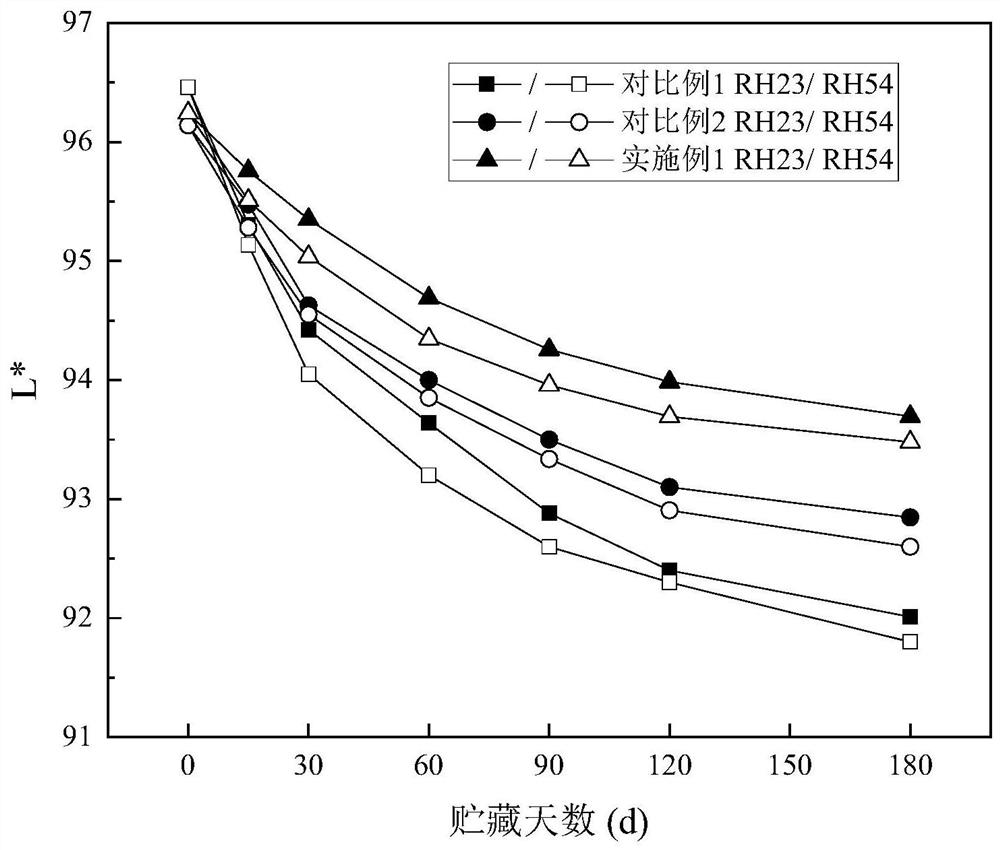
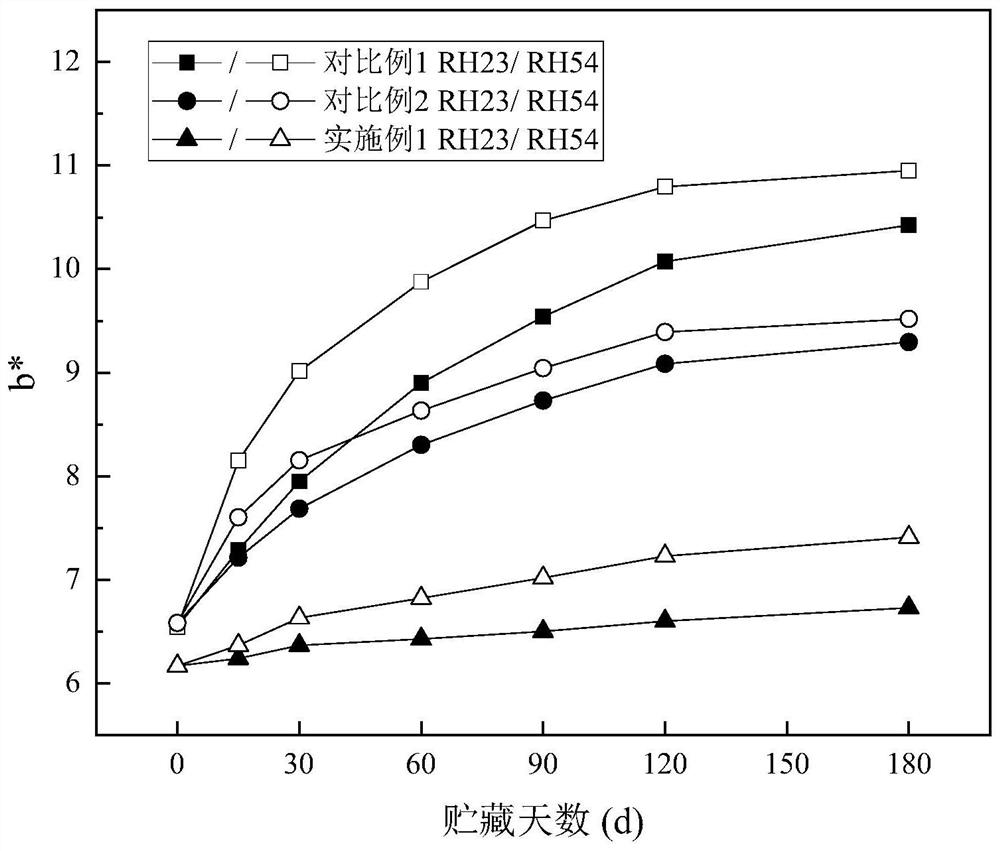
Milk powder with maltodextrin replaced by maltopentaglycosyl trehalose and preparation method
A technology of maltopentasaccharide and maltodextrin, which is applied in the direction of milk preparations, dairy products, applications, etc., can solve the problems of milk powder polymerization, bonding instability, milk powder browning, etc., to achieve stable batch production and low production costs , The effect of simple production process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0036] The preparation of maltopentaosyl trehalose used in Example 1 and Comparative Example 2 refers to the patent CN 111304270 A, which specifically includes the following steps:
[0037] Add β-cyclodextrin to water to obtain a cyclodextrin solution with a concentration of 20g / L; then add 5U / g 环糊精 cyclodextrin degrading enzyme and 30U / g 环糊精 The maltooligosaccharide-based trehalose synthase was added to the cyclodextrin solution, and reacted at 35°C and pH 7.5 for 30 minutes to obtain a reaction solution containing maltodextrin. The reaction solution was deactivated (boiling for 10 minutes), After decolorization (activated carbon decolorization), use a chromatographic column with a size of 1.6cm × 100cm, which is filled with Na-type cation exchange resin, and the height is 70% of the chromatographic column. The solution with a purity of more than 95% was collected at a flow rate of mL / min, and then freeze-dried at -60°C for 24 hours to obtain maltopentaosyl trehalose (non-re...
Embodiment 2
[0081] A formula milk powder with maltopentaosyl trehalose instead of maltodextrin, comprising the following raw materials in parts by mass: 90 parts of fresh milk, 1.5 parts of maltopentaosyl trehalose, 2 parts of rapeseed oil, and 1 part of sunflower oil , 2 parts of coconut oil, 1 part of desalted whey powder, 1.5 parts of concentrated whey protein powder, 0.8 part of lactose, 0.1 part of multivitamin, 0.1 part of multivitamin;
