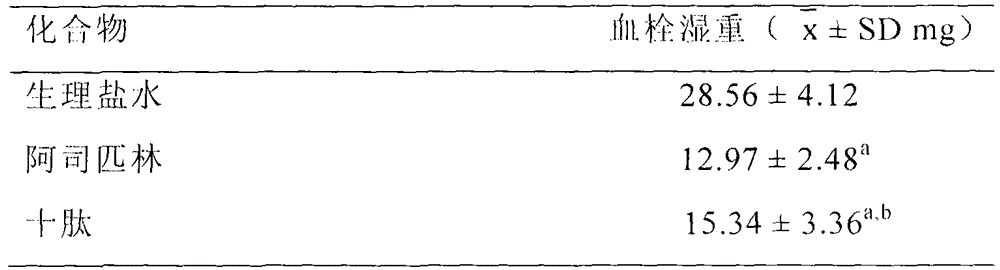
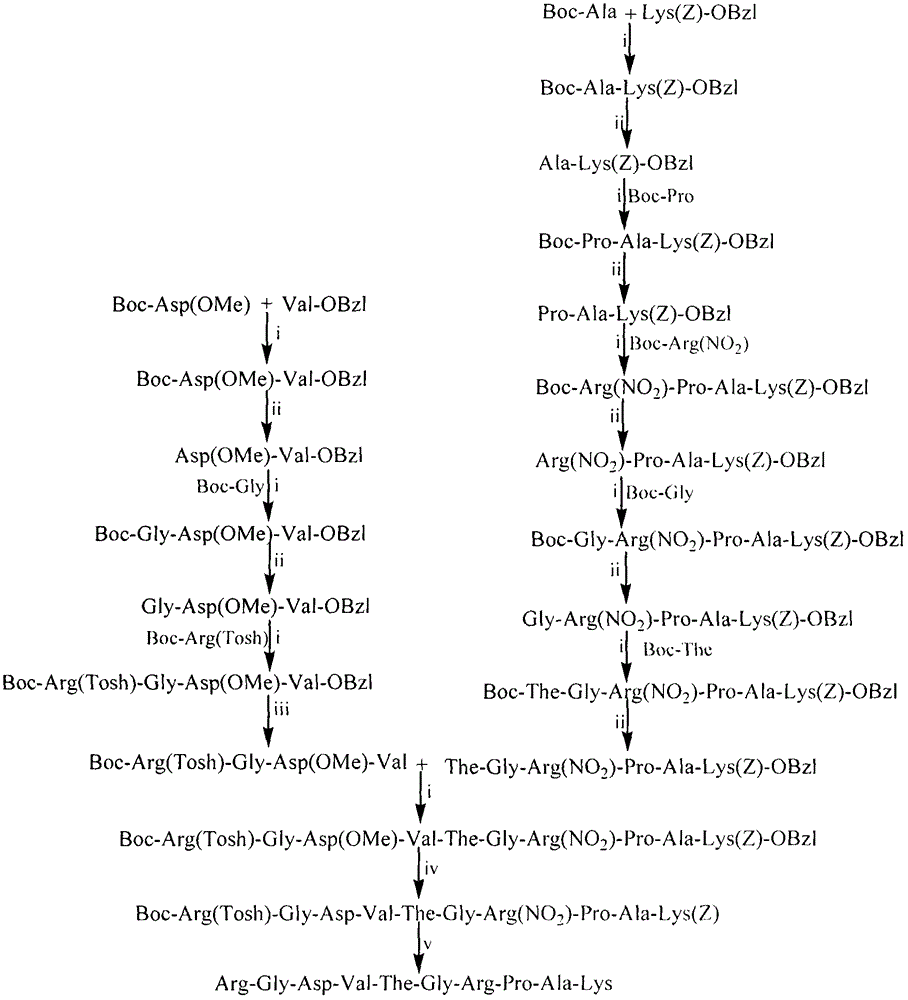RGDV-The-GRPAK decapeptide and its preparation method, activity and use
A technology of -z-lys-obzl and -obzl-asp-val, applied in the preparation method of peptides, peptides, peptide/protein components, etc., can solve the problems of thrombolytic drug bleeding, reembolization, reperfusion injury, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0030] Embodiment 1 prepares Boc-Ala-Lys(Z)-OBzl(1)
[0031] Dissolve 2.08g (11mmol) Boc-Ala in 20mL anhydrous tetrahydrofuran (THF), add 1.35g (10mmol) 1-hydroxybenzotriazole (HOBt) and 2.47g (12mmol) dissolved in anhydrous THF under ice cooling Dicyclohexylcarbonyldiimide (DCC) was stirred for 0.5 hour to obtain reaction solution A. After 30 minutes, 4.07g (10mmol) HCl Lys(Z)-OBzl was dissolved in 15mL THF, and the pH was adjusted to 9 with N-methylmorpholine, then added to the reaction solution A, stirred at room temperature for 12 hours, TLC (developing agent CHCl 3 :MeOH=30:1) showed that HCl·Lys(Z)-OBzl disappeared. The dicyclohexylurea (DCU) in the reaction solution was removed by filtration, the reaction solution was concentrated to dryness under reduced pressure, the residue was dissolved in 100 mL of ethyl acetate, and the insoluble matter was filtered off. The filtrate was successively washed 3 times with saturated aqueous sodium bicarbonate solution, 3 times wit...
Embodiment 2
[0032] Example 2 Preparation of HCl Ala-Lys(Z)-OBzl(2)
[0033] Dissolve 2.0g (3.7mmol) Boc-Ala-Lys(Z)-OBzl in a small amount of dry ethyl acetate, add 20mL of 4N hydrogen chloride-ethyl acetate solution under ice-salt bath, react under ice-cooling for 4 hours, TLC (CHCl 3 :MeOH, 30:1) showed that the raw material point disappeared and the reaction was complete. The reaction solution was concentrated under reduced pressure, and the residue was dissolved in ethyl acetate and concentrated under reduced pressure. This operation was repeated three times to remove free hydrogen chloride, and the residue was washed three times with anhydrous ether to obtain 1.6 g (90%) of the title compound as a colorless powder. ESI-MS(m / z): 442[M+H] + .
Embodiment 3
[0034] Embodiment 3 prepares Boc-Pro-Ala-Lys(Z)-OBzl(3)
[0035]Using the method of Example 1, 1.7 g (78%) of the title compound was obtained as a yellowish oil from 731 mg (3.4 mmol) of Boc-Pro and 1.8 g (3.3 mmol) of HCl·Ala-Lys(Z)-OBzl. ESI-MS(m / z): 639[M+H] + .
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More