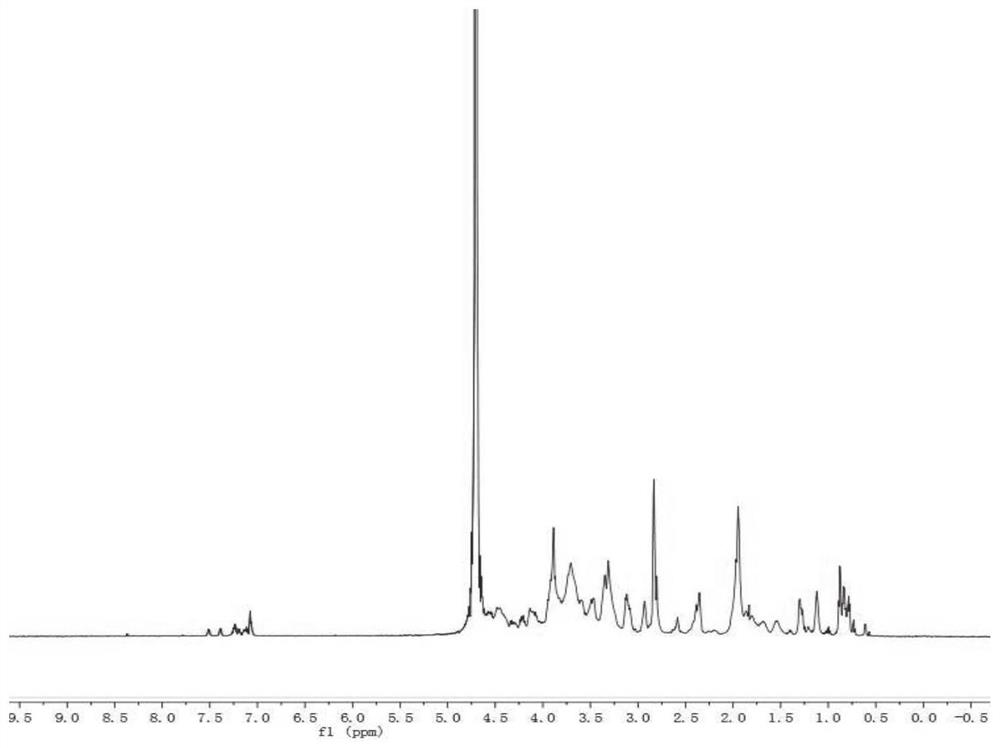
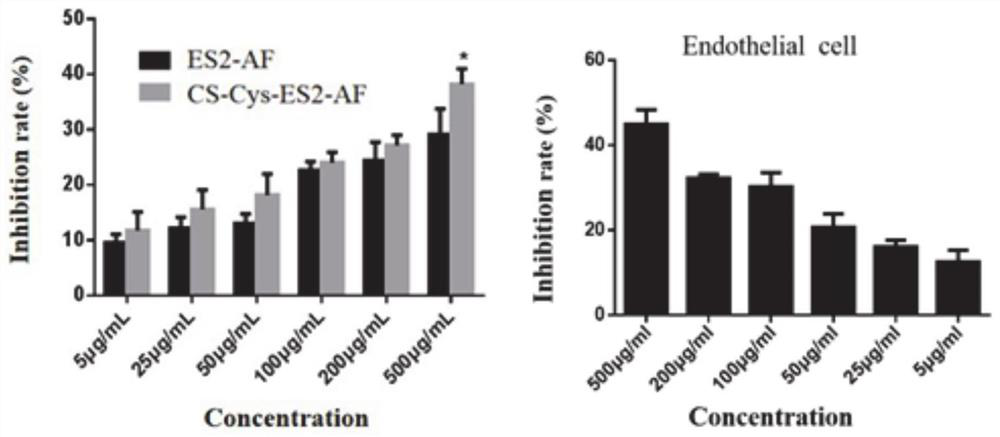
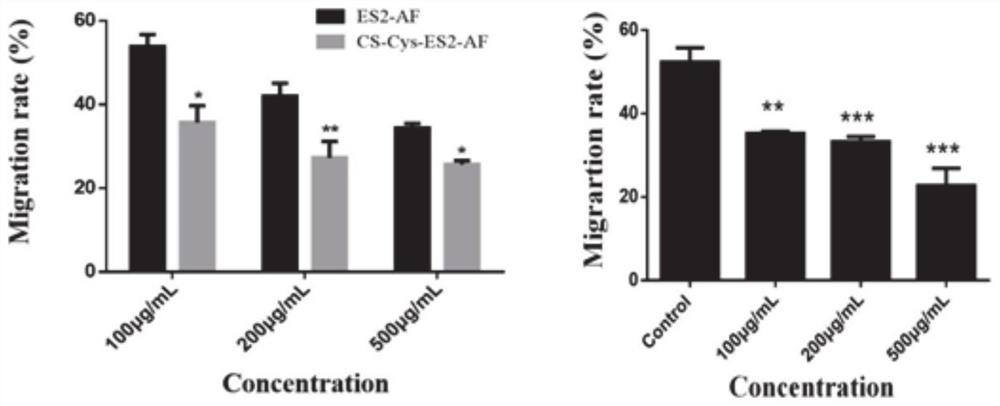
Chondroitin sulfate modifier of anti-angiogenic peptide ES2-AF and preparation method and application of cis-platinum loaded by chondroitin sulfate modifier
A technology of ES2-AF and chondroitin sulfate, applied in the field of biomedicine, can solve the problems of poor targeting, strong toxic and side effects, short half-life, etc., and achieve high stability, high stability and biological activity, good use effect and application value effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0045] In yet another specific embodiment of the present invention, the preparation method of the chondroitin sulfated modification of the anti-angiogenic peptide ES2-AF includes:
[0046] When the chondroitin sulfate modification of the anti-angiogenic peptide ES2-AF is CS-Cys-ES2-AF, its preparation method is:
[0047] S1. Preparation of CS-Cys conjugate: use Cys as a linker to connect ES2-AF and CS, so that ES2-AF can be released rapidly from CS-Cys-ES2-AF in the tumor microenvironment and dissociate to the surrounding tumor Endothelial cells exert anti-angiogenic effects. The specific preparation method is: dissolve CS in water, add EDCI and NHS as catalysts to activate the carboxyl group in CS, add lye to adjust the pH to weak alkaline after activation, and then slowly add Cys solution for reaction. After the reaction is completed, the reaction product After purification, the CS-Cys conjugate is obtained;
[0048] S2. Preparation of CS-Cys-ES2-AF conjugate: Dissolve ES2...
Embodiment 1
[0073] Example 1: Preparation of the chondroitin sulfated modification of the anti-angiogenic peptide ES2-AF (CS-Cys-ES2-AF) The preparation steps are as follows:
