Neuroprotective peptides
a neuroprotective and peptide technology, applied in the direction of peptides/protein ingredients, drug compositions, peptides, etc., can solve the problems of neuronal cell death via acute and delayed damage processing, many potential neuroprotective agents many cpps also exhibit toxicity at low to moderate doses, etc., to reduce cell death and inhibit or ameliorate neurodamaging events/pathways. , the effect of reducing
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
experimental example 4
Animal Trial
[0233]In an initial animal trial that was conducted, it was shown that Arg-9 (R9), R18 and protamine (Ptm) possessed neuroprotective activity in vivo. These trials showed the efficacy of R9D peptide in rat permanent middle cerebral artery occlusion (MCAO) stroke model. R9D peptide was administered intravenously 30 min post-MCAO. Infarct volume (brain injury) was measured 24 h post-MCAO (mean±SEM). This is shown in FIGS. 5 and 27 where it can be seen that (Ns=812 animals for each group) treatment with R9D, R18 and protamine (Ptm) showed a statistically significant neuroprotective effect by reducing infarct volume (brain damage) by approximately 20% after a MCAO stroke.
General Observations and Discussion
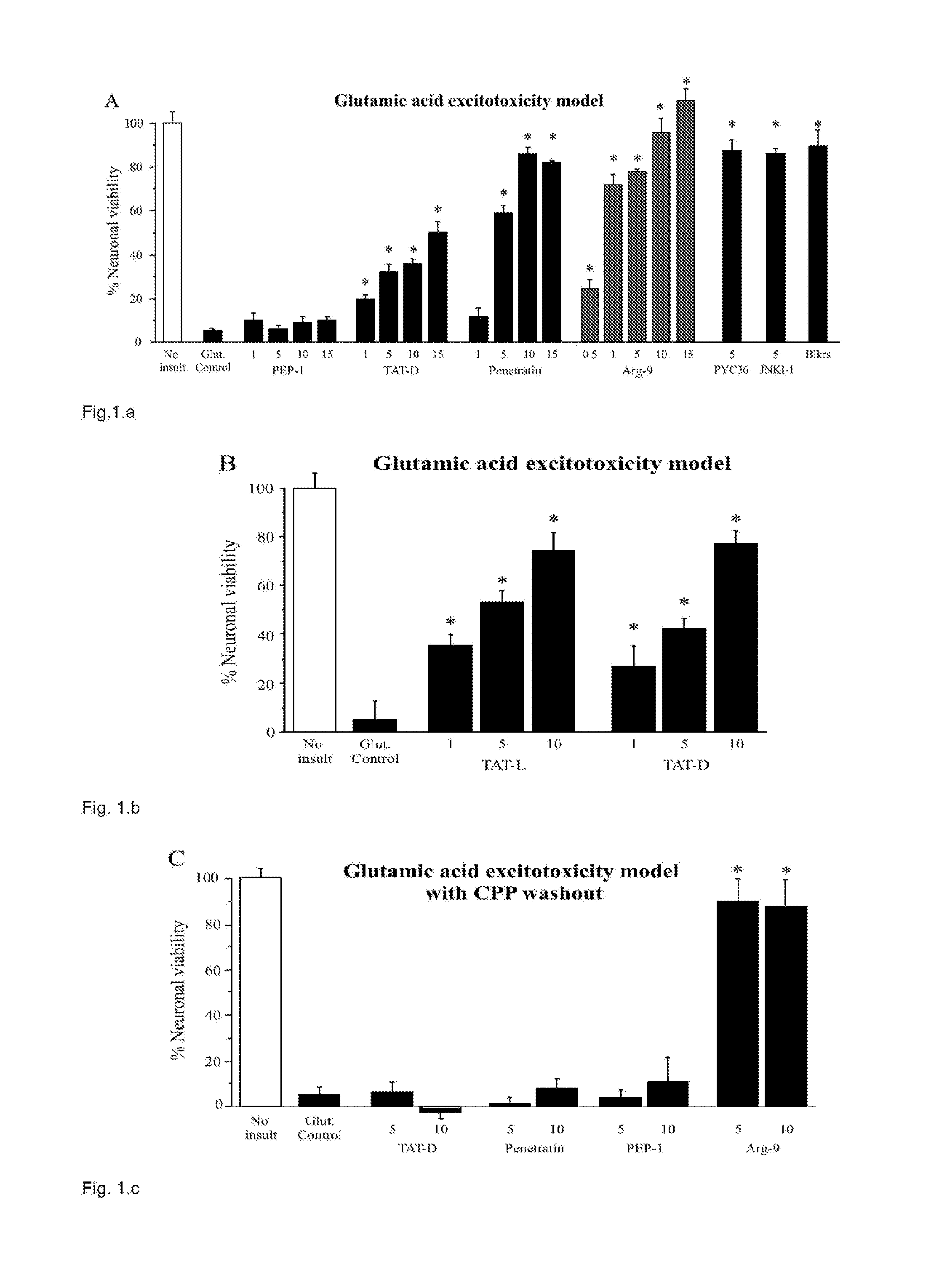
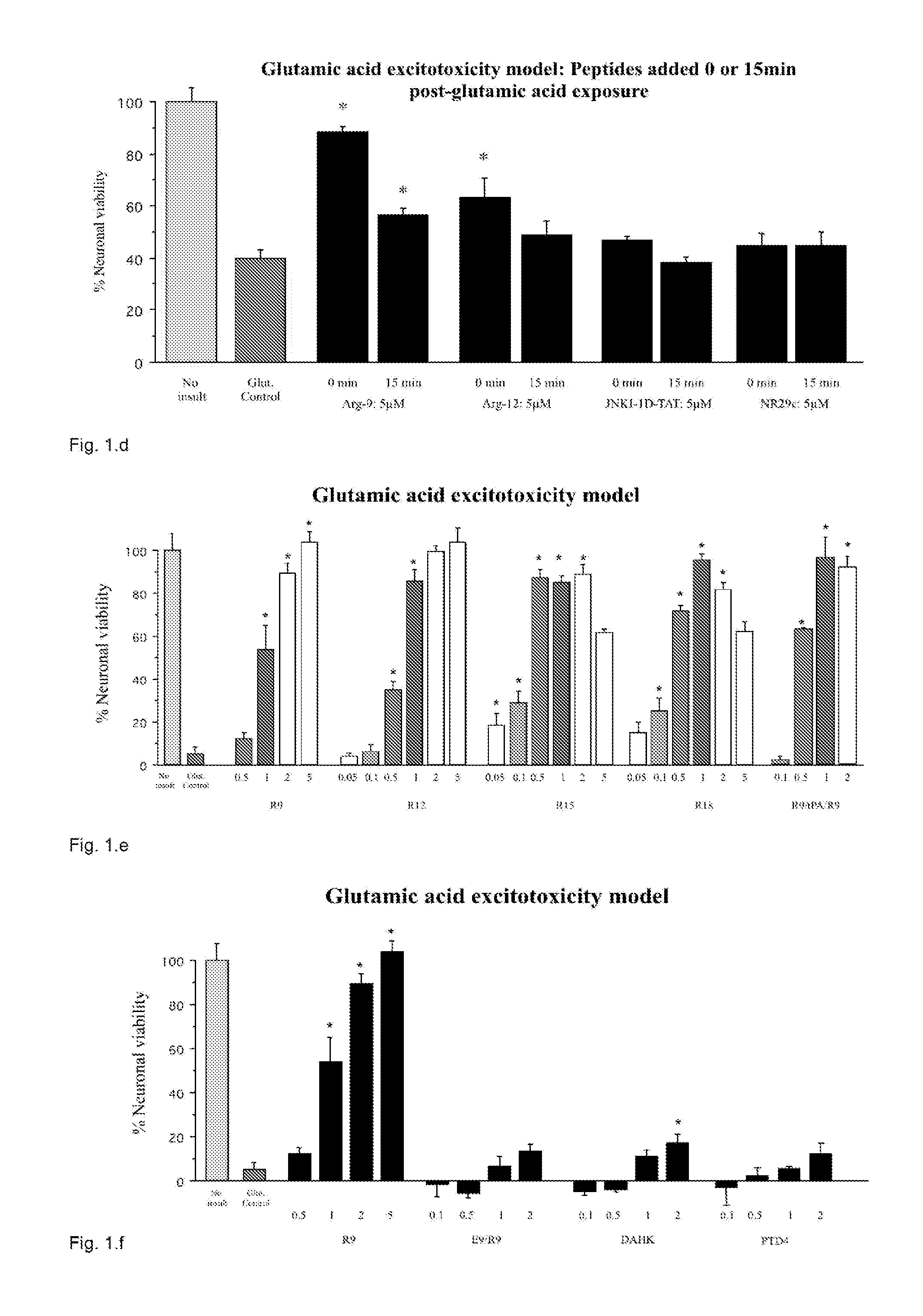
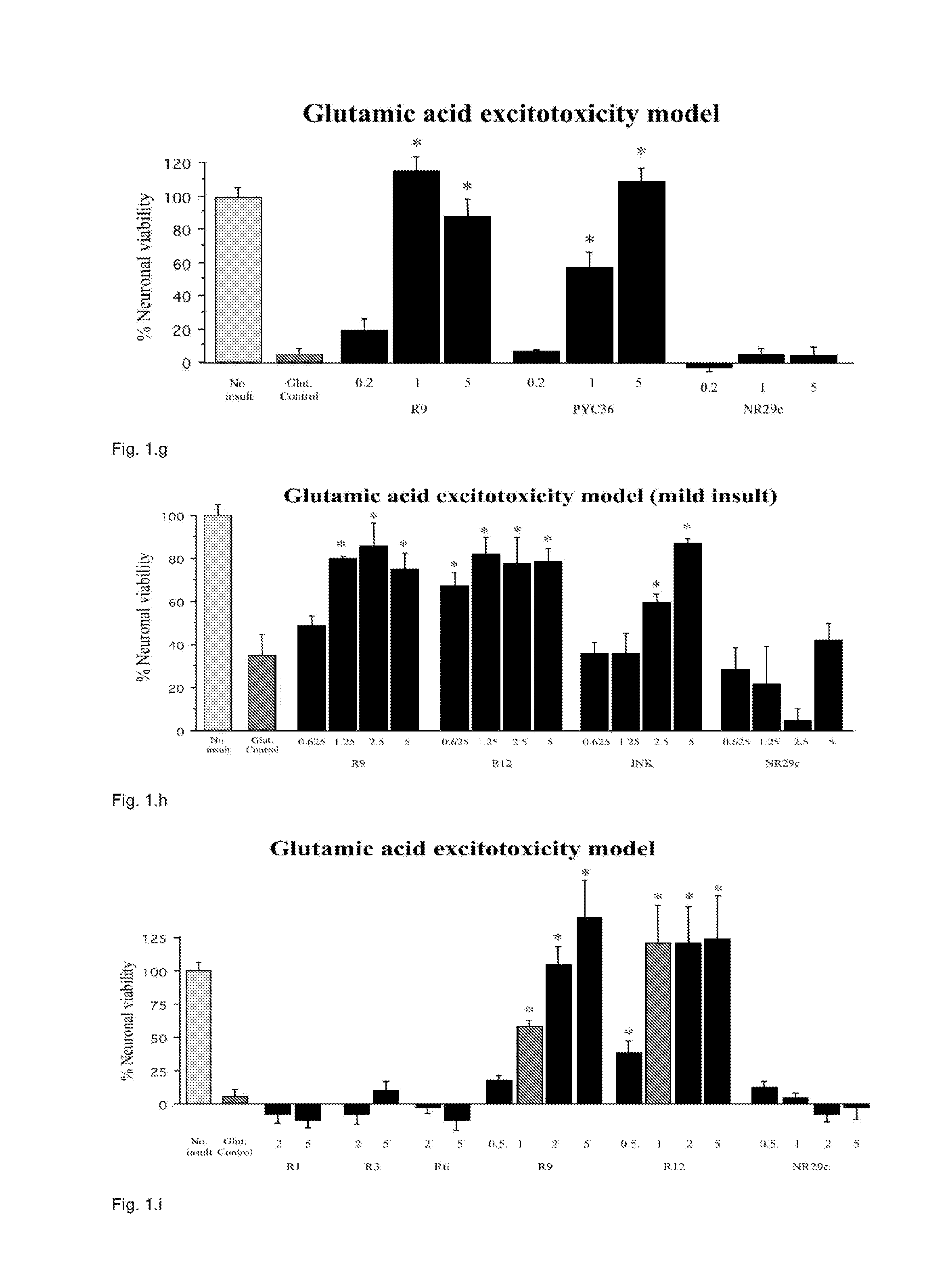
[0234]The Applicant assessed TAT as known neuroprotective example and three other CPPs (penetratin, R9, and Pep-1) for their neuroprotective properties in cortical neuronal cultures following exposure to glutamic acid, kainic acid, or in vitro ischemia (oxygen-glucose depri...
experimental example 1
Neuroprotection Following Glutamic Acid Exposure
[0255]Protamine sulphate (protamine; Ptm) provided significant neuroprotection in a dose response manner (FIGS. 9, 10, 12, 13, 15). Visual assessment of cultures post-injury also confirmed the neuroprotective effect that ranged from for untreated glutamic acid exposed cultures to 85-100%% survival for protamine treated cultures. In addition, a 5 or 10 minute protamine pre-exposure was also highly neuroprotective resulting 100% neuronal survival (FIG. 11). In dose response experiments, low molecular weight (LMWP), protamine 1 (Ptm1), protamine 2 (Ptm2), protamine 3 (Ptm3), protamine 4 (Ptm4), protamine 5 (Ptm5), peptide were also neuroprotective (FIGS. 12, 13, 15).
[0256]In protamine and LMWP pre-exposure experiments, protamine was neuroprotective when neurons where exposed to protamine immediately before and 1 or 2 hours prior to glutamic acid insult, while LMWP was only neuroprotective when exposure was immediately before glutamic acid...
experimental example 2
Neuroprotection Following Oxygen-Glucose Deprivation (OGD)
[0257]In the OGD model protamine was neuroprotective when neurons were treated with peptide 1 hour before insult (FIG. 18) or post-insult (FIG. 16, 17) In addition, when added for 15 or 30 minutes, post-OGD protamine was also neuroprotective (FIGS. 19, 20, 21, 22), LMWP was not neuroprotective when neurons were pre-exposed to the peptide 1 hour before OGD, but it was neuroprotective when added for 15 minutes post-OGD. In addition when protamine peptides (Ptm1-Ptm5) and LMWP were added for 15 minutes post-OGD, peptides Ptm2, Ptm4, Ptm5 and LMWP were neuroprotective (FIG. 22).
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More