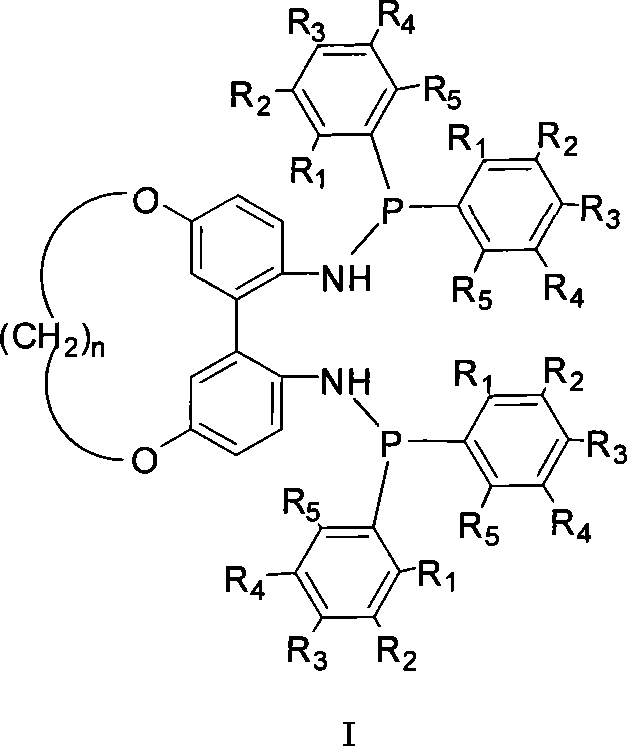
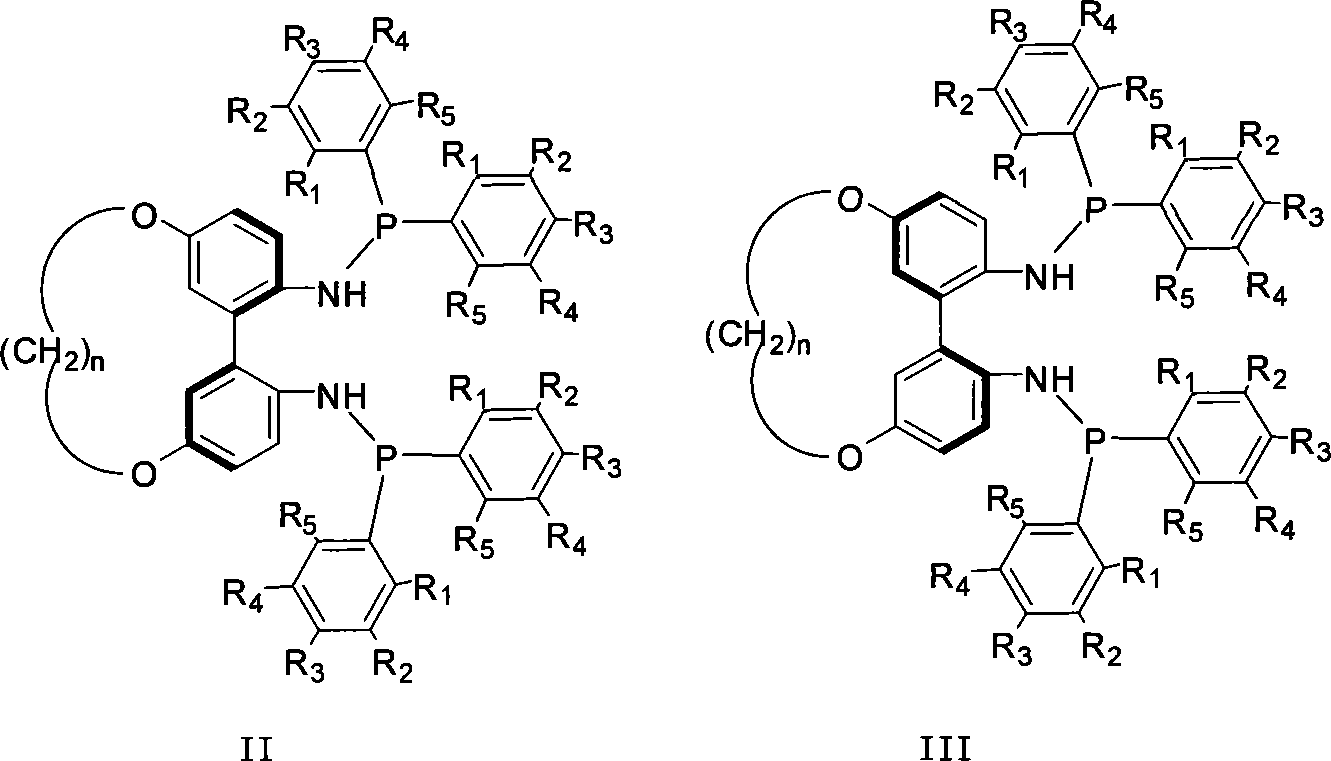
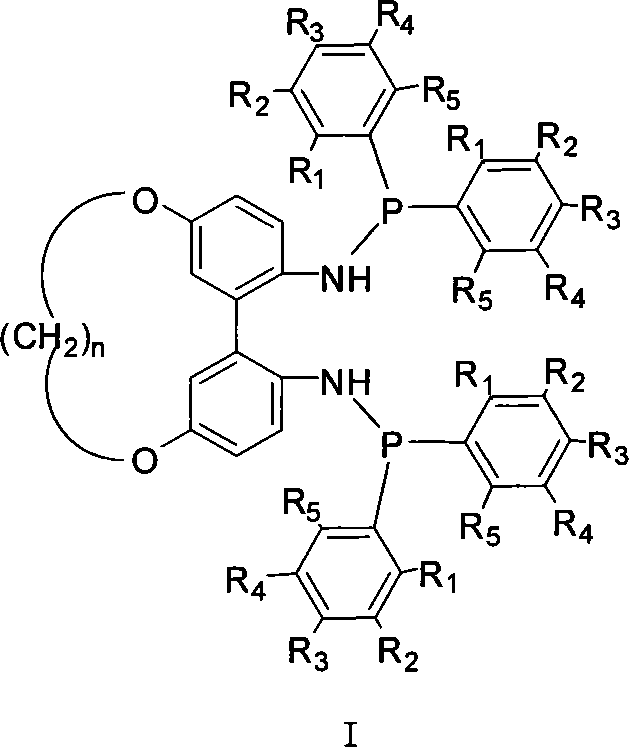
1,1'-biphenyls axial chirality diphosphinidene amide ligand connected at 5,5' position
A bisphosphonite, axial chirality technology, used in the chemical industry to achieve the effect of high reactivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0025] The embodiments of the present invention are described in detail below: the present embodiment is implemented under the premise of the technical solution of the present invention, and detailed implementation and specific operation process are provided, but the protection scope of the present invention is not limited to the following implementation example.
[0026] (1) Preparation of compound V from 3-bromo-4-nitroanisole
[0027] Compound VI (3.2 g, 13.9 mmol) and activated copper powder (3.6 g, 55.6 mmol) were heated to 170° C. for four hours. Washed with benzene and ethyl acetate, the solvent was evaporated, and the residue was recrystallized with acetonitrile to obtain product V (1.7 g, 80%).
[0028] 1 H NMR (400MHz, CDCl 3 ) 8.28 (d, J=8.8, 2H), 7.00 (dd, J=9.2, 2H), 6.70 (d, J=2, 8, 2H), 3.90 (s, 1H)
[0029] (2) Preparation of compound VI from compound V
[0030] Compound V (1.2g, 4.1mmol) and anhydrous lithium chloride (1.0g, 24.5mmol) were heated under re...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More