Catalytic process for the preparation of fluorinated halocarbons
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Hydrofluorination of 1,2-dichloro-1,1-difluoroethane—without Rate Enhancing Reagent
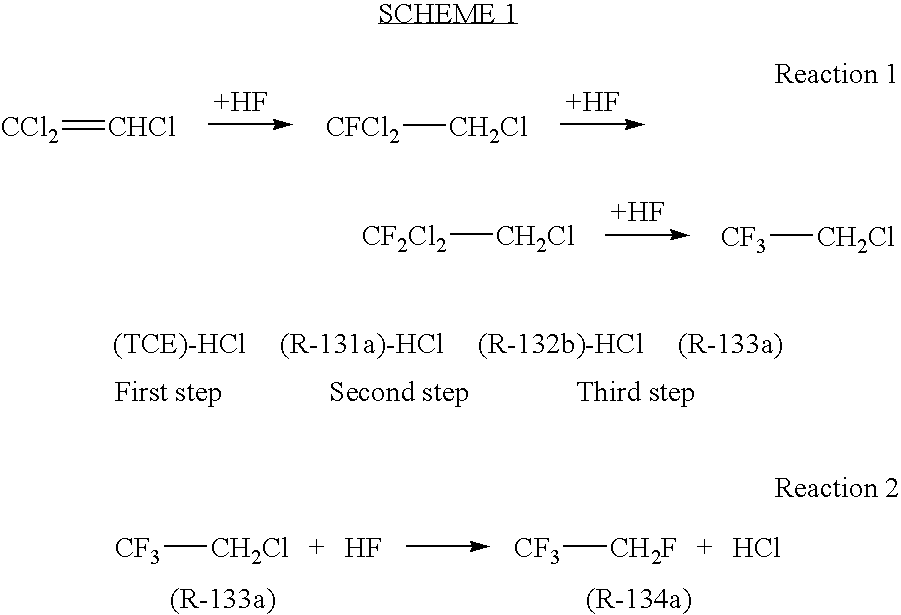
[0016]Seven Hundred and twenty milligrams (0.002 mol) of tantalum pentachloride (TaCl5) was charged into a 250 milliliter reactor. The reactor was evacuated and cooled with ice. Fifty grams (2.5 moles) of anhydrous hydrogen fluoride (HF) was next added to the reactor. The resulting solution was heated with stirring to 140° C. for 60 minutes. The reactor was cooled with ice and 13.4 grams (0.1 mol) of 1,2-dichloro-1,1-difluoroethane (R-132b) was added. The reactor was heated to 140° C. and samples were withdrawn from the reactor headspace. The reaction was monitored by GC. After 1.5 hours, a sample was analyzed. The yield of 2-chloro-1,1,1-trifluoroethane (R-133a) was 36.5%. A repeat example using the same amount of reactants under the same conditions produced a yield of R-133a of 22%.
example 2
Hydrofluorination of 1,2-dichloro-1,1-difluoroethane-15% Rate Enhancing Reagent Added
[0017]Seven Hundred and twenty milligrams (0.002 mol) of tantalum pentachloride (TaCl5) was charged into a 300 milliliter reactor. The reactor was evacuated and cooled with ice. Fifty grams (2.5 moles) of anhydrous hydrogen fluoride (HF) was next added to the reactor. The resulting solution was heated with stirring to 140° C. for 60 minutes. The reactor was cooled with ice and a mixture of 13.4 grams (0.1 mol) of 1,2-dichloro-1,1-difluoroethane (R-132b) and 2.2 grams (0.015 mole) 1-fluoro-1,1,2-trifluoroethane (R-131a) was added. The reactor was heated to 130° C. and samples were withdrawn from the reactor headspace. The reaction was monitored by GC. After 1 hour a sample was analyzed. The yield of 2-chloro-1,1,1-trifluoroethane (R-133a) was 87.5%.
example 3
Hydrofluorination of Trichloroethylene—without Rate Enhancing Reagent
[0018]In a 500 milliliter Hastaloy reactor was placed 7.2 grams of TaF5. The reactor was evacuated, cooled in an ice bath and 120 grams (6 mol) of anhydrous HF were added. The reactor was heated to 100° C. and 131 grams (1 mol) trichloroethylene were added all at one time. The reactor was then rapidly heated to 140° C. and maintained at this temperature for 6 hours. A constant pressure of 500 psi was maintained by venting the hydrogen chloride reaction product. At the conclusion of the reaction period, the contents of the reactor were vented into crushed ice. Analysis of the product obtained from the ice mixture gave a yield of 2-chloro-1,1,1-trifluoroethane of 80%. The residue in the reactor was extracted with 10% hydrochloric acid. The amount of residue was 13 grams and appeared to be a water insoluble oligomeric tar
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More