Methods and compositions for reducing parathyroid levels
A horizontal, biologically effective technology that can be used in drug combinations, chemical instruments and methods, formulations for in vivo testing, etc., and can solve problems such as no joint use recommended
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
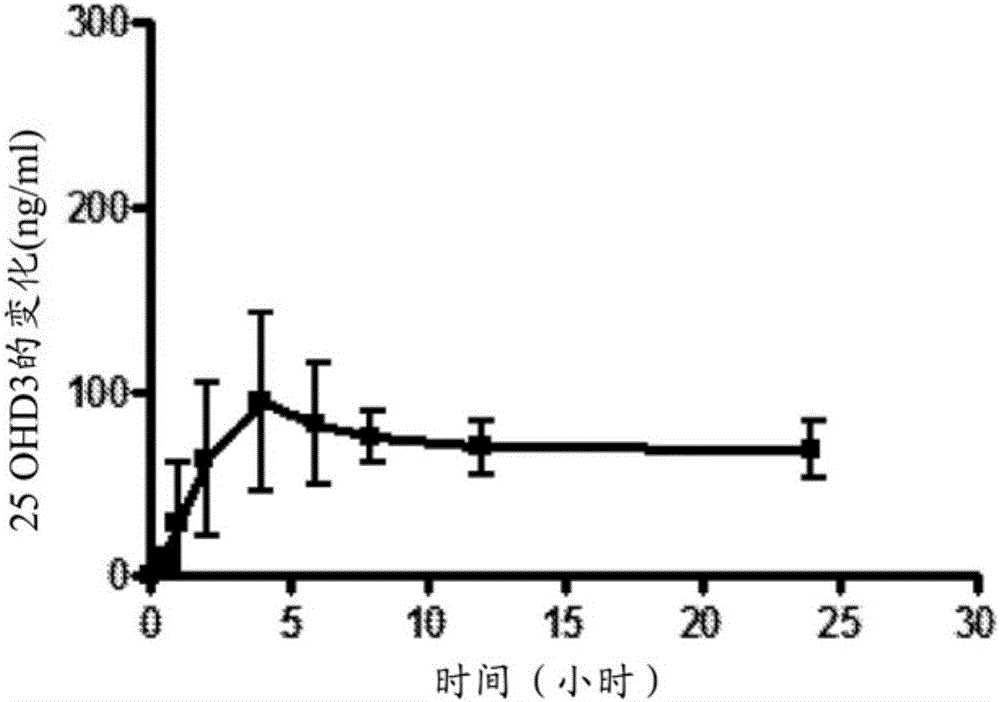
[0132] Example 1 - Sustained release formulation
[0133] Nine oral vitamin D formulations were prepared by uniformly mixing the indicated amounts of the identified components and filling said mixture into hard gelatin capsules according to Table 2 below. Formulation 9 is an immediate release formulation according to the prior art, wherein MIGLYOL 812N is a trade name for caprylic / capric triglycerides, available from CONDEA Chemie GmbH, Cranford, NJ, USA. The formulation was formulated with 250 μg of 25-hydroxyvitamin D 3 A single dose equivalent to Yucatan minipigs (approximately 10kg) group. Each group contained 5 animals. 250 μg equivalent of 25-hydroxyvitamin D 3 A tenth group of Yucatan minipigs was administered intravenously.
[0134] Blood was collected pre-dose, 0.5, 1, 2, 4, 6, 8, 12, 24, 48, 96, 168, 240, 336, 432, 504, 576, and 672 hours post-dose. Analysis of Serum 25-Hydroxyvitamin D by Liquid Chromatography Mass Spectrometry / Mass Spectrometry (LCMS / MS) 3 s ...
Embodiment 2
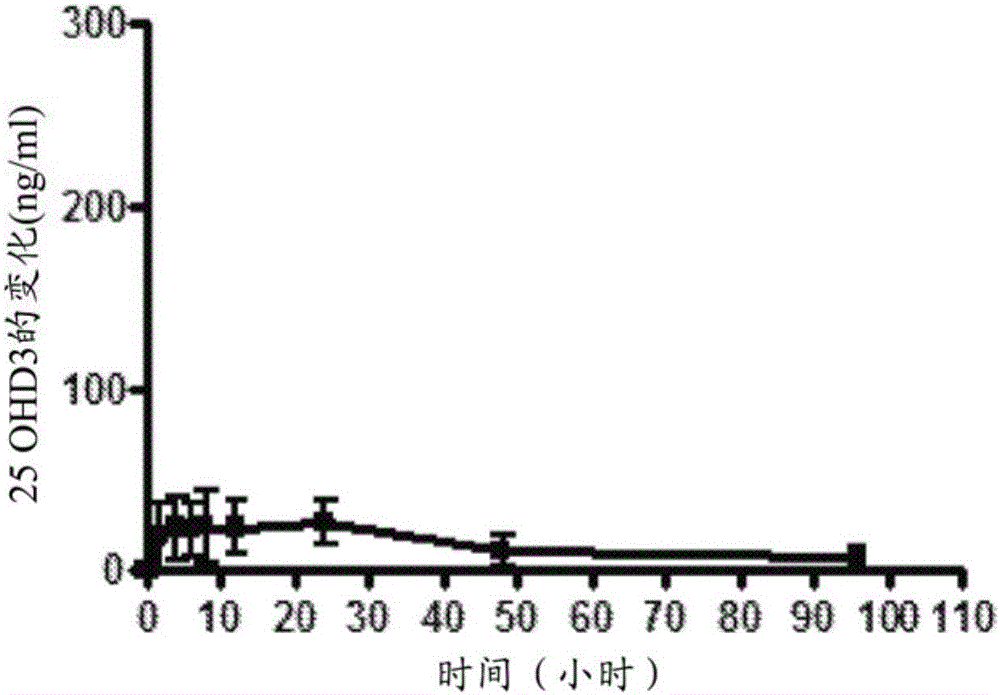
[0148] Example 2 - Pharmacokinetic Study of Minipigs Oral Capsules
[0149] The purpose of the study was to evaluate the administration of 3 consecutive days: a) 1 x 250 μg 25-hydroxyvitamin D 3 Modified release (MR) capsules, b) 2 x 250 μg MR capsules, c) 4 x 250 μg MR capsules, d) 1 x 1000 μg MR capsules, e) 1 x 250 μg immediate release (IR) 25-hydroxyvitamin D 3 25-hydroxyvitamin D in male Yucatan pigs (about 45 kg body weight) after capsules and f) 1 x 250 μg MR capsules 3 systemic absorption.
