Method for catalytically oxidizing organic matter by solid superacid and by taking potassium ferrate as oxidizing agent
A technology of solid superacid and potassium ferrate, which is applied in the fields of oxidative preparation of carbonyl compounds, chemical instruments and methods, organic chemistry, etc., which can solve the problems of fast reaction speed, shortened reaction time, difficult oxidation reaction, etc., to improve oxidation activity , shorten the reaction time, improve the effect of reaction speed
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
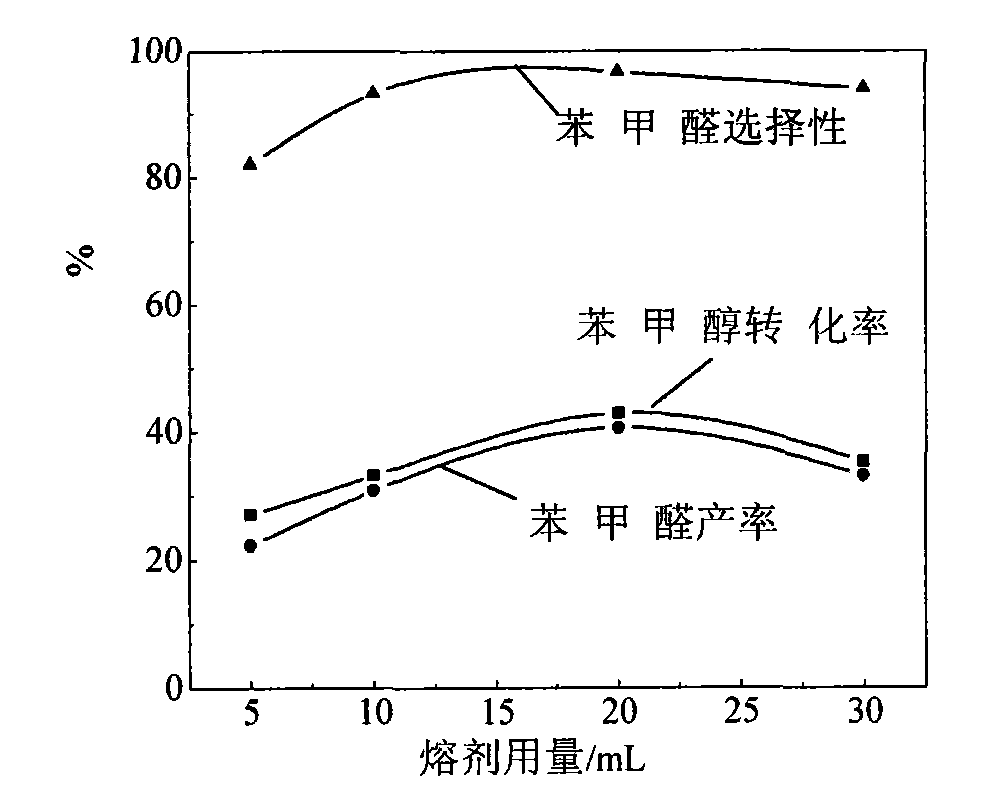
[0015] Embodiment 1: add solid catalyst SO in 50mL volumetric flask 4 2- / Al 2 o 3 -ZrO 2 0.5g, 0.2mL benzyl alcohol, 0.8g potassium ferrate, reaction temperature 30°C, reaction time 2h, change the amount of cyclohexane solvent, and investigate the influence of solvent amount. Filter reaction solution, filtrate is carried out chromatographic analysis, result is as table 1 and figure 1 shown.
[0016] Table 1
[0017]
[0018] Conclusion: As can be seen from the data in Table 1, the conversion rate of benzyl alcohol and the production rate of benzaldehyde first increased and then decreased with the increase of solvent consumption. When the solvent consumption was 20 mL, the conversion rate of benzyl alcohol and the yield of benzaldehyde were the highest.
Embodiment 2
[0019] Embodiment 2: add solid catalyst SO in 50mL volumetric flask 4 2- / Al 2 o 3 -ZrO 2 0.5g, benzyl alcohol 0.2mL, solvent cyclohexane 20mL, potassium ferrate 0.8g, reaction time 2h, change the reaction temperature, investigate the influence of temperature. Filter reaction solution, filtrate is carried out chromatographic analysis, result is as table 2 and figure 2 shown.
[0020] Table 2
[0021]
[0022] Conclusion: As can be seen from the data in Table 2, within a given temperature range, as the reaction temperature increases, the conversion rate of benzyl alcohol and the yield of benzaldehyde do not change much, and the law of change of the yield of benzaldehyde increases first and then decreases , when the temperature is 40℃, the yield of benzaldehyde is the highest.
Embodiment 3
[0023] Embodiment 3: add solid catalyst SO in 50mL volumetric flask 4 2- / Al 2 o 3 -ZrO 2 0.5g, 0.2mL benzyl alcohol, 20mL solvent cyclohexane, reaction temperature 40°C, reaction time 2h, change the amount of potassium ferrate, and investigate the effect of the amount of oxidant. Filter reaction solution, filtrate is carried out chromatographic analysis, result is as table 3 and image 3 shown.
[0024] table 3
[0025]
[0026] Conclusion: As can be seen from the data in Table 3, the conversion of benzyl alcohol increases rapidly as the amount of potassium ferrate increases. After the amount of potassium ferrate is 0.7, the conversion rate of benzyl alcohol increases slowly as the amount of potassium ferrate increases. . With the increase of the dosage of potassium ferrate, the benzaldehyde production first increased and then decreased. When the solvent dosage was 0.8g, the benzaldehyde yield was the highest.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More