[0074] (1) The anti-angiogenic peptide ES2-AF (IVRRADRAAVPGGGGGGNQWFI) was prepared by solid-phase synthesis (synthesized by Ningbo Kangbei Biochemical Co., Ltd.), and the purity of ES2-AF was analyzed by high performance liquid chromatography. The detection wavelength is 220nm, the chromatographic column is a SHIMADZU Inertsil ODS-SP (4.6×250mm, 5 μm) column, the mobile phase A is an aqueous solution containing 0.1% trifluoroacetic acid, and the mobile phase B is an acetonitrile solution containing 0.1% trifluoroacetic acid. The gradient elution was carried out with 15%-75% mobile phase B within 25min at 1.0mL / min.
[0075] (2) Weigh 71mg CS, dissolve it in 30mL double-distilled water, add 710mg EDCI and 70mg NHS after complete dissolution, and activate for 20min. Adjust the pH to 7.4 with 1mol / L NaOH, add 1.65g ...
Embodiment 2
[0077] Example 2: Preparation of chondroitin sulfate-modified anti-angiogenic peptide ES2-AF and cisplatin-loaded conjugate (CDDP-CS-Cys-ES2-AF)
[0078] The preparation process is as follows:
[0079] (1) The anti-angiogenic peptide ES2-AF (IVRRADRAAVPGGGGGGNQWFI) was prepared by solid-phase synthesis (synthesized by Ningbo Kangbei Biochemical Co., Ltd.), and the purity of ES2-AF was analyzed by high performance liquid chromatography. The detection wavelength is 220nm, the chromatographic column is a SHIMADZU Inertsil ODS-SP (4.6×250mm, 5 μm) column, the mobile phase A is an aqueous solution containing 0.1% trifluoroacetic acid, and the mobile phase B is an acetonitrile solution containing 0.1% trifluoroacetic acid. The gradient elution was carried out with 15%-75% mobile phase B within 25min at 1.0mL / min.
[0080] (2) Weigh 71mg CS, dissolve it in 30mL double-distilled water, add 710mg EDCI and 70mg NHS after complete dissolution, and activate for 20min. Adjust the pH to 7...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap