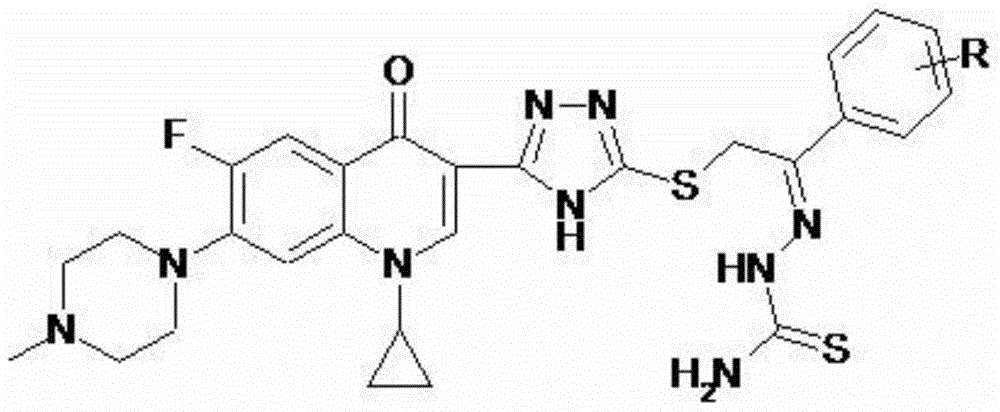
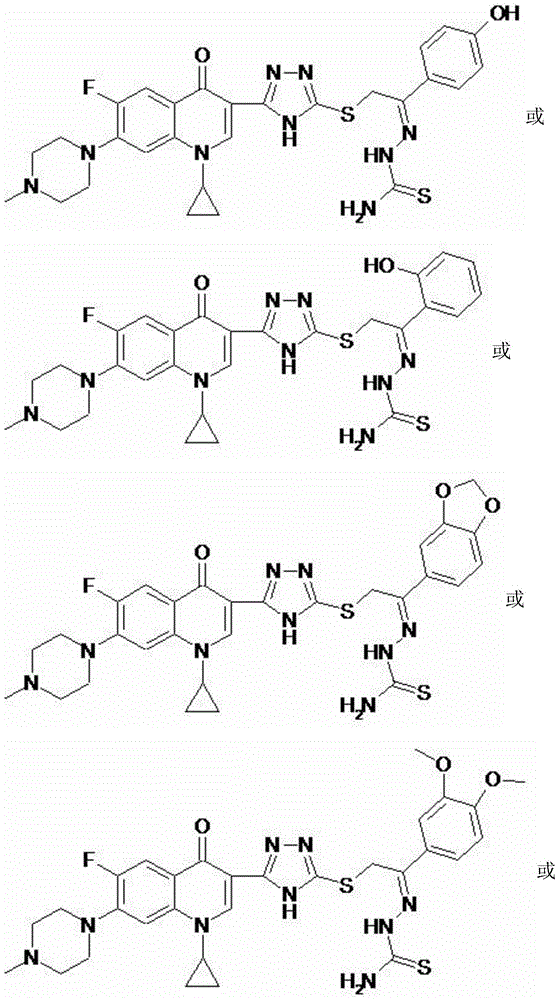
Cyprofluoroquinolone C-3-s-triazole thioetherketone thiosemicarbazone compound and its preparation method and application
A technology of azole thioetherketone thiosemicarbazone and cyprofluoroquinolone, which is applied in the field of drug synthesis and can solve the problems of low tumor therapeutic index and high toxicity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
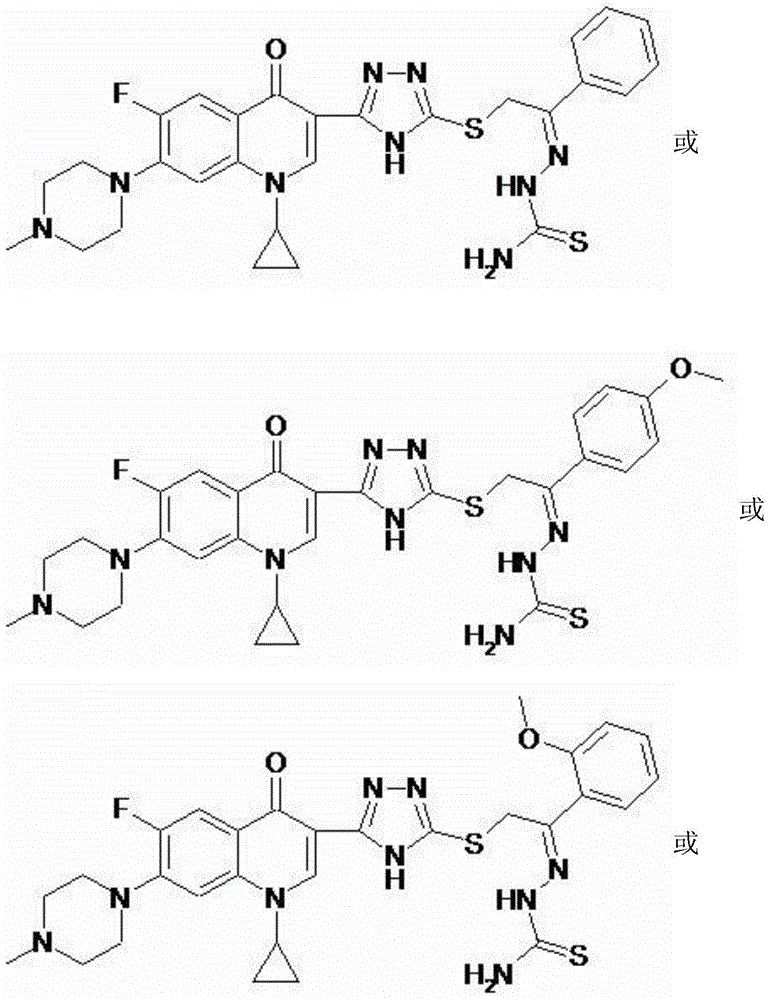
[0046] 2-{5-[1-cyclopropyl-6-fluoro-7-(4-methylpiperazin-1-yl)-quinolin-4(1H)-one-3-yl]-4H-[1 ,2,4] triazole-3-sulfanyl}-1-acetophenone thiosemicarbazone (I-1), its chemical structural formula is:
[0047]
[0048] That is, R in formula I is a hydrogen atom.
[0049] The preparation method of this compound is: take the intermediate (S)-2-{5-[1-cyclopropyl-6-fluoro-7-(4-methylpiperazin-1-yl)-quinoline-4( 1H)-keto-3-yl]-4H-[1,2,4]triazole-3-thio)-1-acetophenone (VI-1) (2.0g, 3.9mmol) was dissolved in glacial acetic acid ( 20 mL), thiosemicarbazide (0.42 g, 4.7 mmol) was added, and the mixture was refluxed for 6 h. Evaporate the solvent under reduced pressure, add water (30 mL) to dissolve, add 0.1 g of activated carbon and stir at 60 °C for 1 h to decolorize. The filtrate was adjusted to neutral with concentrated ammonia water, and the resulting solid was collected by filtration, washed with water, dried, and recrystallized from absolute ethanol to obtain a light yellow cr...
Embodiment 2
[0051] 2-{5-[1-cyclopropyl-6-fluoro-7-(4-methylpiperazin-1-yl)-quinolin-4(1H)-one-3-yl]-4H-[1 ,2,4] triazole-3-sulfanyl}-1-(p-methoxyphenyl)-ethanone thiosemicarbazone (I-2), its chemical structural formula is:
[0052]
[0053] That is, R in formula I is p-methoxy.
