Liposomal camptothecins and uses thereof
a technology of liposomal camptothecin and composition, which is applied in the field of improved liposomal camptothecin composition, can solve the problems of insufficient remarkable activity of lipid formulations, insufficient dose-limiting toxicity, and further complicated fields, and achieves improved liposomal camptothecin, decreased collateral toxicity, and improved clinical efficacy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
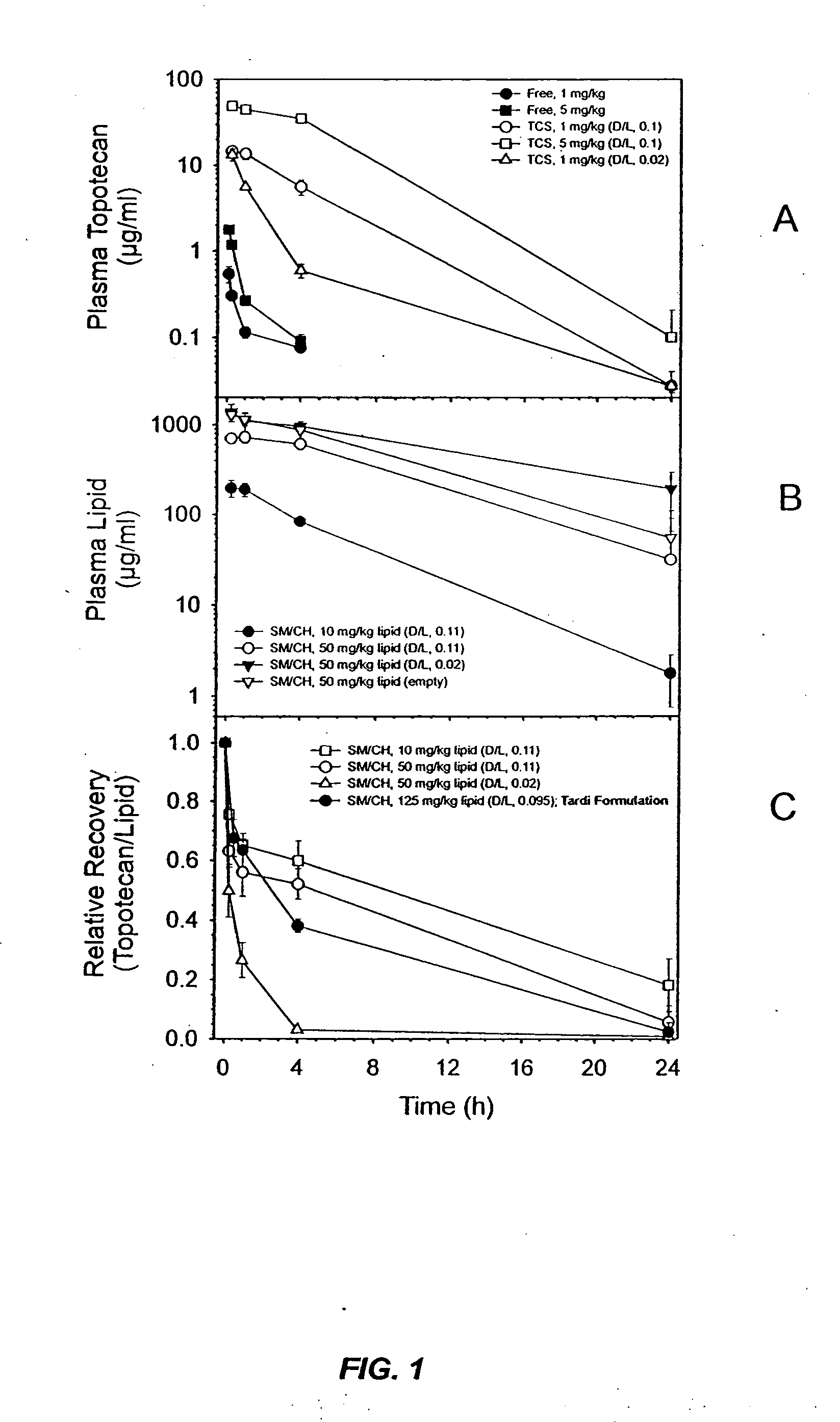
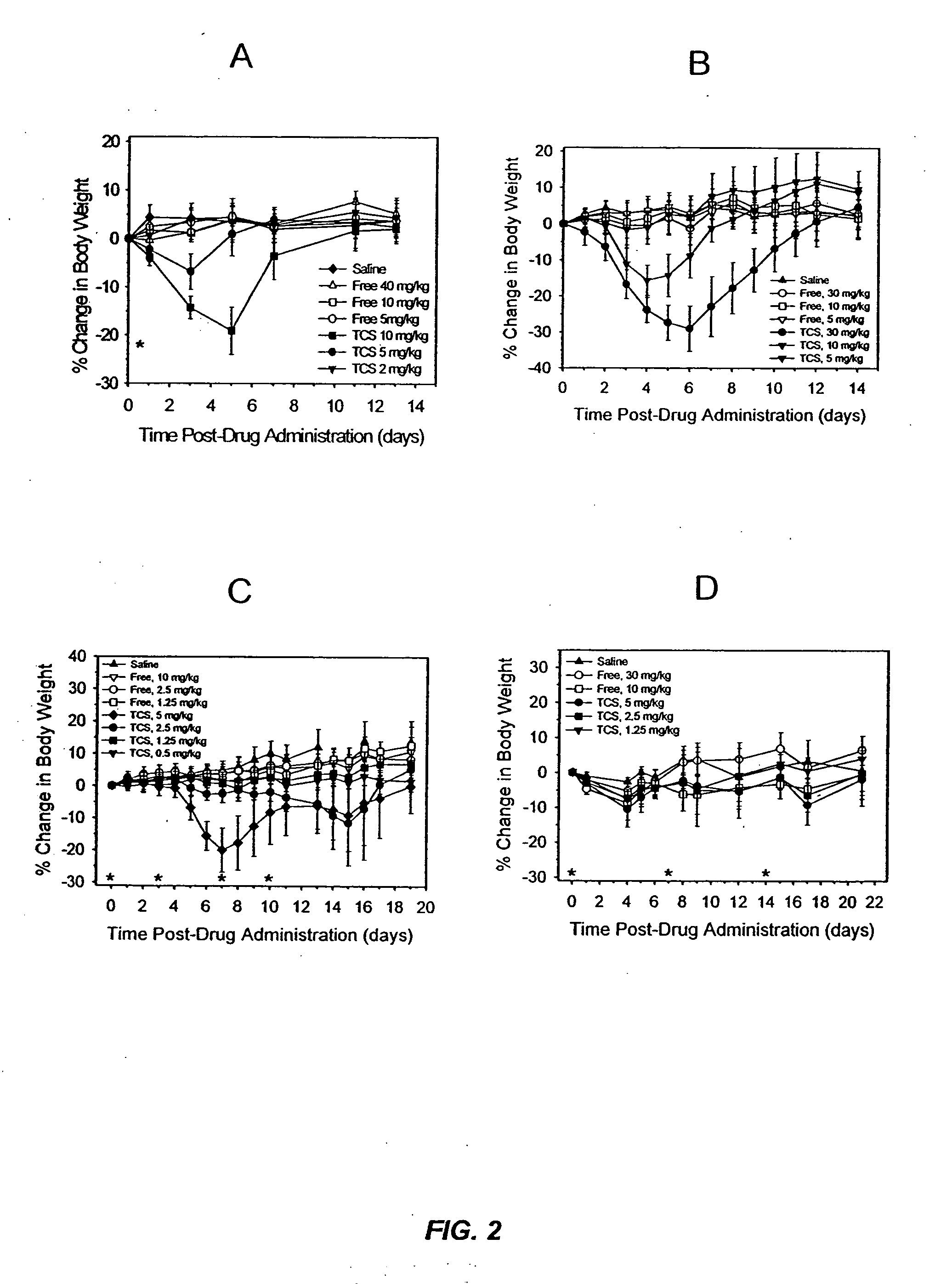