[0150] The MR formulation was prepared according to the formulation of Example 1, Group 7 above. In the case of 1000 μg MR capsules, 25-hydroxyvitamin D 3 The highest concentration of was offset by the relative decrease in ethanol.
[0151] To prepare the IR formulation, 25-hydroxyvitamin D3 (0.12% wt / wt; 250 μg per capsule) was dissolved in ethanol USP (2.32% wt / wt; solubilizer) and mixed with corn oil USP (97.54% wt / wt; vehicle) and butylated hydroxytoluene (0.02% wt / wt; ant...
Embodiment 3
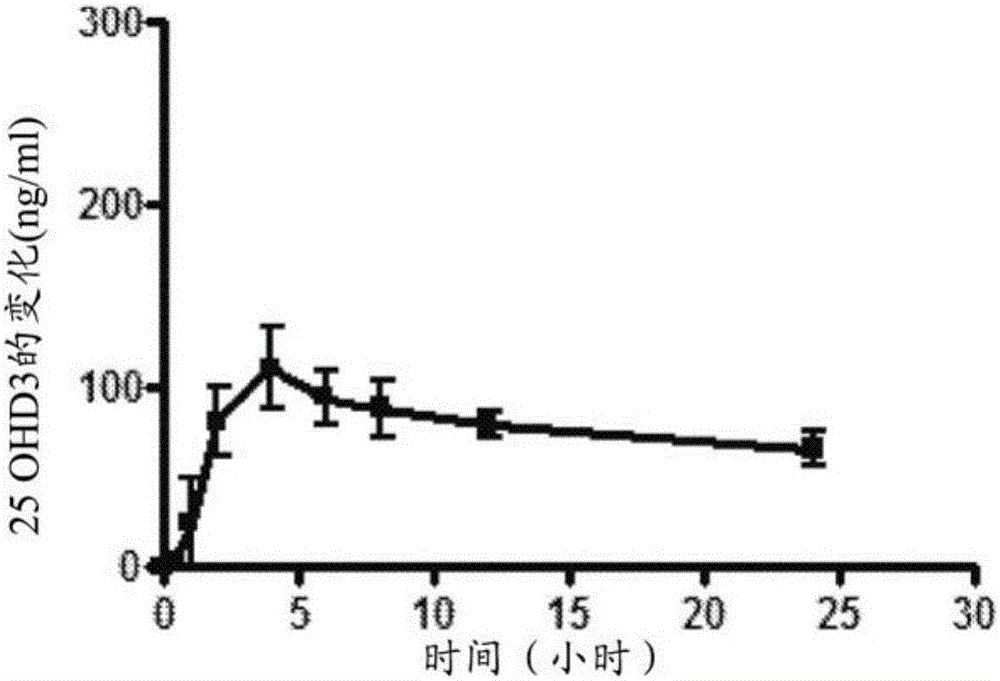
[0167] Example 3 - Study of Systemic Exposure in Minipigs Oral Capsules
[0168] The aim of this study was to evaluate systemic 25-hydroxyvitamin D in healthy normal male Yucatan pigs (up to 50-60 kg body weight) 3 The pigs were maintained on a diet containing adequate vitamin D intake and then dosed daily for 21 days as follows: a) 25 μg immediate release (IR) 25-hydroxyvitamin D 3 Capsules (group 1), b) 25 μg modified release (MR) 25-hydroxyvitamin D 3 Capsules (group 2), and c) 125 μg MR25-hydroxyvitamin D 3 Capsules (group 3).
[0169] Based on Group 7 above, the formulation of Example 1 was prepared as an MR formulation. 25-Hydroxyvitamin D 3 Differences in concentrations were offset by relative changes in ethanol.
[0170] For the preparation of IR formulations, 25-hydroxyvitamin D 3 (0.12% wt / wt, 250 μg per capsule) dissolved in ethanol USP (2.32% wt / wt, solubilizer), and mixed with corn oil USP (97.54% wt / wt, main vehicle) and butylated hydroxytoluene ( 0.02% wt...
PUM
| Property | Measurement | Unit |
|---|---|---|
| melting point | aaaaa | aaaaa |
| kinematic viscosity | aaaaa | aaaaa |
| pour point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More