[0054]The preparation method of this compound is: take the intermediate (S)-2-{5-[1-cyclopropyl-6-fluoro-7-(4-methylpiperazin-1-yl)-quinoline-4( 1H)-keto-3-yl]-4H-[1,2,4]triazole-3-thio)-1-(p-methoxyphenyl)-ethanone (VI-2) (2.0g, 3.6mmol) was dissolved in glacial acetic acid (20mL), thiosemicarbazide (0.37g, 4.0mmol) was added, and the mixture was refluxed for 10h. Evaporate the solvent under reduced pressure, add water (30 mL) to dissolve, add 0.1 g of activated carbon and stir at 60 °C for 1 h to decolorize. The filtrate was adjusted to neutral with concentrated ammonia water, and the resulting solid was collected by filtration, washed with water, dried, and recrystallized from absolute ethanol to obtain ...
Embodiment 3
[0056] 2-{5-[1-cyclopropyl-6-fluoro-7-(4-methylpiperazin-1-yl)-quinolin-4(1H)-one-3-yl]-4H-[1 ,2,4] triazole-3-sulfanyl}-1-(o-methoxyphenyl)-ethanone thiosemicarbazone (I-3), its chemical structural formula is:
[0057]
[0058] That is, R in formula I is o-methoxy.
[0059] The preparation method of this compound is: take the intermediate (S)-2-{5-[1-cyclopropyl-6-fluoro-7-(4-methylpiperazin-1-yl)-quinoline-4( 1H)-keto-3-yl]-4H-[1,2,4]triazole-3-thio)-1-(o-methoxyphenyl)-ethanone (VI-3) (2.0g, 3.6mmol) was dissolved in glacial acetic acid (20mL), thiosemicarbazide (0.49g, 5.4mmol) was added, and the mixture was refluxed for 12h. Evaporate the solvent under reduced pressure, add water (30 mL) to dissolve, add 0.1 g of activated carbon and stir at 60 °C for 1 h to decolorize. The filtrate was adjusted to neutrality with concentrated ammonia water, and the resulting solid was collected by filtration, washed with water, dried, and recrystallized from absolute ethanol to obt...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More