Anti-tnf antibody compositions, and methods for treatment of psoriatic arthritis
A technology for psoriatic arthritis and psoriasis, applied in chemical instruments and methods, antibodies, drug combinations, etc., can solve the problems of low affinity, cell culture, low-yield, low-affinity, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0358] Example 1: Cloning and expression of TNF antibody in mammalian cells .
[0359] A typical mammalian expression vector contains at least one promoter element that mediates transcriptional initiation of the mRNA and antibody coding sequences, as well as signals required for transcriptional termination and polyadenylation of the transcript. Additional elements include enhancers, Kozak sequences, and intervening sequences flanked by donor and acceptor sites for RNA splicing. Efficient transcription can be achieved using the following sequences: early and late promoters from SV40, long terminal repeats (LTRS) from retroviruses such as RSV, HTLVI, HIVI, and early Promoter. However, cellular elements (such as the human actin promoter) can also be used. Suitable expression vectors for practicing the invention include, for example, vectors such as pIRES1neo, pRetro-Off, pRetro-On, PLXSN or pLNCX (Clonetech Labs, Palo Alto, CA), pcDNA3.1 (+ / -), pcDNA / Zeo (+ / -) or pcDNA3.1 / Hy...
Embodiment 2
[0368] Example 2: Production of high-affinity human IgG monoclonal antibodies reactive with human TNF using transgenic mice .
[0369] Summarize. Transgenic mice containing human heavy and light chain immunoglobulin genes have been used to generate high-affinity, fully human monoclonal antibodies that can be used therapeutically to inhibit TNF in the treatment of one or more TNF-mediated effect of the disease. (CBA / J x C57 / BL6 / J)F containing human variable and constant region antibody transgenes for heavy and light chains 2 Hybrid mice were immunized with human recombinant TNF (Taylor et al., Intl. Immunol. 