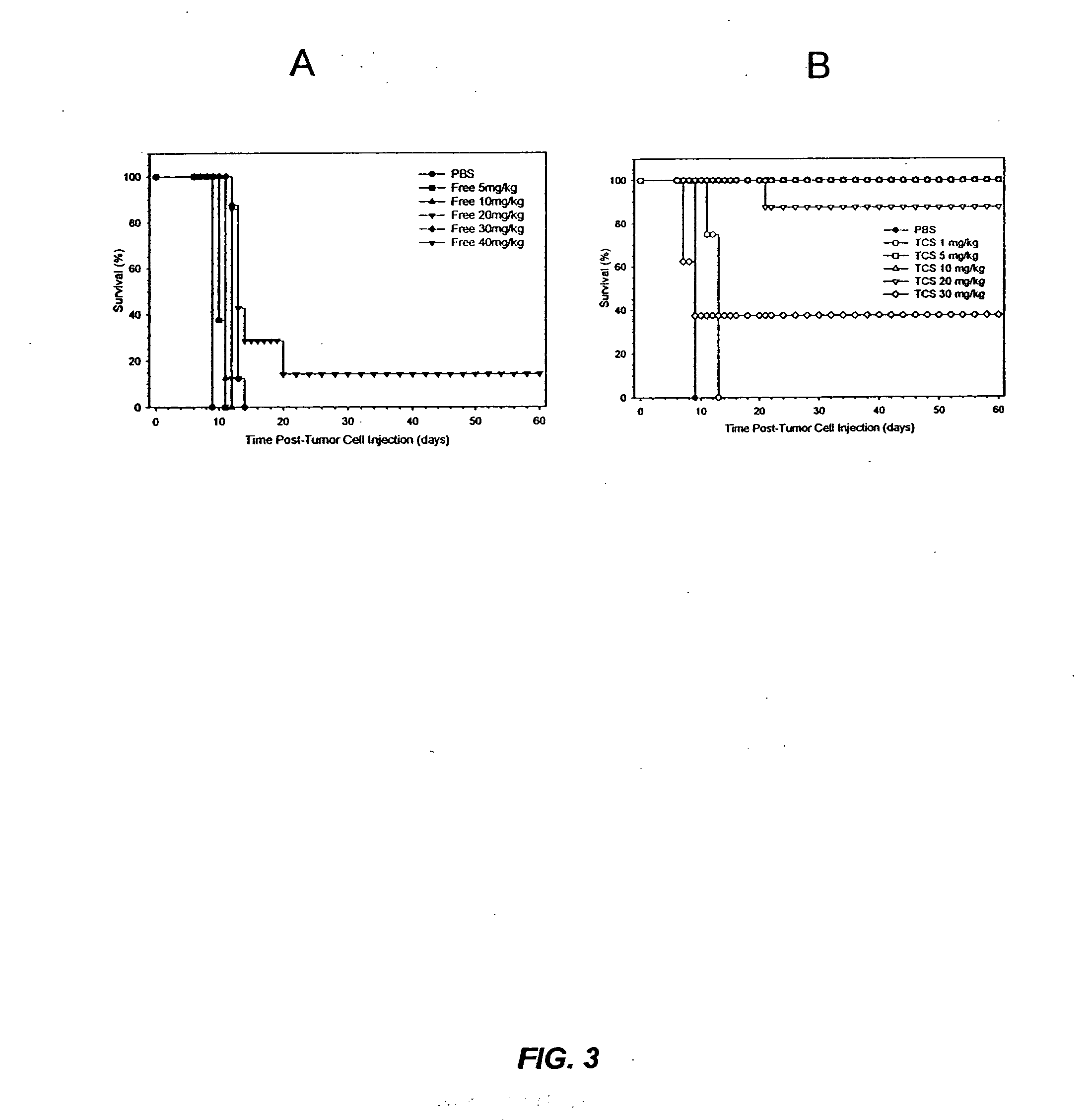
[0022] It has now been discovered that the anti-tumor activity of topotecan hydrochloride (Hycamtin™, SmithKline Beecham) encapsulated in sphingomyelin / cholesterol liposomes, such as sphingomyelin / cholesterol (55:45) liposomes, by a gradient loading method provides surprising clinical efficacy at lower doses, and with lower collateral toxicity, than free topotecan. The data demonstrates a dramatic increase in therapeutic index of liposome-encapsulated topotecan relative to the free drug. The present invention also provides a novel range of different dosages and dosage schedules, and a variety of drug:lipid ratio formulations of liposomal camptothecins, that are useful for treating solid tumors.
I. Compositions and Methods of Making Liposomal Camptothecins
[0023] Liposome, vesicle and liposome vesicle will be understood to indicate structures having lipid-containing membranes enclosing an aqueous interior. The structures can have one or more lipid membranes unless otherwise indicate...
PUM
| Property | Measurement | Unit |
|---|---|---|
| size | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More