[0082] Wherein, the components of the multivitamin include vitamin A, vitamin C, vitamin D, vitamin E, vitamin B1, vitamin B2, vitamin B6, vitamin B12, vitamin K1, folic acid and pantothenic acid, vitamin A, vitamin C, vitamin D The mass ratio of vitamin E, vitamin B1, vitamin B2, vitamin B6, vitamin B12, vitamin K1, folic acid and pantothenic acid is 1:3:0.004:31:6:27:18:0.002:0.008:4:10;
[0083] The components of the composite minerals include calcium carbonate, potassium chloride, magnesium chloride, zinc sulfate and iron pyrosulfate, and the m...
Embodiment 3
[0093] A formula milk powder with maltopentaosyl trehalose instead of maltodextrin, comprising the following raw materials in parts by mass: 88 parts of fresh milk, 1.5 parts of maltopentaosyl trehalose, 3 parts of palm oil, 1 part of soybean oil, coconut 2 parts of oil, 1.6 parts of desalted whey powder, 2 parts of concentrated whey protein powder, 0.8 part of lactose, 0.05 part of multivitamin, 0.05 part of multivitamin;
[0094] Wherein, the components of the multivitamin include vitamin A, vitamin C, vitamin D, vitamin E, vitamin B1, vitamin B2, vitamin B6, vitamin B12, vitamin K1, folic acid and pantothenic acid, vitamin A, vitamin C, vitamin D, The mass ratio of vitamin E, vitamin B1, vitamin B2, vitamin B6, vitamin B12, vitamin K1, folic acid and pantothenic acid is 1:4:0.003:32:8:25:20:0.001:0.01:5:10;
[0095] The components of the composite minerals include calcium carbonate, potassium chloride, magnesium chloride, zinc sulfate and iron pyrosulfate, and the mass rati...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Glass transition temperature | aaaaa | aaaaa |
| Glass transition temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap