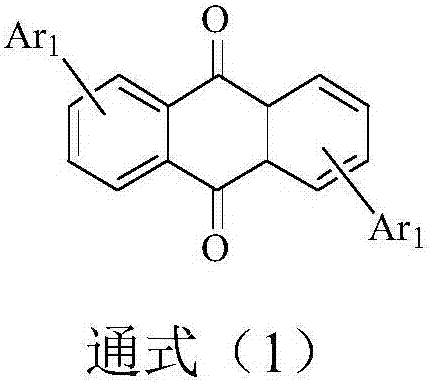
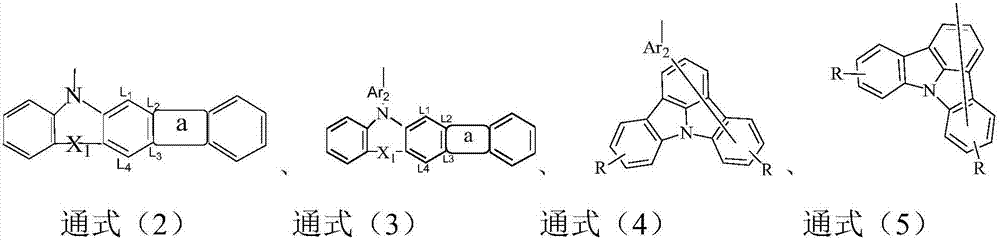
9,10-anthraquinone based compound and application thereof in organic electroluminescent devices
A technology of light-emitting devices and compounds, applied to organic electroluminescent devices, in the field of compounds containing 9,10-anthraquinone, which can solve the problems of efficiency roll-off, low S1 state radiative transition rate, difficult exciton utilization and Problems such as high fluorescence radiation efficiency, to achieve the effect of increasing orbital overlap, good industrialization prospects, and avoiding aggregation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
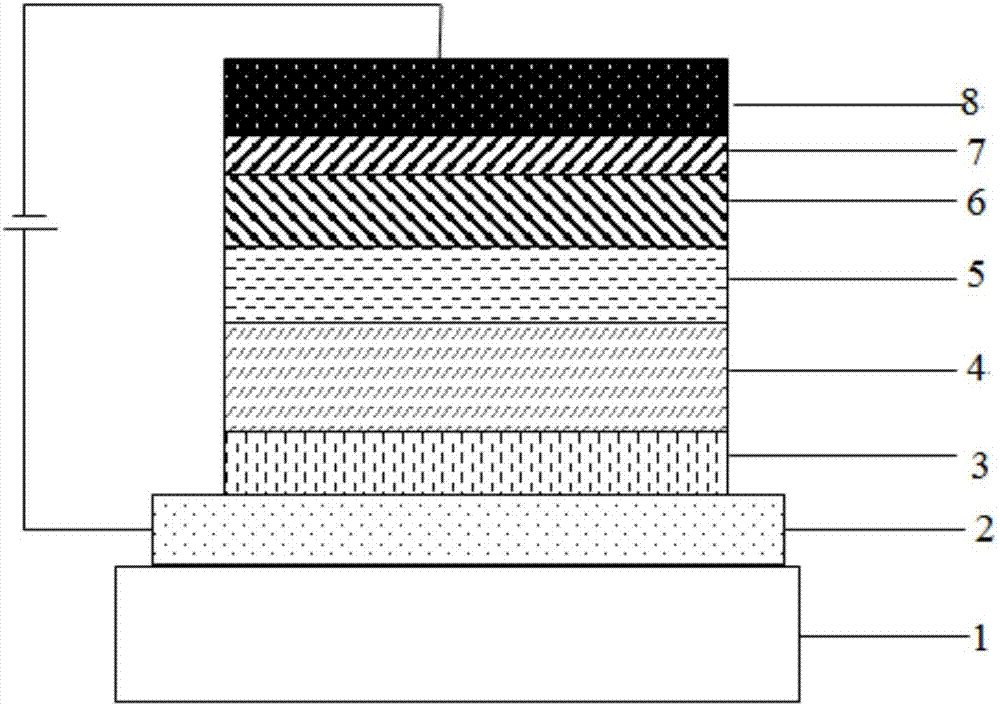
Image
Examples
Embodiment 1
[0044] Example 1 Compound 1
[0045]
[0046]In a 250ml four-necked flask, add 0.01mol 2,6-dibromo-4a,9a-dihydro-anthraquinone, 0.025mol 6,6-dimethyl-6,11-dihydro -13-Oxa-11-aza-indole[1,2-b]anthracene, 0.03mol sodium tert-butoxide, 1×10 -4 mol Pd 2 (dba) 3 , 1×10 -4 mol of tri-tert-butylphosphine, 150ml of toluene, heated to reflux for 24 hours, sampled and plated, the reaction was complete; naturally cooled, filtered, the filtrate was rotary evaporated, and passed through a silica gel column to obtain the target product with a purity of 97.2% and a yield of 75.00%.
[0047] Elemental analysis structure (molecular formula C 56 h 40 N 2 o 4 ): theoretical value C, 83.56; H, 5.01; N, 3.48; 0, 7.95; test value: C, 83.65;
[0048] HPLC-MS: The molecular weight of the material is 804.30, and the measured molecular weight is 804.96.
Embodiment 2
[0049] Example 2 Compound 5
[0050]
[0051] In a 250ml four-neck flask, under a nitrogen atmosphere, add 0.01mol 1,5-dibromo-4a,9a-dihydro-anthraquinone, 0.025mol 6,6,13,13-tetramethyl-11, 13-Dihydro-11-aza-indo[1,2-b]anthracene, 0.03mol sodium tert-butoxide, 1×10 -4 mol Pd 2 (dba) 3 , 1×10 -4 mol of tri-tert-butylphosphine, 150ml of toluene, heated to reflux for 24 hours, sampling plate, the reaction was complete; naturally cooled, filtered, the filtrate was rotary evaporated, and passed through a silica gel column to obtain the target product with a purity of 99.2% and a yield of 72.00%.
[0052] Elemental analysis structure (molecular formula C 62 h 52 N 2 o 2 ): theoretical value C, 86.88; H, 6.12; N, 3.27; O, 3.73; test value: C, 86.93; H, 6.11; N, 3.20;
[0053] HPLC-MS: The molecular weight of the material is 856.40, and the measured molecular weight is 857.10.
Embodiment 3
[0054] Example 3 Compound 6
[0055]
[0056] In a 250ml four-neck flask, add 0.01mol 2,6-bis-(4'-bromobiphenyl-3-yl)-4a,9a-dihydro-anthraquinone, 0.025mol 11H- 6,13-Dioxa-11-aza-indole[1,2-b]anthracene, 0.03mol sodium tert-butoxide, 1×10 -4 mol Pd 2 (dba) 3 , 1×10 -4 mol of tri-tert-butylphosphine, 150ml of toluene, heated to reflux for 24 hours, sampling plate, the reaction was complete; naturally cooled, filtered, the filtrate was rotary evaporated, and passed through a silica gel column to obtain the target product with a purity of 99.5% and a yield of 68.00%.
[0057] Elemental analysis structure (molecular formula C 74 h 44 N 2 o 6 ): theoretical value C, 84.07; H, 4.20; N, 2.65; 0, 9.08; test value: C, 84.21;
[0058] HPLC-MS: The molecular weight of the material is 1056.32, and the measured molecular weight is 1056.80.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More