Preparation method and application of NUC-1031 single isomer
A technology of sp-1 and rp-1, which is applied in the field of compound Sp-1 in crystal form and its preparation, and can solve problems such as difficulties
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
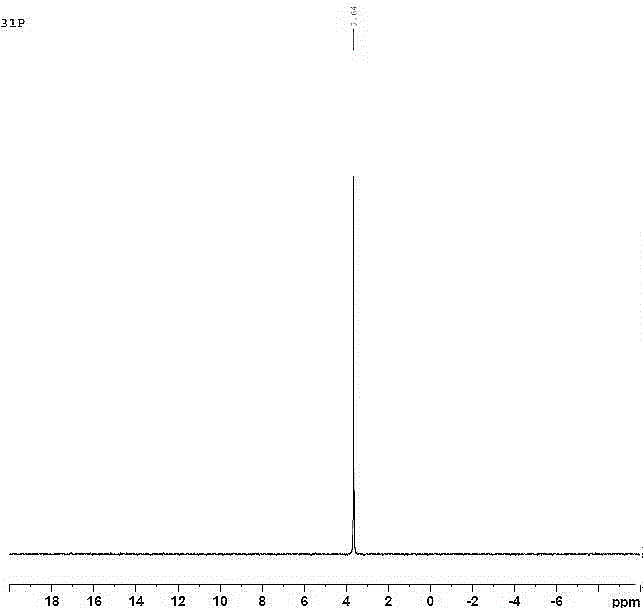
Image
Examples
Embodiment 1
[0081] Preparation of Example 1 compound (61502):
[0082]
[0083] To a solution of 61501h (20g) in dichloromethane (60ml) was added 61501g (20.6g) at -80°C, followed by a solution of 19.3g triethylamine (diluted in 20ml dichloromethane). The mixture was stirred overnight at room temperature. To the mixture was added 61501f, followed by the addition of 19.3g triethylamine (diluted with 20ml dichloromethane) solution, and the mixture was stirred at room temperature for 4h. The mixture was directly precipitated, and the residue was dissolved in ethyl acetate (200ml) and water (400ml). After separating the ethyl acetate, wash the aqueous phase with ethyl acetate (2*100ml), combine the ethyl acetate phase, and wash the acetic acid with brine ethyl ether phase and dried over anhydrous sodium sulfate. Ethyl acetate was distilled off to obtain the target compound (61502), which was directly used in the next step of purification.
Embodiment 2
[0084] Preparation of Example 2 Compound (61501b):
[0085]
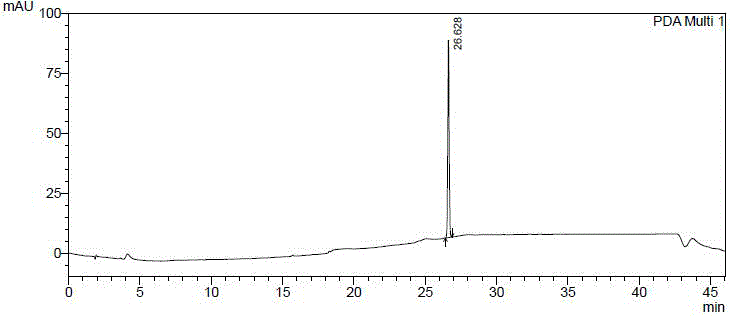
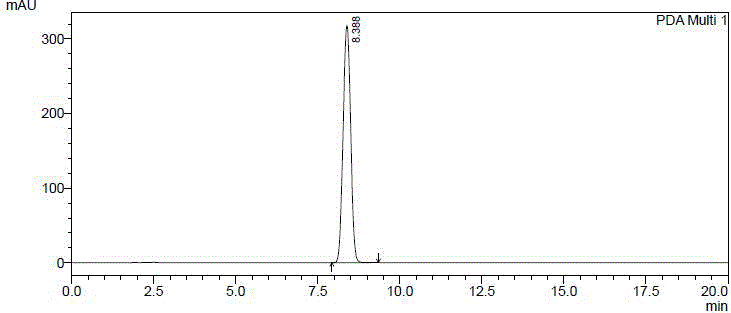
[0086] Compound 61502 (120g) was dissolved in ethyl acetate (240ml), stirred continuously, and petroleum ether (720ml) was slowly added dropwise at room temperature, crystals were precipitated, and the filtrate was removed by filtration to obtain compound 61501b (49.5g), with a yield of 41.2% , HPLC: 100.0% (such as figure 1 shown).
Embodiment 3
[0087] The preparation of embodiment 3 compound (61501c):
[0088]
[0089] Add sodium carbonate (35.4g) to a mixed solution of compound (61501d) (20g) in tetrahydrofuran (200ml) and water (100ml) at room temperature, then add di-tert-butyl dicarbonate (17.5g), and stir at room temperature until the reaction is complete . The mixture was extracted with ethyl acetate (3*200ml), and the combined ethyl acetates were washed with brine and dried over anhydrous sodium sulfate. After distilling off the solvent, the residue was purified by silica gel chromatography (2.5%~10% methanol / dichloromethane) to obtain 18 g of compound (61501c), yield: 67%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More