6:579-591 (1993); Lonberg et al., Nature 368:856-859 (1994); Neuberger, M., Nature Biotech. 14:826 (1996); Fishwild et al., Nature Biotechnology 14:845-851 (1996)). Several fusions produced one or more panels of fully human TNF-reactive IgG monoclonal antibodies. The fully human anti-TNF antibody was further characterized. All were IgG1κ. Such antibodies were found ...
Embodiment 3
[0390] Example 3: Production of Human IgG Monoclonal Antibodies Reactive to Human TNFα .
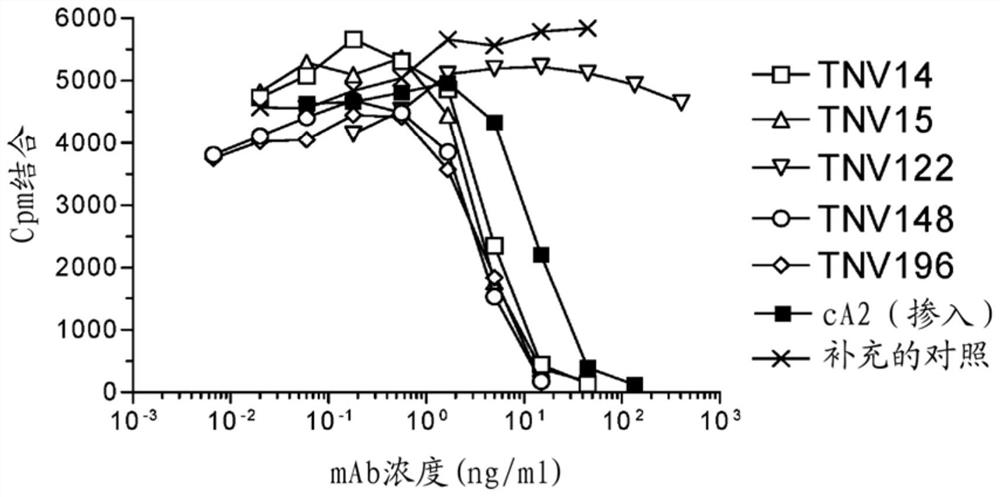
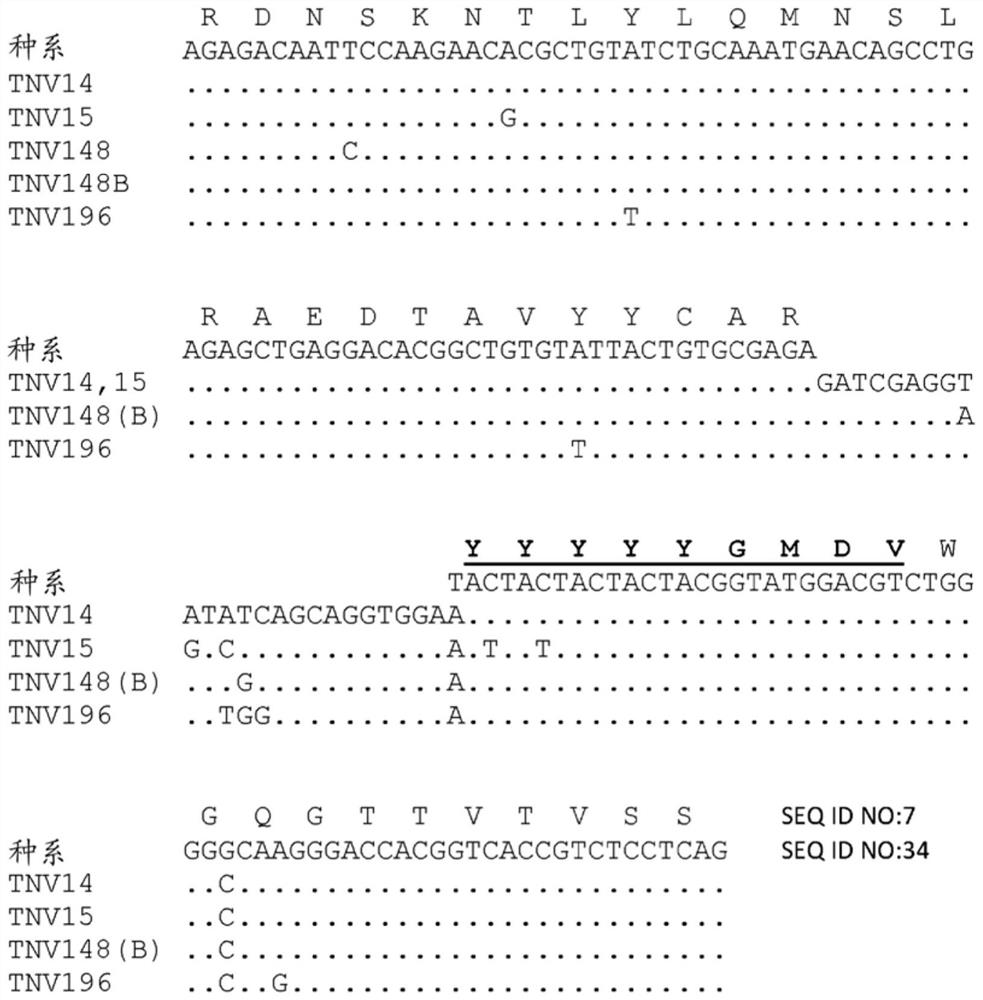
[0391] Summarize. (CBA / J x C57BL / 6J)F containing human variable and constant region antibody transgenes of heavy and light chains 2 Hybrid mice (1-4) were immunized with recombinant human TNFα. One fusion, designated GenTNV, produced eight fully human IgG1κ monoclonal antibodies that bind to immobilized recombinant human TNFα. Shortly after identification, the eight cell lines were handed over to the Institute of Molecular Biology for further characterization. Because these Mabs are fully human in sequence, they are expected to be less immunogenic in humans than cA2(Remigram).
[0392] abbreviation. BSA-Bovine Serum Albumin; CO 2 - carbon dioxide; DMSO - dimethyl sulfoxide; EIA - enzyme immunoassay; FBS - fetal bovine serum; H 2 o 2 - hydrogen peroxide; H - heavy chain; HRP - horseradish peroxidase; ID - intradermal; Ig - immunoglobulin; TNF - tissue necrosis factor alpha; IP - ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
| particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com