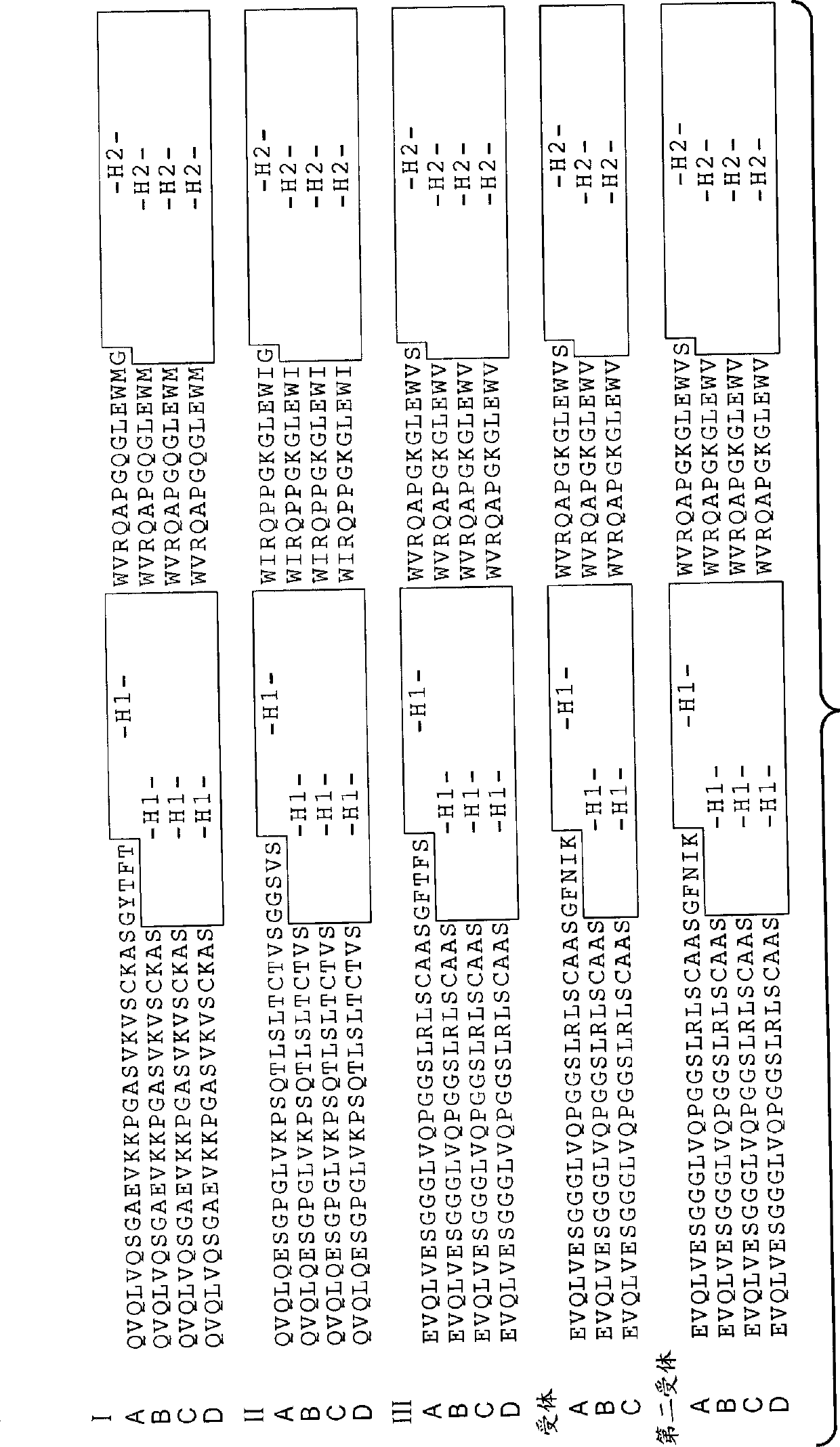
[0542] Example 2: Generation of phage anti-DLL4 antibodies
[0543] A synthetic phage antibody library was constructed on a single framework (humanized anti-ErbB2 antibody, 4D5) by introducing diversity within the complementarity-determining regions (CDRs) of the heavy and light chains (Lee, C.V. et al., J Mol Biol 340, 1073- 93 (2004); Liang, W.C. et al., J Biol Chem 281, 951-61 (2006)). For immobilization in MaxiSorp TM His-tagged human DLL4 (amino acid 1-404) on the immunoplate was implemented against unimmunized ( ) plate panning of the library. After four rounds of enrichment, clones were randomly picked and specific binders were identified using phage ELISA. The resulting hDLL4-binding clones were further screened with His-tagged murine DLL4 protein to identify cross-species clones. For each positive phage clone, the heavy and light chain variable regions were subcloned into a pRK expression vector engineered to express the full-length IgG chain. The heavy and ligh...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More